Successful Management of Acute-onset Torticollis
in a Giraffe (Giraffa camelopardalis reticulata)This section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Journal of Zoo and Wildlife Medicine 2013 (Mar); 44 (1): 181–185 ~ FULL TEXT
Liza I. Dadone, Kevin K. Haussler, Greg Brown,
Melanie Marsden, James Gaynor, Matthew S. Johnston
Research Department,
Palmer College of Chiropractic West,
San Jose, California.
A 2-yr-old male reticulated giraffe (Giraffa camelopardalis reticulata) presented with severe midcervical segmental torticollis upon arrival as an incoming shipment. Despite initial medical management, the giraffe developed marked neck sensitivity, focal muscle spasms, and decreased cervical range of motion. Using operant conditioning to assist patient positioning and tolerance to cervical manipulation, a series of manually applied chiropractic treatments were applied to the affected cervical vertebrae in an effort to restore normal cervical mobility. Laser therapy and cervical range of motion exercises were also used to reduce cervical muscle hypertonicity. The combined application of these nontraditional therapies produced marked clinical improvement. This case highlights the potential benefits of combining traditional medical management with chiropractic treatment and physical therapy techniques for management of severe acute-onset torticollis in a giraffe.
KEYWORDS: Giraffe, Giraffa camelopardalis reticulata, torticollis, low-level laser therapy, chiropractic, physical therapy
From the FULL TEXT Article:
BRIEF COMMUNICATION
A 2-yr-old captive-borne male reticulated giraffe (Giraffa camelopardalis reticulata), estimated to weigh 450 kg, developed severe torticollis during transport between zoos. The giraffe had been given vitamin E (Emcelle Tocopherol 500 IU/ml, Stuart Products, Bedford, Texas 76022, USA; 2.2 IU/kg p.o. q 24 h for 7 days) and was visually normal when loaded. The giraffe was transported for about 20 hr in a standard shipping crate (2.732.1 33.7 m high) and was reported to have remained calm during transport. Before unloading at the destination, the giraffe had severe left lateral torticollis at C4–C5 with compensatory right lateral torticollis at C3–C4 and superficial bleeding of the ossicones with blood streaks on the inside of the trailer at ossicone height (Fig. 1).
The giraffe unloaded with no obvious neurologic deficits, did not seem painful in the cervical region, and started eating immediately from an elevated feeder. Prophylactically, the giraffe was givenvitamin E (Bo-Se Injection, Schering-Plough Animal Health, Union, New Jersey 07083, USA; 1.1 mg/kg i.m.),
phenylbutazone (Phenylbute Boluses 1 g, Phoenix Pharmaceutical, St. Joseph, Missouri 64507, USA; 4.4 mg/kg p.o. loading dose, then 2.2 mg/kg p.o. q 24 h for 2 days), and
methocarbamol (500-mg tablets, Qualitest Pharmaceuticals, Huntsville, Alabama 35811, USA; 24.4 mg/ kg p.o. q 24 h for 13 wk).On day 4, the phenylbutazone was substituted with
oral meloxicam (Mobic 15-mg tablets, Boehringer Ingelheim Pharmaceuticals, Ridgefield, Connecticut 06877, USA; 0.17 mg/kg p.o. q 24 h for 10 days).
Over the first few weeks, the neck was visibly straighter except for continued mild lateral deviation at C3–C5. Target training had been initiated at 6 days of age; therefore, in an effort to evaluate and treat the torticollis directly, additional training was initiated to assist entry into a restraint chute and to tolerate multiple people standing on an elevated platform near its neck. Lateral cervical radiographs, with the giraffe standing and unsedated, were taken over the first 10 wk and revealed no obvious osseous pathology. Desensitization training was initiated to allow palpation and manipulation of the affected cervical region.
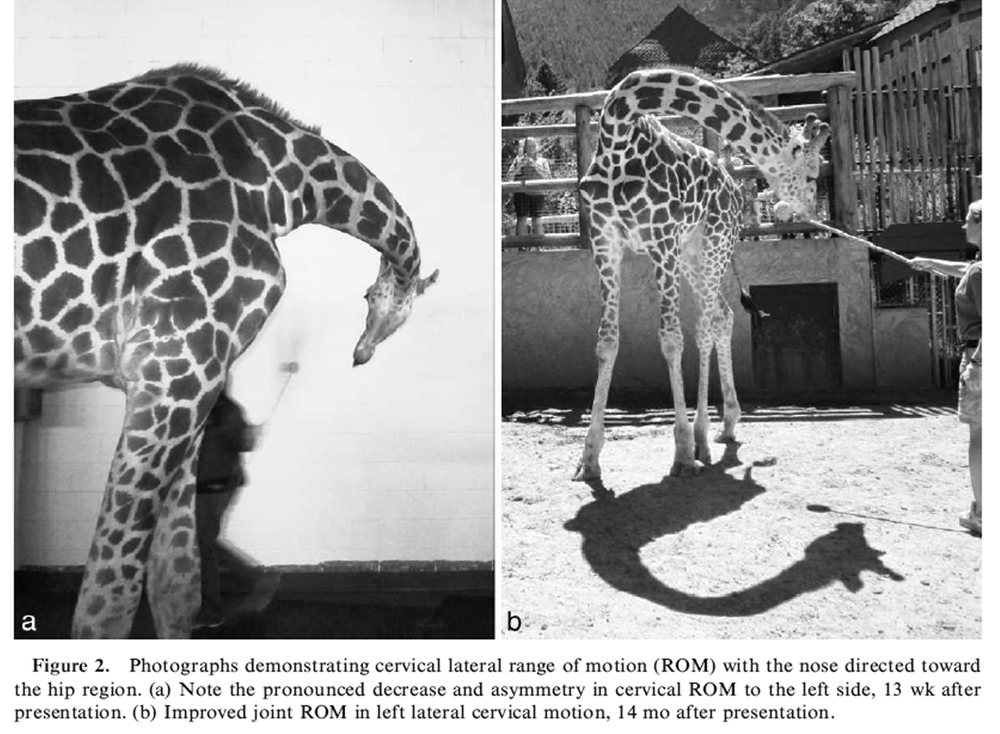
Thirteen weeks after presentation, chiropractic treatment (i.e., manually applied, high-velocity, low-amplitude thrusts) was applied to the left C4- C5 intervertebral articulation in an effort to restore normal segmental motion and motor control because of concerns for long-term clinical deterioration. Gradually increasing manual force was applied laterally over the left C4–C5 articular process until firm resistance to the induced motion was felt (i.e., end range of motion), and then a lateral-to-medial thrust was applied by a reinforced hypothenar contact. [10] In the month before chiropractic treatment, the neck had visually straightened. Cervical range of motion (ROM) was assessed with targeting and directing the giraffe to move its head and nose laterally and toward the hip bilaterally. Pronounced muscle guarding was observed during left lateral bending (Fig. 2a). Methocarbamol was discontinued 13 wk after presentation on a trial basis because it was unclear whether the medication was effective.
Fifteen weeks after initial presentation, a second chiropractic treatment was applied over the left C3–C4 and right C4–C5 intervertebral articulations. At this time, there was visibly increased lateral cervical deviation bilaterally (C3–C4 on the left side and C4–C5 on the right) and obvious soft tissue pain on the left side at C4–C5. These detrimental changes were attributed to the discontinued use of methocarbamol administration. Despite resuming methocarbamol (24.4 mg/kg p.o. q 24 h for 8 wk), keepers reported continued increased sensitivity along the left side of the neck. Chiropractic treatments were repeated only every 4–8 wk because of scheduling conflicts.
At 4 mo, laser therapy (LiteCure, Companion Therapy Laser, Newark, Delaware 19702, USA) was applied over the left side of the neck to help manage soft tissue pain and hypertonicity (Fig. 3). Moderate epaxial muscle hypertonicity and pain on palpation were localized to the left C4–C5 region, with milder changes identified over the right C3–C4 articulation. Laser therapy was applied twice weekly for 4 wk, with treatments focused over sites of muscle hypertonicity. The laser settings included ‘‘edema/swelling’’ (10 W, 1,000 Hz, 4 min/site) and ‘‘arthritic pain’’ (10 W, continuous wave frequency, 6 min/site). The giraffe would stand in the restraint chute longer when laser therapy was applied compared with regular training sessions without laser therapy. One month after initiating laser therapy, the cervical ROM was significantly improved in left lateral bending, muscle hypertonicity and contracture were reduced, and the left side of the neck was less sensitive to palpation. Laser therapy was then continued once weekly for 9 mo. A gradual continued increase in left lateral bending ROM was evident over the next few months.
Figure 3. Photograph of giraffe positioning
in restraint chute for laser therapy treatments.

At 8 mo, the cervical ROM and muscle hypertonicity continued to improve; however, mild lateral deviations were noted over the dorsum of the withers and at the L5–S1 spinal region. The giraffe was subsequently trained for the application of laser therapy and chiropractic treatments at those sites.
One year after initial presentation, the giraffe had nearly normal cervical ROM but continued to have mild thoracolumbar scoliosis. Small regions of muscle hypertonicity continued along the neck, but were markedly improved. Target training was initiated to induce active stretching exercises for the cervical region by placement of the target and nose near the hip region with a prolonged held position (10-sec duration, five repetitions s.i.d.) bilaterally. Because of patient height, the stretching exercises were not able to be performed within the restraint chute; however, target training while free in a paddock was effective after several weeks of training.
One month after initiating consistent stretching exercises and 14 mo after initial presentation, cervical ROM was excellent (Fig. 2b), cervical muscle hypertonicity was minimal, and only mild torticollis remained. On the basis of clinical improvement, laser therapy was discontinued. Two years after presentation, neck stretching exercises are continued daily, and chiropractic treatment is applied every 4–8 wk in an effort to help maintain cervical ROM and muscular stability.
In captive giraffes, cervical injuries and muscle spasms occur in young animals from unknown causes, [4] during immobilizations and recovery,1,5 and as the result of shipping. In two recent cases, shipment-related cervical injuries have resulted in spontaneous death or euthanasia within days or months of onset. In 2009, a 5-yr-old female giraffe sustained cervical vertebral endplate fractures during transport and was euthanatized. [9] In 2010, a 10-yr-old male giraffe had severely avulsed cervical vertebral ligaments and was euthanatized immediately after shipment (Erica Lipanovich, pers. comm., August 2011). The current case is unique in that immediate life-threatening injuries did not occur and conservative treatment approaches were very effective in restoring cervical function.
The etiology of transport-related cervical injuries is unknown, although it is speculated that trauma-induced malalignment of the cervical vertebrae and nuchal ligament displacement could lead to progressive cervical vertebral subluxation, compressive myelopathy, and paralysis (Roy Burns, pers. comm., October 2011). In humans, whiplash-associated injuries and soft tissue damage are reported with velocity changes of as little as 6 km/hr.3 The authors speculate that relatively minor velocity changes during shipment could likewise cause cervical soft tissue injuries in giraffes. The giraffe described in this report was younger at the time of shipment compared with other reported cases of cervical injuries in giraffes; therefore, a younger age or a smaller patient size might have helped minimize the severity of the injury.
Acquired cervical torticollis has been reported in horses and an alpaca from parasite migration of Parelaphostrongylus tenuis within the cervical spinal cord. [2, 8] However, it is not known whether giraffe are susceptible to this parasite, and more cases of spontaneous torticollis without a shipping history would be expected in captive giraffes if this were an underlying etiology.
The nontraditional modalities of chiropractic, laser therapy, and active stretching exercises were essential for successful management of this case. Chiropractic treatment and stretching are well-recognized forms of spinal rehabilitation to address pain, flexibility, and muscle tone in horses. [7] To date, these therapies have not been reported in addressing musculoskeletal issues in exotic animal species. Laser therapy is increasingly utilized in exotic pets. [12] Laser light is considered to produce photobiostimulation at the cellular level, which leads to the normalization of cell function, pain relief, improved circulation, improved nerve function, accelerated healing, and stimulation of acupuncture and trigger points. [11] On the basis of the positive response to these therapies in this case, similar forms of treatment should be considered for managing cervical injuries in giraffes and in select cases in other zoo species. In refractory cervical muscle contractures that produce torticollis, experimental treatment with botulinum toxin-A (Botoxt) could also be considered as a possible treatment option because it is commonly used in humans for the treatment of cervical dystonia. [6]
Acknowledgments.
The authors thank Barb Wolfe, Kay Backues, Erica Lipanovich, Eric Klaphake, and Andrea Goodnight for consulting with this case. We also thank Allison Kao for exceptional work with training the patient, the veterinary and animal departments for help with case management, Pike’s Peak Veterinary Clinic and LiteCure Companion Laser Therapy company for providing access to equipment, and the Oakland Zoo staff for starting patient training at such an early age in this giraffe.
References:
Aprea, F., P. M. Taylor, A. Routh, D. Field, E. Flach, and T. Bouts. 2011.
Spinal cord injury during recovery from anaesthesia in a giraffe.
Vet. Rec. 169: 50.Biervliet, J. V., A. deLahunta, D. Ennulat, M. Oglesbee, and B. Summers. 2003.
Acquired cervical scoliosis in six horses associated with dorsal
grey column chronic myelitis.
Equine Vet. J. 35: 86–92.Brault, J. R., J. B. Wheeler, G. P. Seigmund, and E. J. Brault. 1998.
Clinical response of human subjects to rear-end automobile collisions.
Arch. Phys. Med. Rehabil. 79: 72–80.Burns, R. B., W. C. Shellabarger, and Z. S. Gyumesi. 2009.
Multifocal cervical instability and cervical spinal cord impingement
in a Masai giraffe (Giraffa camelopardalis tippelskirchi).
Proc. 2009 Am. Assoc. Zoo Vets. Conf. 55 pp. [Abstr.]Bush, M., D. G. Grobler, and J. P. Raath. 2002.
The art and science of giraffe (Giraffa camelopardalis) immobilization/anesthesia.
In: Heard, D. (ed.). Zoological Restraint and Anesthesia.
Ithaca, New York: International Veterinary Information Service.Comella, C. L., J. Jankovic, D. D. Truong, A. Hanschmann, S. Grafe,
and U.S. XEOMIN Cervical Dystonia Study Group. 2011.
Efficacy and safety of incobotulinumtoxinA (NT 201, XEOMINt, botulinum neurotoxin
type A, without accessory proteins) in patients with cervical dystonia.
J. Neurol. Sci. 308: 103–109.Haussler, K. K., C. E. Martin, and A. E. Hill. 2010.
Efficacy of spinal manipulation and mobilization in trunk flexibility and
stiffness in horses: a randomized clinical trial.
Equine Vet. J. 42(Suppl. 38): 695–702.Johnson, A. L., C. G. Lamm, and T. J. Divers. 2006.
Acquired cervical scoliosis attributed to Parelaphostrongylus tenuis
infection in an alpaca.
J. Am. Vet. Med. Assoc. 229: 562–655.Kilburn, J. J., D. V. Devine, R. J. Bahr, and K. A. Backues. 2020.
Cervical Salter I physeal fracture in a baringo giraffe
(Giraffa camelopardalis).
Proc. 2010 Am. Assoc. Zoo Vets. Conf. 248 pp. [Abstr.]Peterson, D. H., and T. F. Bergmann. 2002.
Principles of adjustive technique.
In: Peterson, D. H., and T. F. Bergmann (eds.).
Chiropractic Technique, 2nd ed.
Mosby, St. Louis, Missouri. Pp. 97–174.Riegel, R. J. 2008.
Laser Therapy in the Companion Animal Practice: Mechanisms and Protocols
for Class IV Laser Therapy.
LiteCure LLC, Newark, Delaware. Pp. 45–49.Rychel, J. K., M. S. Johnston, and N. G. Robinson. 2011.
Zoologic companion animal rehabilitation and physical medicine.
Vet. Clin. Exot. Anim. 14: 131–140.
Return to PEDIATRICS
Since 3-20-2013


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |