

Chronic Muloskeletal Pain in Children:
Assessment and ManagementThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Rheumatology (Oxford). 2009 (May); 48 (5): 466–474 ~ FULL TEXT
OPEN ACCESS Jacqui Clinch, Christopher Eccleston
Bath Centre for Pain Services,
Royal National Hospital for Rheumatic Diseases,
Bath, UK.Paediatricians and paediatric rheumatologists review a large number of children with ongoing pain (disease and non-disease related). A small number of these develop chronic pain conditions that are complex and distressing. Over recent years studies of the epidemiology, aetiology and rehabilitation of pain and pain-associated disability in children have revealed a large prevalence of clinically relevant pain, and have emphasized the need for early recognition and intervention. Medication has a role when part of a multidisciplinary framework, although there is little evidence for or against the effectiveness of most pharmacotherapy. There is strong evidence to support early targeted psychological and physical intervention, and an understanding that parental education and involvement is essential if progress is to be maintained. In this review, an overview of the assessment is presented and management of childhood chronic musculoskeletal pain conditions with reference to main research findings to date.
Key words: Chronic pain, Children, Pain-associated disability, Diffuse idiopathic pain, Juvenile fibromyalgia, Complex regional pain syndrome, Localized idiopathic pain, Demedicalization, Pain education, Multidisciplinary rehabilitation
From the Full-Text Article:
Introduction
Paediatricians, particularly paediatric rheumatologists, review a large number of children who have a wide variety of musculoskeletal pains [1, 2]. Many of these children do not have easily identifiable inflammatory or other obvious disease processes that can help us understand and explain the cause of pain. However, for most young people presenting to clinic, regardless of whether the cause of their pain is known, the chronic experience of pain has often had a large and wholly negative impact on the physical and psychological well-being of the young person and their family [2]. Recent data have shown that chronic pain also has a serious negative impact on financial well-being, not only for each family but also for the economy as a whole. In the only published cost of illness study of this population we conservatively calculated the financial burden of adolescent chronic pain on the UK economy in 1 year, 2005, to be Ł3840 million [3]. Childhood chronic pain is a modern public health disaster. The extent of unacknowledged suffering and dependency in our healthcare systems is only now coming to light.
In this article, we have five related objectives. First, we review the epidemiology of musculoskeletal pain in childhood. Secondly, we address the current theories of the aetiology of pain. Thirdly, we review the impact of chronic pain, the clinical features of common pain presentations and their relevance to diagnosis and treatment planning. Fourthly, we introduce rehabilitation interventions aimed at the management of chronic pain. Finally, we make recommendations for further action.
Childhood musculoskeletal pain conditions
The most common chronic pain conditions reviewed in paediatric rheumatology settings include diffuse idiopathic musculoskeletal pain (juvenile fibromyalgia), chronic pain related to childhood hypermobility, complex regional pain syndromes (CRPS), chronic back pain and persistent joint pain following previous or controlled inflammation [e.g. juvenile idiopathic arthritis (JIA)].
Epidemiology
There is a historical lack of data on the prevalence and incidence of pain in youth, and on the prevalence of pain-associated suffering. We know, however, that pain is a common feature of childhood. One study showed that 83% of the school-aged children had experienced an episode of pain during the preceding 3 months [4]. Pain is a normal sensation but becomes disabling when it persists and is associated with suffering. In this same study sample, 30.8% of the children and adolescents stated that the pains had been present for over 6 months. Musculoskeletal pains accounted for 64% of all the pains that were reported [4]. Other studies support this finding [5–7]. Whether there is more chronic pain, or whether children with chronic pain are presenting more frequently, is not known. What is known is that there is significant unmet need.
Looking across studies a number of features are clear. First of all, there is a robust finding that girls experience more pain that boys [8–11] (although note that the incidence in boys may be increasing [4]). Children living in low-educated, low-income, families have an ?1.4-fold increase in the odds of having pain [9]. The incidence of chronic musculoskeletal pain peaks at the age of 14 years. A recent large cohort study showed that multiple common symptoms in childhood are associated with a moderately increased risk of chronic widespread pain in adulthood [12].
Clinical features of chronic childhood musculoskeletal pain
It is not unusual for pain to start in a localized area of the body [13]. The pain may intensify and radiate to other areas. It is associated often with a reluctance to mobilize, an avoidance of movement. Discomfort and pain intensity increase and become constant. As discrete pains multiply and continue, the young person may avoid contact with, or use of, an area of the body affected, which can lead to muscular spasms, abnormal posture and gait, and with chronic avoidance a greatly reduced fitness. Such a pattern of avoidance can lead to fear of pain and movement, and a resultant amplification of the pain.
In some circumstances pain has a direct affect on other systems, leading to symptoms that can be as disabling as the pain itself. These include:
Hypervigilance and hypersensitivity Children often report a heightened awareness of pain and pain-associated cues [1, 2]. It is unclear whether this is caused by fear of pain, or hypersensitivity of pain receptors in allodynic and hyperalgesic ranges. Clinically this presents as young people describing unbearable pain on minimal skin contact, and heightened fear of being touched, for example, on examination.
Perceived thermodysregulation This is more common in adolescent girls. Limbs are particularly cool and mottled. Occasionally there will be areas that are very red and hot to touch on a background of the mottled skin. There may also be an abnormal perception of temperature [14] with an increase in thermal pain sensitivity [15].
Autonomic dysfunction Pain is a powerful stressor. Continuous pain signals, immobility and fatigue act directly on the autonomic system [16]. In an environment of physical and emotional anxiety, the sympathetic system is more active. This leads to tachycardia, hyperventilation (compounded with panic attacks), cold sweats, blurred vision, abdominal pain and extreme pallor. Girls particularly complain of nausea, dizziness and episodes of feeling faint. Children look unwell during these episodes of increased pain. It is not unusual for attending paediatricians to investigate cardiovascular, neurological and gastrointestinal systems in an attempt to elicit pathology.
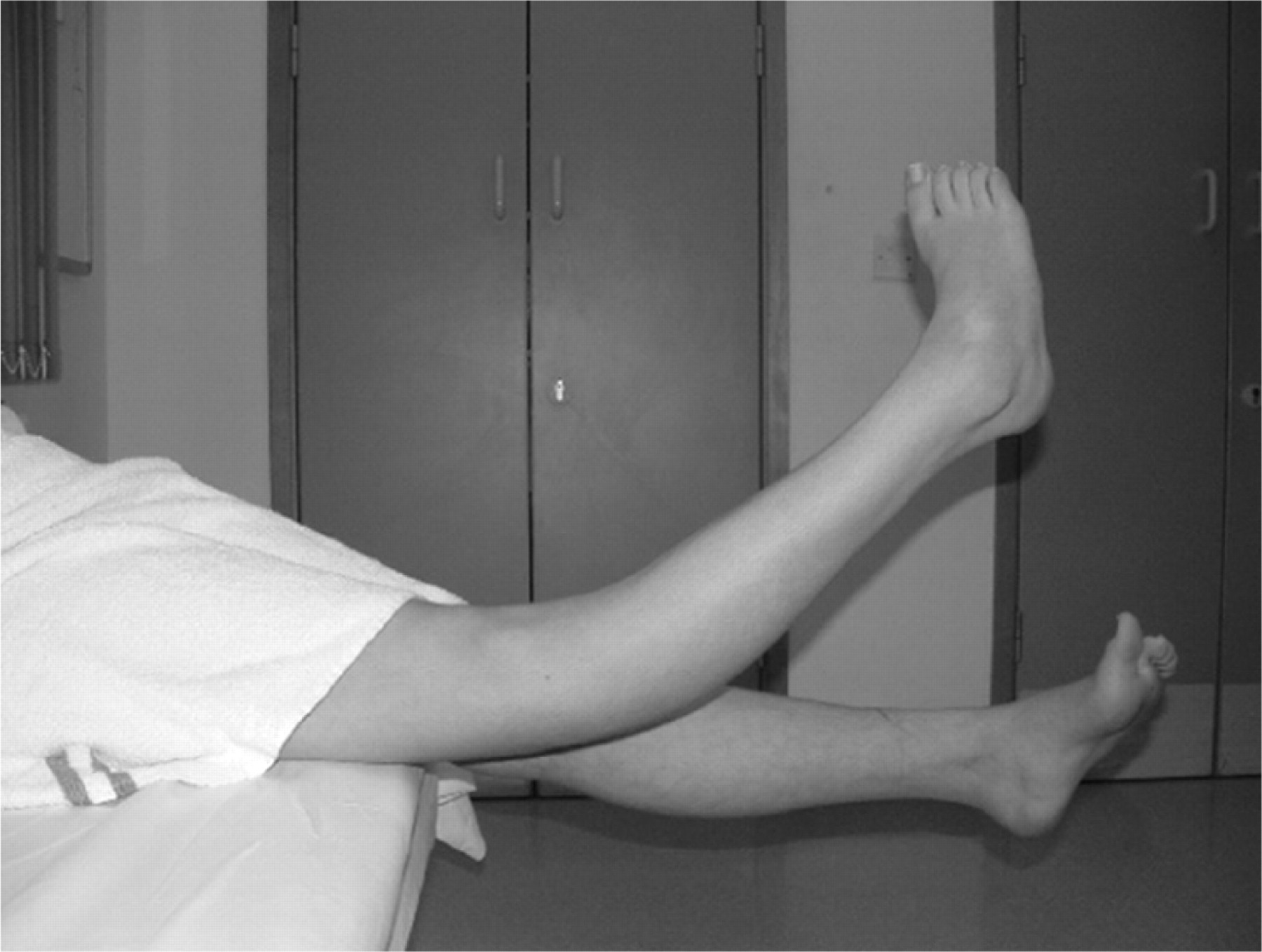
Musculoskeletal disequilibrium These young people are still growing, often in their peripubertal growth spurt, and this can have lasting effects on the final positioning. Proprioceptive signals from the joints are reduced and the limb held in a rigid, fixed position. Legs may ‘give way’. Knees and hips are held flexed, feet are inverted and hands are clenched with flexed wrists. These positions are often described as the most comfortable. Muscles and tendons quickly tighten and this complicates the pain and disability. The adaptive positioning of a young person with leg or abdominal pains particularly affects the gait and resting positions and thus alters the loads on the spine and pelvis [17]. Figures 1 and 2 show early, and then complex positional effect of CRPS. During the growth spurt the malpositioning and hyperextension of the right leg became significant.
There are a number of influences on physical behaviour, including the psychosocial. What can first appear as a structural anomaly or physical constraint may be better explained by fear of pain on movement, or postural compensation due to overprotection of a painful body part. There is very little research in this important area of the effects of a chronic stressor such as pain on a critical period of physical development, or in the measurement of movement constrained by pain.
Figure 1. Shiny, mottled appearance of left leg
indicative of changes seen early in CRPS.
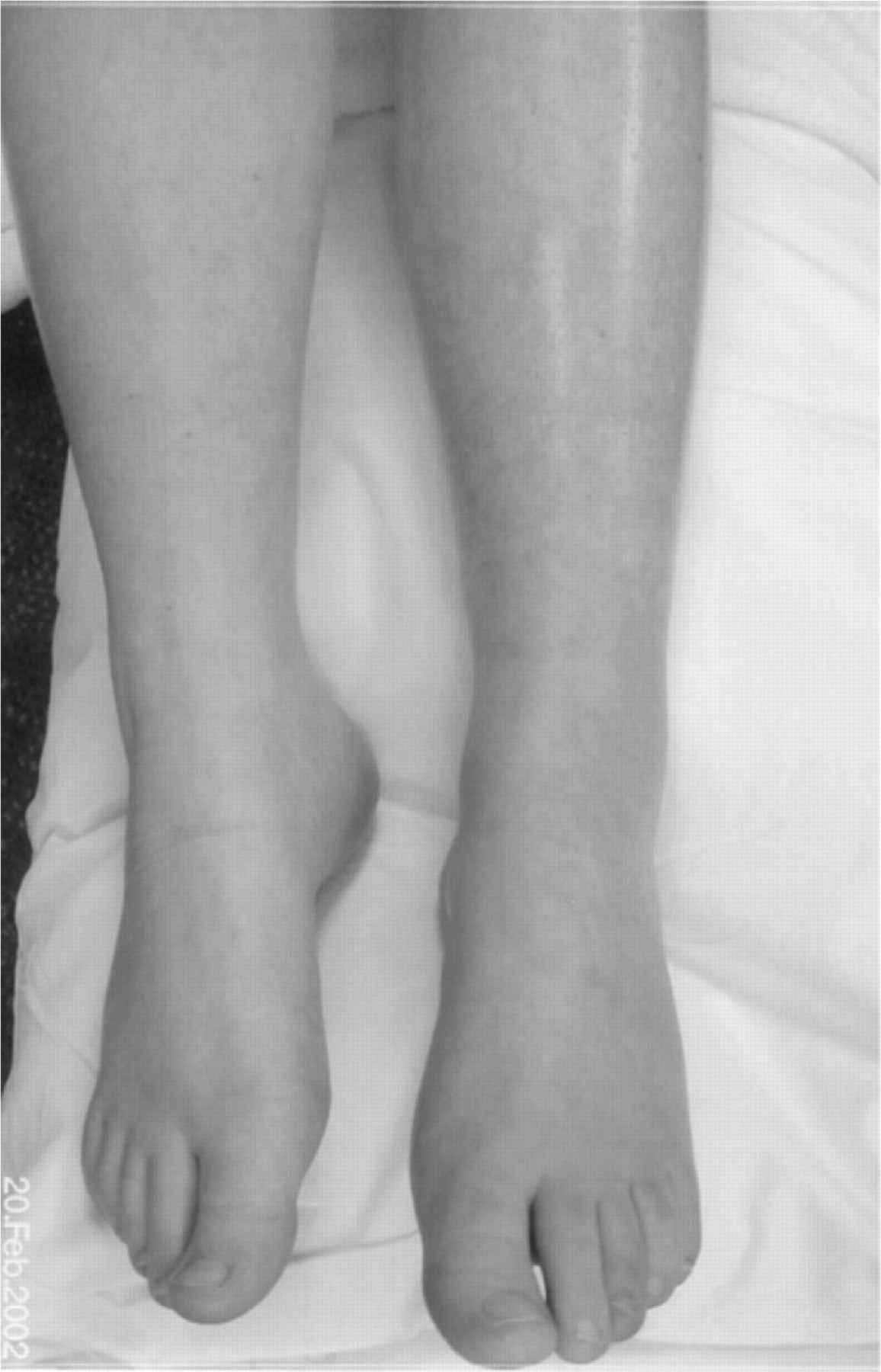
Figure 2. Extreme hyperextension of the right leg
in a child with a 3-yr history of CRPS.
Specific childhood musculoskeletal pain conditions
Diffuse idiopathic pain syndromes (juvenile fibromyalgia)
The onset of pain in diffuse idiopathic pain syndrome (DIP) [18] is often gradual. There may have been an initial insult (infection) or hypermobility [19] but often there is no obvious trigger and only vague recollections of the time of onset. The pain is generalized. There may be areas of allodynia and hyperalgesia but there is often an absence of the autonomic changes that we see in more localized pain conditions [2]. What is striking in the young people with diffuse pain is the fatigue, poor sleep pattern and extremely low mood. It is widely believed, however, that the low moods in adolescents are reactive (to the pain-associated disability) rather than a primary depression [20]. This is in contrast to adults with fibromyalgia where primary depression is frequently seen [21].
The adult fibromyalgia criteria should be viewed with caution [20]; children have a better outcome and there has been no definitive work looking specifically at tender points in this group.
CRPS
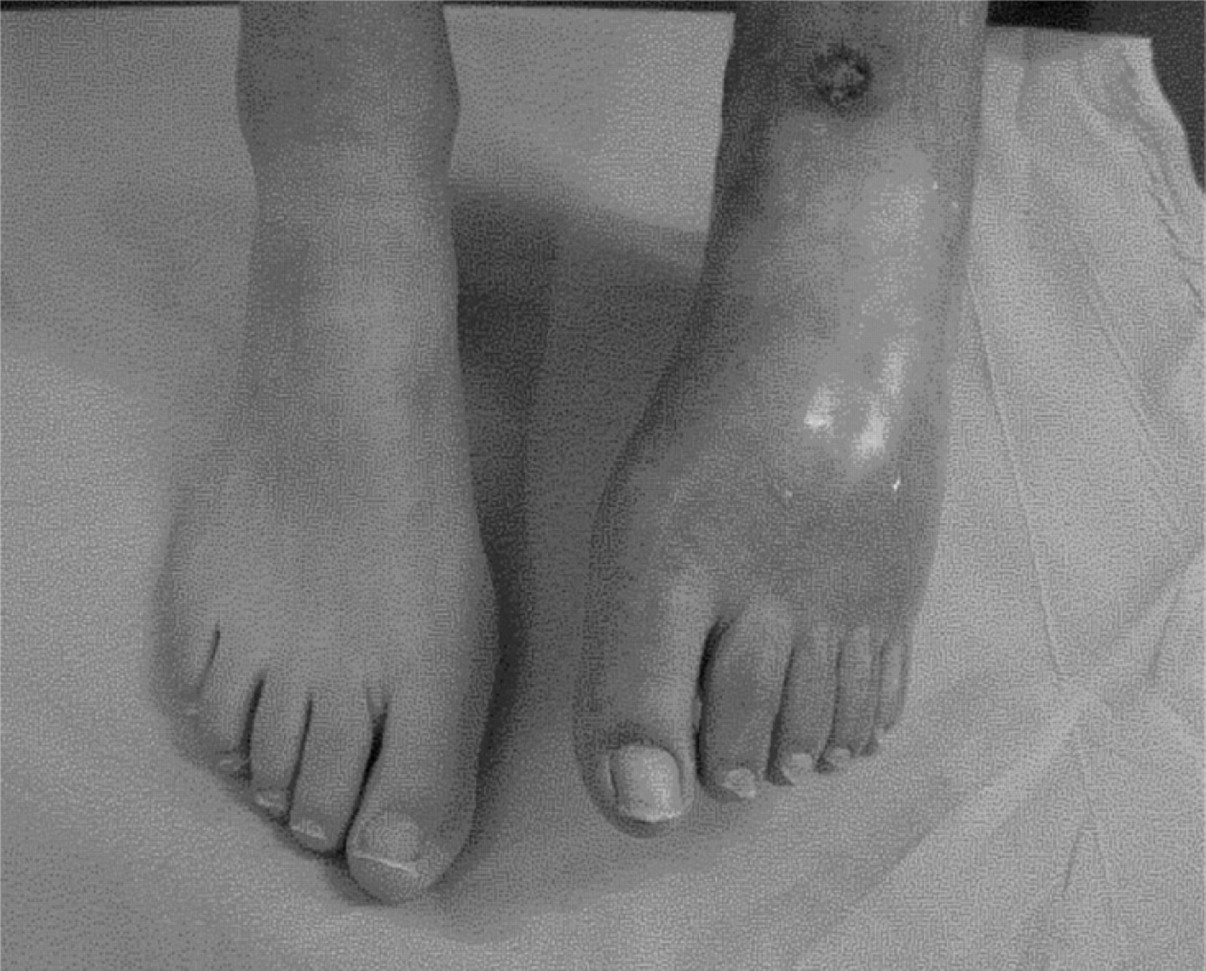
The diagnosis of complex regional pain syndromes (CRPS) remains a clinical one. There may be precipitating trauma. In children, the lower limb is much more commonly involved than the upper limb. The pain is usually out of proportion to the inciting event and accompanied by allodynia. Autonomic changes are present; these include swelling, reduced cutaneous perfusion and thermodynamic instability. There is also a marked reduction in range of movement and, in rare severe cases, trophic changes. Figure 3 shows the marked changes on thermography of the wasted, flexed, ulcerated foot that is shown in Figure 4.
Figure 3. Thermography shows poor cutaneous
circulation in affected left foot (barely visible).
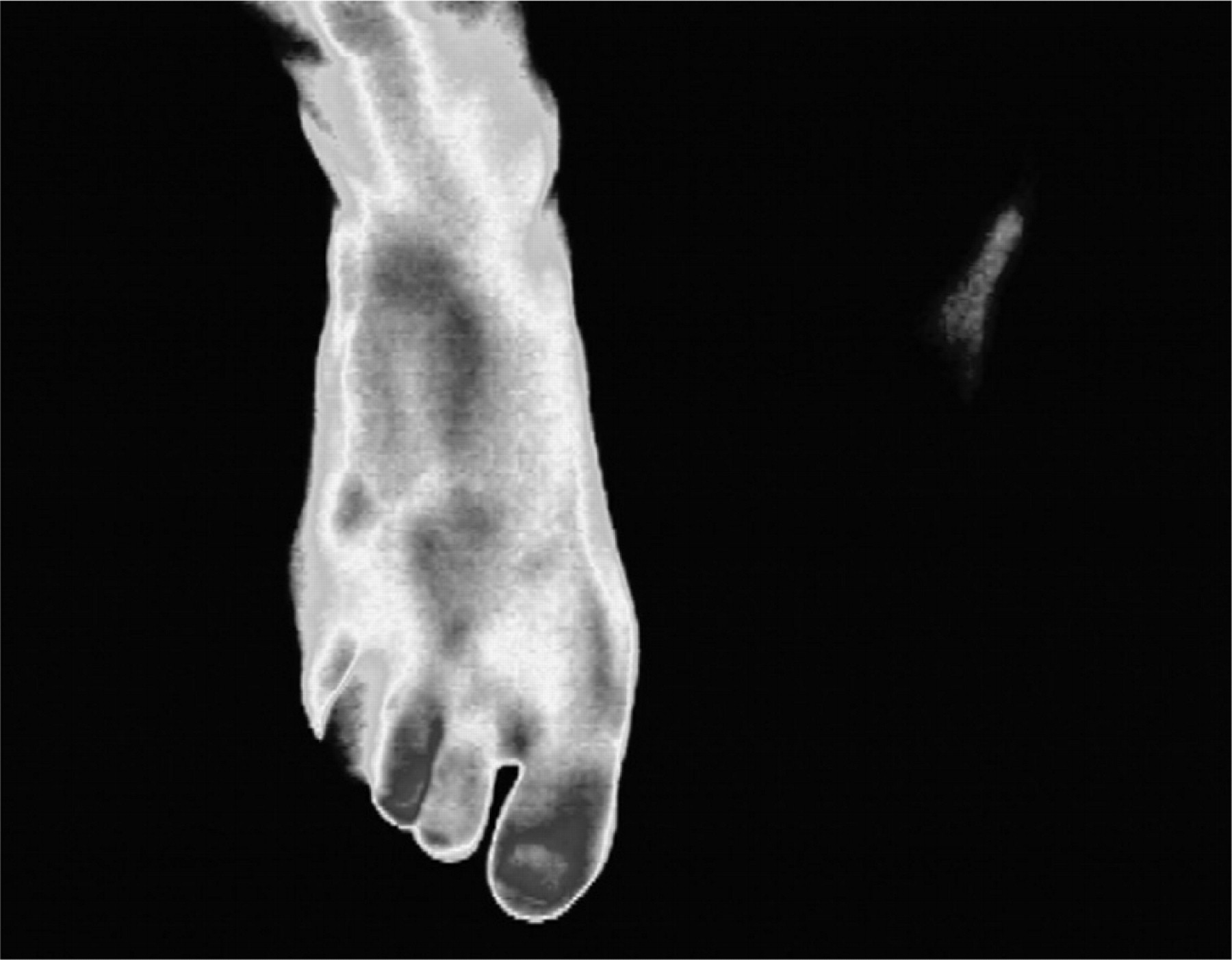
Figure 4. Severe CRPS with trophic changes
and ulceration.
The International Association for the Study of Pain (IASP) has diagnostic criteria for adults with CRPS. Although these diagnostic criteria hold true for children and adolescents, it is widely believed that the dystrophic changes and long-term disability are less common when compared with adults.
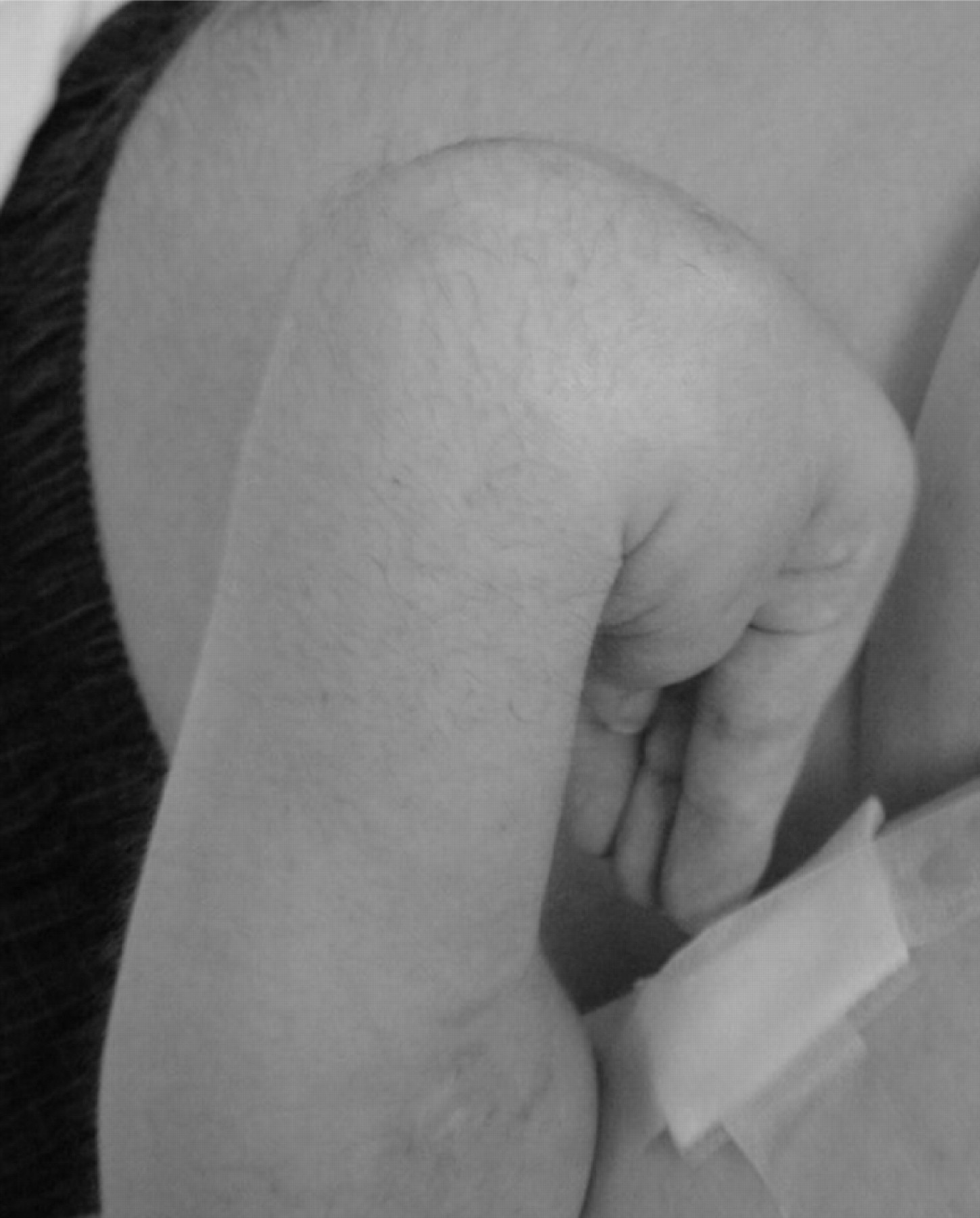
Occasionally more than one limb may be affected at presentation. It is not unusual for a hand or other leg to develop CRPS months after a leg has been affected. This may be due to the use of crutches and subsequent pain amplification but may also have no obvious trigger. Figure 5 shows the fixed, flexed position adopted after a mild injury.
Prior to the 1970s CRPS was seldom reported in children. More recently, however, hundreds of paediatric patients have been reported [22–24]. CRPS is seen most commonly around puberty although there are a few cases reported below the age of 10. CRPS type 1 is more commonly seen in girls with a ratio of 4 to 1 (female to male) [24].
Figure 5. Fixed flexion following minor injury.
Juvenile hypermobility and pain
There is undoubtedly an association between joint hypermobility in children and diffuse pain [19]; however, the nature of this is poorly understood.
Hypermobility or ligamentous laxity of joints is increasingly recognized as an aetiological factor in the presentation [25]. Such conditions include recurrent lower-limb arthralgia, anterior knee pain syndromes and back pain. In a recent study, a significant number of these children were shown to suffer recurrent joint sprains, actual subluxation/dislocation of joints, problems with handwriting tasks, limitations of school-based or other physical education activities, and having missed significant periods of schooling because of symptoms [26]. However, much disagreement remains as to the validity of hypermobility as an aetiological factor. What seems clear is that not all hypermobile individuals will be symptomatic or indeed possibly have any risk for specific musculoskeletal disorders in later life. Screening tools such as the Beighton score are likely to be inadequate in children as they are generally more mobile than adults. The challenge remains to interpret symptoms correctly as being related to the hypermobility and to predict why such children become symptomatic. The answer is likely to involve physiological and psychosocial factors. In addition, early identification and modification of risk factors may have major implications for subsequent prevalence of many adult chronic pain conditions.
The delay in diagnosis can result in poor control of pain and disruption of normal home life, schooling and physical activities. Knowledge of the diagnosis and simple interventions are likely to be highly effective in reducing the morbidity and cost to the health and social services.
Back pain
Low back pain specifically is commonly reported in the adolescent population. In a recent cross-sectional study the prevalence of low back pain that lasted for at least 1 month was 24% [27]. Often this is thought to be related to lifestyle influences on a developing spine such as postural habit (slouching), load bearing on the back (e.g. school bags) or engagement in sedentary activity (e.g. computer use). There are insufficient data to bring clarity to these arguments although it is fair to say that there is no conclusive evidence for any one of these factors being significantly related to the increase in back pain. As with adult back pain, if there are any red flags (such as nocturnal pain) in children then these should be fully investigated prior to rehabilitation.
Childhood disease and chronic pain
Diffuse and localized chronic pains can also complicate almost any other chronic childhood illness [28] including juvenile arthritis, IBD [29], cerebral palsy, cancer [30], sickle cell disease [31], muscular dystrophies [32] and cystic fibrosis [33].
The relationship between juvenile arthritis and chronic pain is well recognized [34]. As with other diseases the degree of disabling pain does not always mirror inflammatory joint activity. A growing body of research in rheumatic diseases, such as JIA, highlights the importance of environmental and cognitive behavioural influences in the pain experience of children in addition to the contribution of disease activity.
The aetiology of chronic pain in childhood
Idiopathic pain syndromes, in adolescents, seem to be related (either singularly or in combination) to illness, injury, psychological distress and environmental factors [2]. Children reporting diffuse pain conditions may have a history of a preceding childhood illness, such as glandular fever, tonsillitis and influenza.
Procedural pain
Although there is some evidence that early repeated exposure to invasive medical procedures that typically involve tissue cutting or puncture is a risk factor for lowered threshold to pain [35] there is, to date, no evidence to suggest that any of the common childhood procedural pains, such as on immunization, is a risk factor for adult chronic pain.
It is difficult to know whether a primary infectious or inflammatory condition (such as JIA) has an effect on the evolution of chronic pain or whether the pain is a consequence of the immobility, medical therapies and environmental changes that are associated with these conditions, or a combination. The research has not been undertaken.
Injury
It is not unusual for a child with a localized chronic pain to recall a sporting injury, operation or other trauma around the onset of chronic pain [36, 37]. Hypermobility has also been associated with falls and subsequent pain problems. There may be a period of enforced immobilization that can be an additional factor in the development of a chronic pain syndrome.
Psychological influences
There is no evidence for purely psychologically generated pain conditions in children. Although there are case reports of conversion neuroses manifesting as widespread pain complaints, this is highly unusual. There is evidence for psychosocial influences as complicating factors and as being implicated in the maintenance of pain complaints [38] but not their aetiology [39–43].
Genetic influences
There is emerging evidence that patients with CRPS may have a genetic predisposition [44] in Caucasian women, but the underlying genomics are far from clear. There have also been reports of chronic idiopathic musculoskeletal pains in siblings [45–47] and parent–child pairs [48], but this field is in its infancy and we are far from having information that is clinically useful.
Environmental influences
Although there is no strong evidence for the intergenerational transmission of pain and pain-related behaviour [49] there is a lack of study of common environmental risk factors other than social learning. Social history may show a recent life event (moving house/school, illness or death) [43, 44, 50] that has relevance to the presentation.
Developmental influences
There is current interest in the neuropathic mechanisms of paediatric pain. Considerable post-natal development occurs within nociceptic pathways over the first few post-natal weeks [51]. Interruption of this development may have a direct role in the later susceptibility of a child to suffer neuropathic pain. Neonatal pain experience may also have a role in long-term alterations in pain processing and development, with a greater tendency to amplified pain behaviour as a child [52]. There has been interest in the preliminary findings that pain sensitivity may be higher in adolescents who were born prematurely [53].
CRPS in paediatric patients is clinically distinct from the adult condition. In children, there is often complete resolution of its signs and symptoms within several months to a few years. With functional MRI it has recently been shown that considerable CNS circuitry changes take place in young people with a diagnosis of CRPS [54].
Pain sensitivity varies substantially among humans. It has recently been shown that catecholamine-O-methyltransferase (COMT) activity substantially influences pain sensitivity, and the three major haplotypes determine COMT activity in humans that inversely correlates with pain sensitivity and the risk of developing a chronic pain condition [55].
The impact of chronic pain on the child and family
Persistent or recurrent chronic pain brings persistent and recurrent distress, disability, adult attention and widespread family disruption [5]. Most families have relatively successful mechanisms for dealing with short-lived demands or disruption. However, a young person with chronic pain typically demands sustained physical, emotional and financial resource [3]. Young people with chronic pain report sleep disturbance, disordered mood, appetite disruption, low feelings (depression is often masked in this population), social isolation and unwelcome dependency on parents. All of these are experienced chronically and can serve to maintain pain and disability. They are also difficult to manage in isolation from other symptoms. It can be useful to graphically represent this with patients and others as shown in Figure 6.
Figure 6. A diagrammatic representation of the widespread impact of
chronic pain (after Bursch et al. [97]) illustrated with typically child quotations.
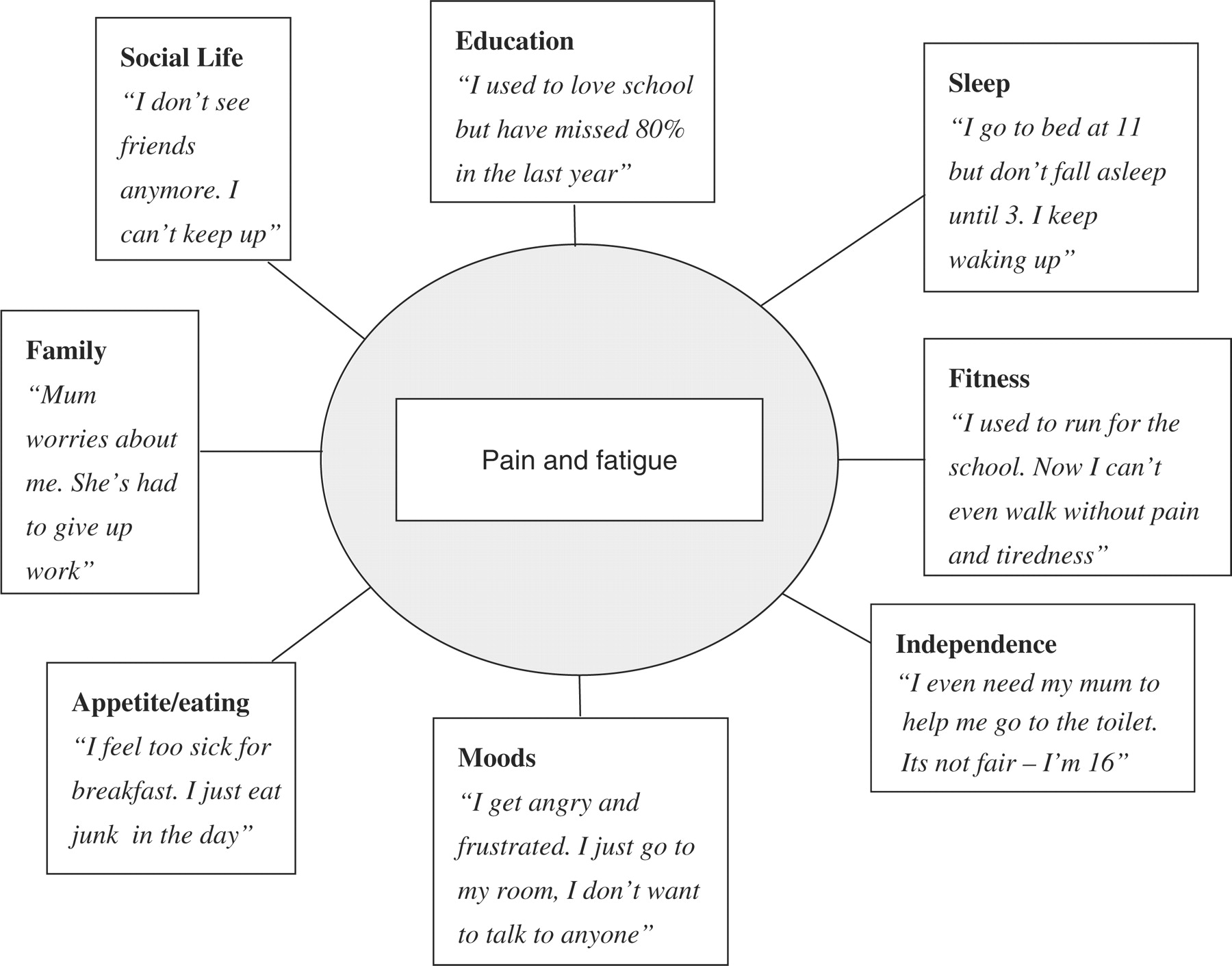
It is important to recognize that chronic pain is likely to bring severe disruption to the social and family structure [5]. Humans are highly organized and habitual creatures. Attention to altered routines can often yield relevant information. Children with chronic pain are commonly outside of formal education systems, absent from normal schooling [4]. The routines of normal school, rigidly applied, are often unmanageable for children who experience severe pain when sitting, concentration and memory difficulties, interpersonal problems in explaining confusing problems to peers, frequent anxiety-provoking ‘emergency-events’ associated with pain and time away for hospital visits.
Related to this is the social developmental context in which children are learning to live with chronic pain. Adolescence is by definition a time of change and experimentation. A recent study found that adolescents with chronic pain report that they are less socially developed on virtually every metric than their peers. Strong peer relationships were an important factor in inoculating children from further emotional distress and maladaptive coping [56].
Parents also report significant distress from living with a child in persistent pain. Levels of parental stress are clinically significant [57] and parents experience severe distress and conflict in parenting their child. Typically they report struggling with the desire to cure their child's pain, and comfort their child, recognizing that the desire to protect may be counterproductive [58].
Assessment of the young person (and family) with chronic pain
It is important to exclude significant disease. Common clinical behaviour is to be conservative in approach. Although this is necessary, it can also lead to a diagnostic vacuum and lack of treatment planning that allows for fear, disability and depression to grow. With serious medical causes excluded one can then work to move on to de-medicalize and rehabilitate safely. In paediatrics this is, in practice, more difficult as there can be a tendency to catastrophize about persistent pain by the family, and sometimes by the physician. The need to find a cause for the pain can be overwhelming and, in this way, the child may be overmedicalized.
History
Table 1 summarizes the important areas that should be covered when taking a history from a child and their carers.

Table 1 Ostensibly the features of the history taking, such as previous medical history, specific history leading to pain, family circumstances and medication review, are not unusual. However, a number of principles are helpful to keep in mind, which is specific to this process. For the physician the goal of the history taking is often to exclude serious possible causes, identify key problems, build a trusting relationship with the patient (and family) and, if possible, identify a treatment plan. However, the average child with persistent pain not only has a chronic experience of pain and distress, but a chronic experience of medical settings, feelings of being doubted, having the validity of their pain contested, and often a history of failed and pain exacerbating interventions. Three key psychological features of this setting are worth keeping in mind. First, both child and family are likely to be fearful, hoping for a cure, desperate to be helpful, but often ‘on their best behaviour’, so may not immediately appear to be distressed, and may have flat or neutral affect. Secondly, because of their history, any attempt to shift the treatment goal from one of cure to management or symptom control may be heard to be patient and carer(s) blaming, and so should be done only after building trust. Thirdly, what matters is not necessarily what information has been given to the patient and carers, but what they believe to be true. Typically both patient and family will have heard a variety of stories about what may be happening to them.
Physical examination
Time spent fully on examining at the beginning may prevent repetition and unnecessary, distressing investigations at a later date [34]. If there is concern regarding the diagnosis then this is the time to order all investigations and ensure that these are followed up. Undue delay leads to fear and often a worsening of pain symptoms and associated disability.
Psychometric instruments
A number of questionnaire or interview techniques have been used in researching the impact of pain on the lives of children. First, there is now excellent guidance on the optimal methods for the assessment of pain itself in self-report [59] and in observation [60]. There has been recent systematic review of all of the instruments that have been used to assess aspects of adolescent chronic pain [61] and all of the instruments used to assess the impact of adolescent chronic pain on parents [62]. Both reviews observed that there are few assessment tools designed specifically for this group. There are some well-validated and commonly used tools, most notably the Varni/Thompson Paediatric Pain Questionnaire [63] focusing on pain, and the Functional disability Index [64], focusing on disability.
Pain intensity in children has long been measured using simple severity measurement tools, such as the visual analogue scale (VAS) [65]. These give a subjective measure of the pain intensity according to the adolescent and/or that pain perceived by their carer. While these are useful measures, prospectively they give no indication of the impact of pain on the young person's life. For example, one adolescent with a pain score of 7 on the VAS may still get to school where another adolescent with the same score may not be getting out of bed.
Two multidisciplinary tools have recently been developed, one specifically for use in measuring the impact of pain on adolescents with chronic pain (Bath Adolescent Pain Questionnaire) [66] and one specifically for use in measuring the impact of adolescent chronic pain on parents (Bath Adolescent Pain Questionnaire for Parents) [67].
Physiological measures of pain-related indexes
There are little data on the role or utility of measuring physiological processes in the context of childhood chronic pain. The role of thermography and bone scintigraphy in CRPS has been studied [68, 69], evaluating use in the early and late stages of this pain condition. Opinion is divided on their usefulness. Functional MRI has recently been shown to be a useful tool in evaluating the role of the CNS in childhood CRPS [54].
Rehabilitation and self-management
The primary treatment approach to the young person with chronic pain is one of symptom management and psychosocial rehabilitation not a curative one. To complicate matters further, although it may be tempting to focus solely on the amelioration of the suffering of the child, one of the most important aspects of rehabilitation is ‘inclusion’. A dedicated team that works consistently with the adolescent and family will facilitate communication, ensure effective delivery of therapy, reduce iatrogenic influences and enable goals to be reached earlier [18, 70, 71]. Most approaches to rehabilitation share common features including education, symptom control, behavioural science and physical therapy.
Education
Although the evidence for education alone as a treatment for behaviour change is wanting, the provision of a rationale for what are often counter-intuitive instructions in self-management is essential. Additionally, one should never underestimate that the average understanding of anatomy and physiology is at best partial, and more typically fantastic. A critical first step in all rehabilitation is to offer, reoffer and reinforce an understanding of how one's body may be working to maintain pain. Education about pain may be difficult to grasp because there is a dominant cultural view of pain as a warning sign that a disease process or abnormality is present. There is a need for the young person and family to identify a cause, and often skepticism from health professionals that the pains are present at all.
It can be helpful to show and reinforce to the family the fact that many childhood pains have no function; or have outlived their usefulness. Children and their parents often have inflexible or rigid ideas not only about the cause of pain, but also of pain as an important signal of damage or disease. Rhetorical devices (metaphors, stories, examples, pictures) that counter this rigid thinking can be helpful. Headaches are common but are only rarely due to conditions, such as brain tumours or vascular events. Back pain is rarely related to malignancy or arthritis. Explaining the fascinating case of phantom limb experiences can also be helpful, to introduce flexibility to the idea that brain signals must signify peripheral damage.
The young person will often be experiencing the pain-related symptoms and signs mentioned earlier in this chapter. Education can play a valuable role as a fear reduction strategy, exposing children to the counter information that they have unique medical complication, or that their symptoms are related to underlying mysterious disease.
Pharmacotherapy
The number of analgesics and interventions used is a sign that there are no well-controlled therapeutic trials in childhood chronic pain. It is becoming widely accepted, however, that any analgesic intervention should be used alongside multidisciplinary therapy [72–75]. Oral treatments that can be used, with variable success, include tricyclic anti-depressants, NSAIDs, opioids, anti-convulsants and glucocorticoids [76–78]. Sympathetic blockade [79] and botulinum toxin injections have been used in localized muscular pain [76]. Gabapentin and pregabalin may have a role in addressing neuropathic pain in CRPS [77]. A Cochrane review in 2005 showed that tricyclic anti-depressants do have a role in modifying aspects of neuropathic pain in some patients [80].
The evidence supporting the efficacy of any of these therapies in children is lacking. Elsewhere we have described the lack of evidence for pharmacotherapy as embarrassing [81]. This may have been an understatement. Needed is a comprehensive and systematic review on the existing evidence of pharmacotherapies for childhood chronic musculoskeletal pain, and a programme of trials to establish the evidence base.
Medicine review is often an essential task in this population. Polypharmacy, risk of dosing and combination errors, and confusion over the utility of previously and currently prescribed medication is common. Drug withdrawal, detoxification and replacement should be carefully managed.
Psychological therapies
A Cochrane systematic review of psychological therapy reported on the effectiveness of psychological treatments for pain control in common chronic pain problems [82]. Brief psychological therapies such as relaxation, habit reversal and attention-based interventions are highly effective for use with persistent and recurrent pain such as headache [83].
For the complexly disabled patient there are no randomized controlled trials of multidisciplinary multicomponent cognitive behaviour therapy; however, there is evidence that an interdisciplinary programme for adolescents complexly disabled with chronic pain has a positive significant impact [86]. The targets of these therapies are multiple, in fitting with the widespread impact of chronic pain. Disengagement from the pursuit of pain relief and engagement with normal life activities is the overall goal. Its effectiveness is measured by success in return to normal schooling, reduced depression and anxiety, reduction in parental stress and increased activity. In such a programme often the frame of the programme is cognitive behavioural, but the content is delivered by different therapists. The role of the physician is often as an educator, to oversee analgesic withdrawal and to support the overall message that it is safe to increase activity despite pain and reduce reliance on medical support.
Physical therapy
Exercise is key to the rehabilitation of young people with persistent pain. In some conditions, such as CRPS, early intensive physiotherapy is the treatment of choice [23]. The aim of this is accelerated mobilization [84]. Many cases of diffuse pain will require a gentle, paced approach. In all cases, the increase of activity should be consistent despite the pain. Dose seems to be important with high doses being more effective [85]. With musculoskeletal pains the more active the musculoskeletal system becomes the more likely the muscle spasms and tightening are to reduce. Proprioception improves and autonomic changes subside. Where possible the young person should work to devise his or her own ‘fitness plan’. Using a local gym rather than a hospital physiotherapy gym allows them to start to return to a more normal environment [86]. Working in this consistent and paced manner can be extremely hard for the young person and their parents. The pain invariably continues at the beginning (if not throughout) and motivation is poor. Parental anxiety is high [87] and there is a fear that damage will be done. Psychological support during this time is important. The young person will need help setting goals, learning how to communicate pain to peers and family, maintaining motivation on ‘bad days’, managing low mood, dealing with anger and frustration, and overcoming fears. Often they have not been at school for a long period of time and need help in preparing again for this difficult environment. In some cases, there may be other mental health needs that can be identified and appropriately treated. This approach to physical therapy is described in more detail elsewhere [88].
Other therapies
There are ongoing studies that show promise, specifically with CRPS patients and visual counter-stimulation. Mirror therapy focusing on the hypothesis that incongruence between motor output and sensory input produces CRPS is underway with adults [89]. With children this has not been investigated but, in some settings, a trial of mirror therapy may be appropriate. More recently there has been interest in deep brain stimulation in severe cases of CRPS where there is significant disability due to dystonia. There are no ongoing studies, as yet, in children. Spinal cord stimulation has also been used for patients with lower limb CRPS, but while this has been studied in adults, there is no evidence of efficacy in children [90]. Transcutaneous electrical nerve stimulation for musculoskeletal pain has some evidence in general muscular pains but more study is needed in particular, for how such stimulation methods can fit within an overall rehabilitation philosophy [91].
Complementary therapies are commonly utilized by patients with chronic pain [92]. The evidence supporting many of these therapies in children and adolescents is poor [93], but anecdotally many young adults find certain therapies such as acupuncture, massage and aromatherapy helpful.
Natural history and long-term outcomes
The natural history of chronic musculoskeletal pain in children shows that, in many cases, outcome is improved compared with that in adults [94–97]. Early, multidisciplinary input (including cognitive behavioural therapy) has favourable outcome [86]. CRPS in children generally has a favourable prognosis if early physiotherapy is initiated (with psychological support) [24]. This is improved if the parents are involved in the rehabilitation process. However, a prolonged time to treatment and the presence of marked autonomic changes are not good prognostic indicators in this condition.
Relapses of pain are relatively common but if the young person and his/her family recognize the onset of similar pains and put into practice physical and emotional strategies that have previously been taught, then the impact of the pain can be reduced. This is particularly true with regard to musculoskeletal pain. The outlook for childhood abdominal pain and headaches however is not as rosy. Two birth cohorts (1946—Medical Research Council and 1958—National Child Development Study) showed that children with persistent abdominal pain and headaches go on to suffer more physical symptoms in adult life, more anxiety and more depression than healthy children.
In some cases, despite a variety of interventions, the impact of pain remains disabling on the young person and family. We do not yet have prognostic indicators to show us, at initial assessment, how to identify this very small, but complex group.
Conclusions
It is widely accepted that many health care professionals are reviewing a significant number of children and young adults who have pain that is impacting on their development and lifestyle [1, 2, 4]. Paediatric rheumatologists and other paediatric health care providers should understand the epidemiology of childhood musculoskeletal pain, diagnose pain syndromes in children, rule out rheumatic disease and be willing to initiate treatment of pain in children and adolescents. Practitioners’ ability to carry out these tasks is enhanced by an awareness of the biopsychosocial model of pain, which integrates biological, environmental and cognitive behavioural mechanisms in describing the causes and maintenance of children's pain.
Within rheumatology we are seeing increasing numbers of children with hypermobility and chronic joint pain, CRPS and other localized limb pains. This increase may be in part due to the growing expectation for permanent and total well-being, or by an increasingly sedentary lifestyle, but the data cannot yet give us a clear direction for any target of public health intervention. Whatever the cause, we have a population of ?1–2% of children with severe disabling chronic pain beginning to find their way to clinic.
However, there are now well-developed assessment techniques and tools, and a good understanding of the context of assessment and the importance of a broad multidimensional review. There are also promising treatments aimed at the self-management of disability and distress, able to return children and families to normal functioning. However, there is much that is not known. For just two examples, we do not know the role that pain may play in affecting neurodevelopmental pathways of the evolving system in a child. Learning this may be crucial. We also do not know the optimal methods for the involvement of parents in the rehabilitation of their child in pain.
Many of the barriers to good pain management are not primarily scientific or medical, but organizational. For example, we are ignorant about the possible methods of optimal pharmacotherapy. Early appropriate pharmacotherapy may be helpful in altering the trajectory of the child becoming a chronic pain patient, but guidance is lacking. There has been no investment programme in paediatric analgesia by any government or pharmacology company to the best of our knowledge. For a further example, the evidence for brief psychological therapies delivered by nurses in a school environment for the management of chronic pain is overwhelmingly positive [82]. However, there are no national initiatives to train school staff to deliver these programmes. As a final example, although there is evidence that untreated complex pain is personally, socially and financially burdensome for individuals, families and societies, there are scandalously few psychologists and physiotherapists in paediatric settings with training in the rehabilitation of the child with chronic pain.
The consequence of this current failure to address pain before it becomes chronic is that there is a growing population of children who are developing disability, psychological distress and who are socially handicapped. A significant number of these children with musculoskeletal chronic pain will become adults with chronic pain who are dependent on the health care and social care services. If untreated they will present as complex cases to the adult rheumatology services.

Disclosure statement
The authors have declared no conflicts of interest.
REFERENCES:
Sherry DD. Pain syndromes in children, Curr Rheumatol Rep , 2000, vol. 2 (pg. 337-42)
Malleson P, Clinch J. Pain syndromes in children, Curr Opin Rheumatol , 2003, vol. 15 (pg. 572-80)
Sleed M, Eccelston C, Beecham J, Knapp M, Jordan A. The economic impact of chronic pain in adolescence: methodological considerations and a preliminary costs-of-illness study, Pain , 2005(pg. 183-90)
Roth-Isigkeit A. Pain among children and adolescents: restrictions in daily living and triggering factors, Pediatrics , 2005, vol. 115 (pg. 152-62)
Palermo TM. Impact of recurrent and chronic pain on child and family daily functioning: a critical review of the literature, J Dev Behav Pediatr , 2000, vol. 21 (pg. 58-69)
Brattberg G. Do pain problems in young school children persist into early adulthood? A 13-year follow-up, Eur J Pain , 2004, vol. 8 (pg. 187-99)
Perquin CW, Hazebroek-Kampschreur AA, Hunfield JA, et al. . Pain in children and adolescents: a common experience, Pain , 2000, vol. 87 (pg. 51-8)
Lamberg L. Girls’ and boys’ differing response to pain starts early in their lives, J Am Med Assoc , 1998, vol. 280 (pg. 1035-6)
Groholt EK. Recurrent pain in children, socio-economic factors and accumulation in families, Eur J Epidemiol , 2003, vol. 18 (pg. 965-75)
Brattberg G. The incidence of back pain and headache among Swedish school children, Qual Life Res , 1994, vol. 3 Suppl. 1(pg. S27-31)
Smedbraten BK, Natvig B, Rutle O, Bruusgaard D. Self-reported bodily pain in schoolchildren, Scand J Rheumatol , 1998, vol. 27 (pg. 273-6)
Jones GT, Silman AJ, Power C, Macfarlane GJ. Are common symptoms in childhood associated with chronic widespread body pain in adulthood? Results from the 1958 British Birth Cohort Study, Arthritis Rheum , 2007, vol. 56 (pg. 1669-75)
Buskila D, Neumann L. Musculoskeletal injury as a trigger for fibromyalgia/posttraumatic fibromyalgia, Curr Rheumatol Rep , 2000, vol. 2 (pg. 104-8)
Gibson SJ, Littlejohn GO, Gorman MM, Helme RD, Granges G. Altered heat pain thresholds and cerebral event-related potentials following painful CO2 laser stimulation in subjects with fibromyalgia syndrome, Pain , 1994, vol. 58 (pg. 185-93)
Geisser ME, Casey KL, Brucksch CB, Ribbens CM, Appleton BB, Crofford LJ. Perception of noxious and innocuous heat stimulation among healthy women and women with fibromyalgia: association with mood, somatic focus, and catastrophizing, Pain , 2003, vol. 102 (pg. 243-50)
Cohen H, Neumann L, Kotler K, Buskila D. Autonomic nervous system derangement in fibromyalgia syndrome and related disorders, Isr Med Assoc J , 2001, vol. 3 (pg. 755-60)
Clarke J, Eccleston C. Assessing the quality of walking in adults with chronic pain: the development and preliminary psychometric evaluation of the Bath Assessment of Walking Inventory (BAWI), Eur J Pain (in press)
Malleson PN, Connell H, Bennett SM, Eccelston C. Chronic musculoskeletal and other idiopathic pain syndromes, Arch Dis Child , 2001, vol. 84 (pg. 189-92)
Gedalia A, Press J, Klein M, Buskila D. Joint hypermobility and fibromyalgia in schoolchildren, Ann Rheum Dis , 1993, vol. 52 (pg. 494-6)
Buskila D. Fibromyalgia in children-lessons from assessing nonarticular tenderness, J Rheumatol , 1996, vol. 23 (pg. 2017-9)
Buskila D, Neumann L, Herschman E, Gedalia A, Press J, Sukenik S. Fibromyalgia syndrome in children-an outcome study, J Rheumatol , 1995, vol. 22 (pg. 525-8)
Bernstein BH. Reflex neurovascular dystrophy in childhood, J Pediatr , 1978, vol. 93 (pg. 211-5)
Murray CS, Cohen A, Perkins T, Davidson JE, Sills J. Morbidity in reflex sympathetic dystrophy, Arch Dis Child , 2000, vol. 82 (pg. 231-3)
Sherry DD, Wallace CA, Kelley C, Kidder M, Sapp L. Short- and long-term outcomes of children with complex regional pain syndrome type I treated with exercise therapy, Clin J Pain , 1999, vol. 15 (pg. 218-23)
Murray KJ. Hypermobility disorders in children and adolescents, Best Pract Res Clin Rheumatol , 2006, vol. 20 (pg. 329-51)
Adib N, Davies K, Grahame R, Woo P, Murray KJ. Joint hypermobility syndrome in childhood. A not so benign multisystem disorder?, Rheumatology , 2005, vol. 44 (pg. 744-50)
Watson KD, Papageorgiou AC, Jones GT, et al. . Low back pain in schoolchildren: occurrence and characteristics, Pain , 2002, vol. 97 (pg. 87-92)
Varni JW, Walco GA. Chronic and recurrent pain associated with pediatric chronic diseases, Issues Compr Pediatr Nurs , 1988, vol. 11 (pg. 145-58)
Buskila D. Fibromyalgia in inflammatory bowel disease, J Rheumatol , 1999, vol. 26 (pg. 1167-71)
Jay SM, Elliott C, Varni JW. Acute and chronic pain in adults and children with cancer, J Consult Clin Psychol , 1986, vol. 54 (pg. 601-7)
Stinson J, Naser B. Pain management in children with sickle cell disease, Paediatr Drugs , 2003, vol. 5 (pg. 229-41)
Engel JM, Kartin D, Jaffe KM. Exploring chronic pain in youths with Duchenne muscular dystrophy: a model for pediatric neuromuscular disease, Phys Med Rehabil Clin North Am , 2005, vol. 16 (pg. 1113-24) xii
Ravilly S, Robinson W, Berde CB. Chronic pain in cystic fibrosis, Pediatrics , 1996, vol. 98 (pg. 741-7)
Anthony KK, Schanberg LE. Assessment and management of pain syndromes and arthritis pain in children and adolescents, Rheum Dis Clin North Am , 2007, vol. 33 (pg. 625-60)
Taddio A. The effects of early pain experience in neonates on pain responses in infancy and childhood, Paediatr Drugs , 2005, vol. 7 (pg. 245-57)
Ashwal S. Reflex sympathetic dystrophy syndrome in children, Pediatr Neurol , 1988, vol. 4 (pg. 38-42)
Kristjansdottir G, Rhee H. Risk factors of back pain frequency in schoolchildren: a search for explanations to a public health problem, Acta Paediatr , 2002, vol. 91 (pg. 849-54)
Jones GT, Silman AJ, Macfarlane GJ, et al. . Predicting the onset of widespread body pain in children, Arthritis Rheum , 2003, vol. 48 (pg. 2615-21)
Ciccone DS, Bandilla EB, Wu W. Psychological dysfunction in patients with reflex sympathetic dystrophy, Pain , 1997, vol. 71 (pg. 323-33)
Sherry DD, Weisman LR. Psychologic aspects of childhood reflex neurovascular dystrophy, Pediatrics , 1988, vol. 81 (pg. 572-8)
Pillemer FG, Micheli LG. Psychological considerations in youth sports, Clin Sports Med , 1988, vol. 7 (pg. 679-89)
Aasland A, Flato B, Vandvik IH. Psychosocial factors in children with idiopathic musculoskeletal pain: a prospective, longitudinal study, Acta Paediatr , 1997, vol. 86 (pg. 740-6)
Greene JW. Stressful life events and somatic complaints in adolescents, Pediatrics , 1985, vol. 75 (pg. 19-22)
Mailis A, Wade J. Profile of Caucasian women with possible genetic predisposition to reflex sympathetic dystrophy: a pilot study, Clin J Pain , 1994, vol. 10 (pg. 210-7)
Bruscas IC, Beltran ACH, Jimenez ZF. [Polytopic and recurrent reflex sympathetic dystrophy in lower limbs in two siblings.], An Med Interna , 2004, vol. 21 (pg. 183-4)
Postacchini F, Lami R, Pugliese O. Familial predisposition to discogenic low-back pain. An epidemiologic and immunogenetic study, Spine , 1988, vol. 13 (pg. 1403-6)
Rush PJ, Wilmot D, Shore A. Severe reflex neurovascular dystrophy in childhood, Arthritis Rheum , 1985, vol. 28 (pg. 952-6)
Buskila D, Neumann L, Carmi R. Familial aggregation in the fibromyalgia syndrome, Semin Arthritis Rheum , 1996, vol. 26 (pg. 605-11)
Jones GT, Silman AJ, Macfarlane GJ. Parental pain is not associated with pain in the child: a population based study, Ann Rheum Dis , 2004, vol. 63 (pg. 1152-4)
Imbierowicz K, Egle UT. Childhood adversities in patients with fibromyalgia and somatoform pain disorder, Eur J Pain , 2003, vol. 7 (pg. 113-9)
Fitzgerald M. The development of nociceptive circuits, Nat Rev Neurosci , 2005, vol. 6 (pg. 507-20)
Peters JW, Schouw R, Anand KJ, van Dijk M, Duivenvoorden HJ, Tibboel D. Does neonatal surgery lead to increased pain sensitivity in later childhood?, Pain , 2005, vol. 114 (pg. 444-54)
Buskila D, Neumann L, Press J. Pain sensitivity in prematurely born adolescents, Arch Pediatr Adolesc Med , 2003, vol. 157 (pg. 1079-82)
Lebel A, Becerra L, Wallin D, et al. . fMRI reveals distinct CNS processing during symptomatic and recovered complex regional pain syndrome in children, Brain Advance Access published June 20, 2008
Diatchenko L, Slade GD, Nackley AG, et al. . Genetic basis for individual variations in pain perception and the development of a chronic pain condition, Hum Mol Genet , 2005, vol. 14 (pg. 135-43)
Eccleston C, Wastell S, Crombez G, Jordan A. Adolescent social development and chronic pain, Eur J Pain (in press)
Eccleston C, Crombez G, Scotford A, Clinch J, Connell H. Adolescent chronic pain: patterns and predictors of emotional distress in adolescents with chronic pain and their parents, Pain , 2004, vol. 108 (pg. 221-9)
Jordan AL, Eccleston C, Osborn C. Being a parent of the adolescent with complex chronic pain, Eur J Pain , 2007, vol. 11 (pg. 49-56)
Stinson JN, Kavanagh T, Yamada J, Gill N, Stevens B. Systematic review of the psychometric properties, interpretability and feasibility of self-report pain intensity measures for use in clinical trials in children and adolescents, Pain , 2006, vol. 125 (pg. 143-57)
von Baeyer CL, Spagrud LJ. Systematic review of observational (behavioral) measures of pain for children and adolescents aged 3 to 18 years, Pain , 2007, vol. 127 (pg. 140-50)
Eccleston C, Jordan AL, Crombez G. The impact of chronic pain on adolescents: a review of previously used measures, J Pediatr Psychol , 2006, vol. 31 (pg. 684-97)
Jordan AL, Eccleston C, Crombez G. The impact of adolescent chronic pain on parents: a review of previously used measures, J Pediatr Psychol (in press)
Varni JW, Thompson KL, Hanson V. The Varni/Thompson Pediatric Pain Questionnaire. I. Chronic musculoskeletal pain in juvenile rheumatoid arthritis, Pain , 1987, vol. 28 (pg. 27-38)
Walker LS, Greene JW. The functional disability inventory: measuring a neglected dimension of child health status, J Pediatr Psychol , 1991, vol. 16 (pg. 39-58)
Carlsson AM. Assessment of chronic pain. I. Aspects of the reliability and validity of the visual analogue scale, Pain , 1983, vol. 16 (pg. 87-101)
Eccleston C, Jordan A, McCracken LM, Connell H, Clinch J. The Bath Adolescent Pain Questionnaire (BAPQ): development and preliminary psychometric evaluation of an instrument to assess the impact of chronic pain on adolescents, Pain , 2005, vol. 118 (pg. 263-70)
Jordan A, Eccleston C, McCracken LM, Connell H, Clinch J. The Bath Adolescent Pain – Parental Impact Questionnaire (BAP-PIQ): development and preliminary psychometric evaluation of an instrument to assess the impact of parenting an adolescent with chronic pain, Pain , 2008, vol. 137 (pg. 478-87)
Lightman HI, Pochaczevsky R, Aprin H, Ilowite N. Thermography in childhood reflex sympathetic dystrophy, J Pediatr , 1987, vol. 111 (pg. 551-5)
Goldsmith DP, Vivino FB, Heyman S, et al. . Nuclear imaging and clinical features of childhood reflex neurovascular dystrophy: comparison with adults, Arthritis Rheum , 1989, vol. 32 (pg. 480-5)
Christie D, Wilson C. CBT in paediatric and adolescent health settings: a review of practice-based evidence, Pediatr Rehabil , 2005, vol. 8 (pg. 241-7)
Aitkenhead S. Managing chronic pain in children, Nurs Times , 2001, vol. 97 (pg. 34-5)
McGrath PA. An assessment of children's pain: a review of behavioral, physiological and direct scaling techniques, Pain , 1987, vol. 31 (pg. 147-76)
Stanton-Hicks M. Complex regional pain syndromes: guidelines for therapy, Clin J Pain , 1998, vol. 14 (pg. 155-66)
Shapiro BS. Treatment of chronic pain in children and adolescents, Pediatr Ann , 1995, vol. 24 (pg. 148-50)(pg. 153-6)
Kashikar-Zuck S. Treatment of children with unexplained chronic pain, Lancet , 2006, vol. 367 (pg. 380-2)
Gordon N. Reflex sympathetic dystrophy, Brain Dev , 1996, vol. 8 (pg. 257-62)
van de Vusse AC, Stomp-van den Berg SG, Kessels AH, Weber WE. Randomised controlled trial of gabapentin in complex regional pain syndrome type, BMC Neurol , 2004, vol. 4 pg. 13
Crofford LJ. Pain management in fibromyalgia, Curr Opin Rheumatol , 2008, vol. 20 (pg. 246-50)
Mailis A, Furlan A. Sympathectomy for neuropathic pain, Cochrane Database Syst Rev , 2003, vol. (2) pg. CD002918
Saarto T, Wiffen PJ. Antidepressants for neuropathic pain, Cochrane Database Syst Rev , 2007, vol. (4)
Eccleston C, Malleson P. Managing chronic pain in children and adolescents. We need to address the embarrassing lack of data for this common problem, Br Med J , 2003, vol. 326 (pg. 1408-9)
Eccleston C, Morley S, Williams A, Yorke L, Mastroyannopoulou A. Psychological therapies for the management of chronic and recurrent pain in children and adolescents (Cochrane review), In: The Cochrane Library, Issue 1. , 2003OxfordUpdate Software
Martin PR, Forsyth MR, Reece J. Cognitive-behavioral therapy versus temporal pulse amplitude biofeedback training for recurrent headache, Behav Ther , 2007, vol. 38 (pg. 350-63)
Littlejohn GO. Reflex sympathetic dystrophy in adolescents: lessons for adults, Arthritis Rheum , 2004, vol. 51 (pg. 151-3)
Lee BH, Scharff L, Sethna NF, et al. . ‘Physical therapy and cognitive-behavioral treatment for complex regional pain syndromes’, J Pediatr , 2002, vol. 141 (pg. 135-40)
Eccleston C, Malleson PM, Clinch J, Connell H, Sourbut C. Chronic pain in adolescents: evaluation of a programme of interdisciplinary cognitive behaviour therapy, Arch Dis Child , 2003, vol. 88 (pg. 881-58)
Eccleston C, Crombez G, Scotford A, Connell H, Clinch J. Adolescent chronic pain: patterns and predictors of emotional distress in adolescents with chronic pain and their parents, Pain , 2004, vol. 108 (pg. 221-9)
Eccleston Z, Eccleston C. Interdisciplinary management of adolescent chronic pain: developing the role of the physiotherapist, Physiotherapy , 2004, vol. 90 (pg. 77-81)
McCabe CS, Haigh RC, Ring EF, Halligan PW, Wall PD, Blake DR. A controlled pilot study of the utility of mirror visual feedback in the treatment of complex regional pain syndrome (type 1), Rheumatology , 2003, vol. 42 (pg. 97-101)
Kemler MA, de Vet HC, Barendse GA, van den Wildenberg FA, van Kleef M. Effect of spinal cord stimulation for chronic complex regional pain syndrome Type I: five-year final follow-up of patients in a randomized controlled trial, J Neurosurg , 2008, vol. 108 (pg. 292-8)
Ashwal S, Tomasi L, Neumann M, Schneider S. Reflex sympathetic dystrophy syndrome in children, Pediatr Neurol , 1988, vol. 4 (pg. 38-42)
Carter B. Complementary therapies and management of chronic pain, Paediatr Nurs , 1995, vol. 7 (pg. 18-22)
Tsao JC, Zeltzer LK. Complementary and alternative medicine approaches for pediatric pain: a review of the state-of-the-science, Evid Based Complement Altern Med , 2005, vol. 2 (pg. 149-59)
Ledingham J, Doherty S, Doherty M. Primary fibromyalgia syndrome – an outcome study, Br J Rheumatol , 1993, vol. 32 (pg. 139-42)
Gedalia A, Garcia CO, Molina JF, Bradford NJ, Espinoza LR. Fibromyalgia syndrome: experience in a pediatric rheumatology clinic, Clin Exp Rheumatol , 2000, vol. 18 (pg. 415-9)
Krilov LR, Fisher M, Friedman SB, Reitman D, Mandel FS. Course and outcome of chronic fatigue in children and adolescents, Pediatrics , 1998, vol. 102 (pg. 360-6)
Bursch B, Walco GA, Zeltzer L. Clinical assessment and management of chronic pain and pain-associated disability syndrome, Dev Behav Pediatr , 1998, vol. 1 (pg. 45-53)

Return to PEDIATRICS
Return to NECK AND BACK PAIN
Since 4–03–2017


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |