

Disability-adjusted Life Years (DALYs) for 291 Diseases
and Injuries in 21 Regions, 1990-2010: A Systematic
Analysis for the Global Burden of Disease Study 2010This section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Lancet. 2012 (Dec 15); 380 (9859): 2197–2223 ~ FULL TEXT
Christopher J L Murray, MD, Theo Vos, PhD, Rafael Lozano, MD, Mohsen Naghavi, PhD, Abraham D Flaxman, PhD, Catherine Michaud, MD et al.
Institute for Health Metrics and Evaluation,
Seattle 98121, WA, USA.
BACKGROUND: Measuring disease and injury burden in populations requires a composite metric that captures both premature mortality and the prevalence and severity of ill-health. The 1990 Global Burden of Disease study proposed disability-adjusted life years (DALYs) to measure disease burden. No comprehensive update of disease burden worldwide incorporating a systematic reassessment of disease and injury-specific epidemiology has been done since the 1990 study. We aimed to calculate disease burden worldwide and for 21 regions for 1990, 2005, and 2010 with methods to enable meaningful comparisons over time.
METHODS: We calculated DALYs as the sum of years of life lost (YLLs) and years lived with disability (YLDs). DALYs were calculated for 291 causes, 20 age groups, both sexes, and for 187 countries, and aggregated to regional and global estimates of disease burden for three points in time with strictly comparable definitions and methods. YLLs were calculated from age-sex-country-time-specific estimates of mortality by cause, with death by standardised lost life expectancy at each age. YLDs were calculated as prevalence of 1160 disabling sequelae, by age, sex, and cause, and weighted by new disability weights for each health state. Neither YLLs nor YLDs were age-weighted or discounted. Uncertainty around cause-specific DALYs was calculated incorporating uncertainty in levels of all-cause mortality, cause-specific mortality, prevalence, and disability weights.
FINDINGS: Global DALYs remained stable from 1990 (2·503 billion) to 2010 (2·490 billion). Crude DALYs per 1000 decreased by 23% (472 per 1000 to 361 per 1000). An important shift has occurred in DALY composition with the contribution of deaths and disability among children (younger than 5 years of age) declining from 41% of global DALYs in 1990 to 25% in 2010. YLLs typically account for about half of disease burden in more developed regions (high-income Asia Pacific, western Europe, high-income North America, and Australasia), rising to over 80% of DALYs in sub-Saharan Africa. In 1990, 47% of DALYs worldwide were from communicable, maternal, neonatal, and nutritional disorders, 43% from non-communicable diseases, and 10% from injuries. By 2010, this had shifted to 35%, 54%, and 11%, respectively. Ischaemic heart disease was the leading cause of DALYs worldwide in 2010 (up from fourth rank in 1990, increasing by 29%), followed by lower respiratory infections (top rank in 1990; 44% decline in DALYs), stroke (fifth in 1990; 19% increase), diarrhoeal diseases (second in 1990; 51% decrease), and HIV/AIDS (33rd in 1990; 351% increase). Major depressive disorder increased from 15th to 11th rank (37% increase) and road injury from 12th to 10th rank (34% increase). Substantial heterogeneity exists in rankings of leading causes of disease burden among regions.
INTERPRETATION: Global disease burden has continued to shift away from communicable to non-communicable diseases and from premature death to years lived with disability. In sub-Saharan Africa, however, many communicable, maternal, neonatal, and nutritional disorders remain the dominant causes of disease burden. The rising burden from mental and behavioural disorders, musculoskeletal disorders, and diabetes will impose new challenges on health systems. Regional heterogeneity highlights the importance of understanding local burden of disease and setting goals and targets for the post–2015 agenda taking such patterns into account. Because of improved definitions, methods, and data, these results for 1990 and 2010 supersede all previously published Global Burden of Disease results.
There is more like this at our
Global Burden of Disease PageFUNDING: Bill & Melinda Gates Foundation.
From the FULL TEXT Article:
Introduction
Summary measures of population health combine information on mortality and non-fatal health outcomes to provide unique perspectives on levels of health and key contributing causes to loss of health. [1] There are three related but distinct uses of summary measures of population health at the global, regional, national, or subnational levels. Summary measures can be used, first, to compare overall population health across communities and over time; for example, national estimates of healthy life expectancy (HALE) have been published for 191 countries. [2] The second and more common use of summary measures is to provide a coherent overall picture as to which diseases, injuries, and risk factors contribute the most to health loss in a given population. The comparative view provided by summary measures helps decision-makers, researchers, and citizens understand what the most important problems are and whether they are getting better or worse. This information, along with information on the costs, intervention effectiveness, and equity implications of health interventions and policy options, lays the foundation for a debate on priorities for health policy action and research that is clearly informed by the best available evidence. Third, summary measures can help guide an assessment of where health information systems are strong or weak by identifying which data sources required for their calculation are missing, of low quality, or highly uncertain. Different users in different contexts will make use of summary measures for any of the three purposes.
The only comprehensive effort to date to estimate summary measures of population health for the world, by cause, is the ongoing Global Burden of Diseases, Injuries, and Risk Factors (GBD) enterprise. For a summary measure of population health, the GBD study uses disability-adjusted life years (DALYs), which are the sum of years of life lost due to premature mortality (YLL) and years lived with disability (YLD). While the term disability has taken on many different meanings in different settings, [3–7] in the GBD lexicon it refers to any short-term or long-term health loss, other than death. The construct of health in the GBD study is defined in terms of functioning, which encompasses multiple domains of health such as mobility, pain, affect, and cognition.8 Final GBD results for 1990 were published in 1996 and 1997. [9–14] GBD estimates were produced for 1999, 2000, 2001, 2002, and 2004 by WHO. [15–19] Although GBD results have been estimated by WHO for 1999–2004, and incorporated new approaches to mortality measurement, [20] these updates undertook systematic analysis of the epidemiological data for only a subset of disease sequelae. [21] DALY results have been referenced extensively in global health debates and decision-making. The first results from the GBD study for 1990 were published in the World Development Report 1993: Investing in Health. [22] The study has led to many national burden of disease studies in developed and developing countries using similar methods. [23–75] Subnational studies have also been done in many countries. [76–81] Quantifying health loss in terms of DALYs has led to increased attention to mental health problems [82] and injuries, [83] non-fatal health effects of neglected tropical diseases, [84] and more generally non-communicable diseases (NCDs). [85]
The Global Burden of Diseases, Injuries, and Risk Factors Study 2010 (GBD 2010) [86] has been implemented as a collaboration of seven institutions: the Institute for Health Metrics and Evaluation (IHME) as the coordinating centre providing academic leadership; the University of Queensland School of Population Health; WHO; the Johns Hopkins Bloomberg School of Public Health; the Harvard School of Public Health; Imperial College London; and the University of Tokyo. The GBD 2010 has been undertaken to apply comparable, systematic, and rigorous epidemiological assessment of all diseases and injuries. The number of disease and injury sequelae has expanded from 483 to 1160. The study also uses a much more detailed set of age groups, 20 instead of eight; and 21 regions instead of the 14 used in the GBD 2000 study. [86]
In the GBD 1990 study, results were computed with several variants of DALYs reflecting different social-value choices for discounting and age-weighting. The base case reported for DALYs used a 3% discount rate and age weights that placed the greatest emphasis on health outcomes in young adults. WHO has continued in its updates for 1999, 2000, 2001, 2002, and 2004 to use this base case set of social-value choices although other variants have been calculated. One publication for 2001 reported discounted DALYs without age-weighting. [87] On the basis of broad consultation, [86] the base case for DALYs in GBD 2010 has been simplified to omit both discounting and age-weighting. YLLs are calculated with reference to a new reference-standard life expectancy at each age; for example, a death at age 5 years counts as 81·4 YLLs and a death at age 60 counts as 27·8 YLLs. [86] The reference standard has been computed on the basis of the lowest age-specific death rates recorded across countries in 2010. YLDs are based on the product of the prevalence of a sequela and its associated disability weight. Of note, the empirical basis for disability weights in the GBD 2010 derives from judgments of the general public about health severity, by contrast with the GBD 1990 study that relied on judgments of health-care professionals. [3] A key tenet of the GBD analytical philosophy is not to allow advocates for the importance of specific diseases to choose the disability weights associated with specific disorders (Panel).
Disability-adjusted life years and Global Burden of Disease definitions
Disability-adjusted life years (DALYs) are a summary metric of population health. DALYs represent a health gap; they measure the state of a population’s health compared to a normative goal. The goal is for individuals to live the standard life expectancy in full health.
DALYs are the sum of two components: years of life lost due to premature mortality (YLLs) and years lived with disability (YLDs).
YLLs are computed by multiplying the number of deaths at each age x by a standard life expectancy at age x. The standard selected represents the normative goal for survival and has been computed based on the lowest recorded death rates across countries in 2010.
YLDs are computed as the prevalence of different disease-sequelae and injury-sequelae multiplied by the disability weight for that sequela. Disability weights are selected on the basis of surveys of the general population about the loss of health associated with the health state related to the disease sequela.
DALYs are an absolute measure of health loss; they count how many years of healthy life are lost due to death and non-fatal illness or impairment. They reflect the number of individuals who are ill or die in each age-sex group and location. Population size and composition influences the number of DALYs in a population.
The GBD 2010 disease-and-injury-cause list is a hierarchical list of 291 diseases and injuries. At the first level of disaggregation causes are divided into three broad groups: communicable, maternal, neonatal, and nutritional disorders; non-communicable diseases; and injuries. At each level in the hierarchy, the cause list provides a set of mutually exclusive and collectively exhaustive categories.
Sequelae — in total, we have identified 1160 sequelae of the 291 diseases and injuries. For example, diabetic neuropathy is a sequela of diabetes mellitus. To avoid double counting, a sequela can only appear in the cause-sequela list once even if the same outcome might be claimed by more than one disease.
Health states — across the 1160 sequelae, 220 unique health states were identified. For example, both malaria and hookworm have mild anaemia as a sequela. Mild anaemia is a unique health state. The list of unique health states serves two purposes:
(a) to allow assessment of the total burden of some health states such as anaemia across various causes; and
(b) to simplify the task of measuring disability weights for sequelae.DALYs presented in this study are not age-weighted and are not discounted for time preference. Base case tabulations for the GBD 1990 and GBD 2000 studies used age-weighting and a 3% discount rate.
Because of improved data and methods, comparisons between 1990 and 2010 should be based exclusively on the results of this study.
The goal of the GBD 2010 has been to synthesise available data on the epidemiology of all major diseases and injuries to provide a comprehensive and comparable assessment of the magnitude of 291 diseases and injuries and their associated sequelae in 1990, 2005, and 2010. In this Article, we summarise the results of a large and complex study involving hundreds of researchers. The findings draw on millions of observations of epidemiological parameters over the past three decades. By the synthetic nature of the work, we provide a high-level overview of key findings. Because this study uses consistent definitions and improved methods to assess the GBD over two decades, the findings supersede all previously published GBD results.
Methods
Study design
The division of countries into 21 epidemiological regions, the choice of 20 age groups, and the primary methods for each of the 18 components of the study are described by Murray and colleagues.86 We provide only a brief description here. The GBD cause list has 291 diseases and injuries, which are organised in a hierarchy with up to four levels of disaggregation. For each cause, there are from one to 24 sequelae. In total, the study includes 1160 sequelae. The expansion of the cause list and the criteria used to add causes and sequelae across various revisions of the GBD study is described elsewhere.86
Causes of death
YLLs have been computed on the basis of cause-of-death estimates for 235 of 291 causes of death for 20 age groups, both sexes, and 187 countries. Two disorders, sudden infant death syndrome (SIDS) and aortic aneurysm, cause only YLLs. Cause of death estimates have been developed with a comprehensive database of vital registration, verbal autopsy, surveillance, and other sources covering 187 countries from 1980 to 2010. Quality of each observation has been assessed, and various revisions of the International Classification of Diseases and Injuries (ICD) have been mapped. Deaths assigned to causes that are not likely to underlie causes of death have been reassigned with standardised algorithms.88, 89 All observations were converted to the 20 standard GBD age groups. For 133 causes, including all major causes of death excluding HIV/AIDS, we used the Cause of Death Ensemble model (CODEm) strategy to develop ensembles of the best performing models that meet two plausibility criteria. The first criterion is that the direction of the regression coefficient for a covariate is in the expected direction, and the second is that the coefficient has a p value less than 0·05. Performance is assessed in terms of rigorous out-of-sample predictive validity testing based on the root-mean-squared error of the log of the age-specific death rates, the percentage of time that trend is accurately predicted, and the coverage of the uncertainty intervals (UIs).
For HIV/AIDS, we have used CODEm for countries with high-quality vital registration systems and the UNAIDS 2012 revision estimates by age and sex for the remaining countries. Natural history models have been used for African trypanosomiasis, measles, whooping cough, hepatitis E, typhoid and paratyphoid fevers, leishmaniasis, HIV/AIDS, and congenital syphilis. Aetiologies or subcauses for diarrhoea, lower respiratory infections, meningitis, chronic kidney diseases, maternal disorders, cirrhosis, and liver cancer have been based on meta-regression of published studies on aetiology, disease registry data, and, where appropriate, vital registration data. For some rarer causes such as diphtheria or varicella, negative binomial regression has been used; for a few causes that rarely account for mortality, fixed proportions of the parent cause in the hierarchy have been used by age, sex, and region. A key aspect of the GBD method is to enforce consistency between the sum of cause-specific mortality and independently assessed levels of all-cause mortality derived from demographic sources (see Wang and colleagues90 for details on the all-cause-mortality analysis). Uncertainty in cause-of-death model predictions has been captured with standard simulation methods by taking 1000 draws91 for each age, sex, country, year, and cause (see Lozano and colleagues92 for more details on causes-of-death methods). Consistency with all-cause mortality is enforced at the draw level. Final uncertainty for YLLs reflects uncertainty in the levels of all-cause mortality in each age-sex-country-year as well as uncertainty in the estimation of each cause of death for that age-sex-country-year.
Years lived with disability
The second component of DALYs is YLDs. YLDs have been estimated for 1160 sequelae of the diseases and injuries in the hierarchical cause list. YLDs are the product of prevalence times the disability weight for a sequela. Prevalence estimation for each sequela begins with a systematic analysis of published and available unpublished data sources for prevalence, incidence, remission, and excess mortality. For most sequelae, estimates have been made based on the database for all age-sex-country-year groups, with a Bayesian meta-regression method developed for the GBD 2010 (DisMod-MR). The meta-regression can handle data reported for any age interval and can use two types of covariates: those that explain true variation in prevalence; and those that explain variation across studies due to study design, case definitions, or diagnostic technology. Nested super-region, region, and country random intercepts are also included.
A map of regions and super-regions is published elsewhere. [86] Where appropriate, DisMod-MR uses data on incidence, prevalence, remission, excess mortality, and cause-specific mortality to generate prevalence estimates assuming these rates are stable over time. Using data on multiple epidemiological parameters to estimate prevalence is especially important when prevalence data are sparse. Where rates are changing rapidly, DisMod-MR can be used to undertake meta-regression without assuming equilibrium rates. Alternative strategies have been used for the prevalence of selected sequelae (see elsewhere for details). [93] DisMod-MR and alternative methods generate uncertainty distributions for the prevalence of each sequela by age, sex, country, and year. For nine residual cause categories such as other mental and behavioural disorders, YLDs have been approximated with the relation between YLLs and YLDs reported for similar disease groupings.
For the GBD 2010, disability weights have been measured for 220 unique health states that encompass the 1160 disease and injury sequelae. The number of health states is lower than the number of sequelae because the same health status such as anaemia appears in the cause sequela list multiple times (eg, mild anaemia from malaria, or mild anaemia from chronic kidney diseases). Disparate outcomes across some diseases have been grouped into a small number of more homogeneous outcomes. For example, disability from all acute infectious disease episodes was captured by a mild, moderate, or severe health state. Disability weights have been generated using data collected from more than 31,000 respondents through population-based surveys in five countries—USA, Peru, Tanzania, Bangladesh, and Indonesia—and an open internet survey. The primary elicitation method used was pairwise comparisons of two randomly selected health states where the respondent selects which health state represents the higher level of health. Results for health-state severities were consistent across levels of educational attainment and cultural groups. [3] Uncertainty in the disability weight for each sequela has been propagated into the estimates of YLDs for each disease and injury. Salomon and colleagues3 provide detail on the methods used to analyse the results of pairwise comparisons to yield disability weights.
Ranking lists
For presentation of the leading causes of DALYs, we need to choose the level in the cause hierarchy at which we rank disorders. Because the leading causes of burden tend to have some influence on the perception of disease-control priorities, the choice of aggregation is at once important and subject to debate. To help convey the complexity of the burden of disease results, we show information at the second level of the GBD cause hierarchy (21 causes); we have also identified a ranking list with 176 causes selected to distinguish and cluster disorders that might have programmatic or public-health significance. We aggregated detailed causes within the broader categories of maternal disorders, diarrhoeal diseases, lower respiratory infections, stroke, and road injury for this reason. The full ranking list is included in the report by Murray and colleagues. [86] Results in the tables are provided for all 291 causes; the ranking list is used only for the figures illustrating the leading cause of DALYs. The 176 causes do not include residual categories such as other parasitic or other cardiovascular diseases because these categories represent complex aggregations of detailed causes for which no clear public health programme exists. The 176 causes along with the excluded residual categories are also mutually exclusive and collectively exhaustive.
Regional ordering and uncertainty
For figures where we present information by region, we order regions by the mean age of death.90 Mean age of death reflects both population age-structure and age-specific death rates and is a simple summary measure of the demographic and epidemiological transition. Mean age of death is a particularly useful metric because average age of the population and age-specific death rates are negatively correlated.
The models used to generate estimates of YLLs and YLDs produce uncertainty intervals that include correlation of uncertainty across age, sex, and time for a given outcome. In the absence of data and a method that would allow one to estimate the correlation of uncertainty between YLLs and YLDs, we had to assume that, for estimating DALYs in an age-sex-country-year-cause, YLL and YLD uncertainty distributions were independent. We computed many different aggregations of DALYs, for example global and regional DALYs for an age group or aggregations for developed or developing regions. For all geographic aggregates, we assumed that uncertainty distributions of the components across countries were independent. In practice, uncertainty from all inputs into the calculations of YLLs, YLDs, and DALYs are propagated with Monte Carlo techniques where 1000 samples are from the posterior distribution. Aggregations are made at the level of the 1000 draws for all estimates that are being summarised. The uncertainty interval (UI) around each quantity of interest is presented as the 2·5th and 97·5th centile values. These ranges can be interpreted as a 95% UI.
Decomposition of change from 1990 to 2010
To help understand the drivers of change in the numbers of DALYs by cause, we have decomposed change from 1990 to 2010 into growth in total population, change in population age-structure and sex-structure, and change in age-specific and sex-specific rates. We compute two counterfactual sets of DALY numbers:(1) a population growth scenario computed as the number of DALYs expected in 2010 if only total population numbers increased to the level of 2010 but the age-sex structure of population stayed the same as in 1990 and age-specific and sex-specific rates remained at 1990 levels and
(2) a population growth and population ageing scenario computed as the number of DALYs expected in 2010, using 1990 age-specific and sex-specific rates and 2010 age-specific and sex-specific population numbers.The difference between 1990 numbers and the population growth scenario is the change in DALY numbers due strictly to the growth in total population. The change from the population growth scenario to the population growth and ageing scenario is the number of deaths due to ageing of the population. The difference between 2010 DALYs and the population growth and ageing scenario is the difference in DALY numbers due to epidemiological change in age-specific and sex-specific death rates. Each of these three differences is also presented as a percentage change with reference to the 1990 observed death number.
Further details on the data and methods used for specific diseases and injuries are available on request.
Role of the funding source
The sponsor of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results

Figure 1 In 2010, there were a total of 2·490 billion DALYs, or 361 DALYs per 1000 population. Globally, 31·2% of DALYs in 2010 were from YLDs and 68·8% from YLLs. YLDs make very little contribution to the burden in the neonatal age groups but increase to a peak in age group 10–14 years when mortality rates are generally the lowest (Figure 1). In nearly all age groups, YLDs make up a larger share of DALYs in women than in men. Globally, YLDs in women caused 50% or more of DALYs up until age 45 years and then declined slowly but still caused about 30% of DALYs over the age of 70 years.

Figure 2 Across broad cause groups, the distribution of DALYs in 2010 reflected a predominance of NCDs globally, with 54% of all DALYs due to non-communicable diseases, compared with 35% due to communicable, maternal, neonatal, and nutritional disorders, and 11% due to injuries. The composition of global DALYs in 2010 shows the diversity of causes that make major contributions to the burden of disease. Cancers and circulatory diseases accounted for 19% of global DALYs, while about a third of the global burden of disease was from other NCDs including chronic respiratory, digestive, neurological, mental and behavioural, endocrine, kidney, musculoskeletal, and other disorders. In the early and late neonatal age groups, neonatal disorders, diarrhoea, lower respiratory infections, and the category other NCDs, which includes congenital anomalies, were most common (Figure 2). For children older than the age of 1 month, the cluster of diarrhoea, lower respiratory infections and other infections, nutritional deficiencies, malaria and neglected tropical diseases, and a diverse set of other causes start to play an increasing part. For young adult men from 15–39 years of age, the main causes of DALYs were HIV/AIDS and tuberculosis, mental and behavioural disorders, road injuries, unintentional injuries other than transport, intentional injuries, and wars or disasters. In young women, the same set of causes plus deaths and YLDs due to maternal disorders occurred. At older ages, cancers, cardiovascular diseases, musculoskeletal disorders, chronic respiratory diseases, digestive diseases, and diabetes are important.

Table 1 The wide range of causes making up the burden of disease is borne out by examining the cumulative burden as a function of a rank list of specific causes. The top ten causes account for 37% of DALYs, the top 25 account for 61% of DALYs, and the top 50 causes account for 78%. Results for all 20 GBD age groups, by male, female, and combined sexes are shown in the appendix. While the results for many causes have public health significance, we highlight causes that lead to more than 15 million DALYs. Tuberculosis accounts for 2·0% of all DALYs, HIV/AIDS 3·3% of DALYs, and malaria 3·3% (Table 1). Diarrhoea and lower respiratory infections were very large causes of burden accounting for 3·6% and 4·6% of global DALYs, respectively. Within the broad group of communicable, maternal, neonatal, and nutritional disorders, meningitis (1·2%), maternal disorders (0·6%), protein-energy malnutrition (1·4%), and iron-deficiency anaemia (1·8%) were all substantial causes. Neonatal disorders collectively caused 8·1% of all DALYs because of the large number of deaths at young ages and some lifelong disability. Each of the four causes in neonatal disorders was a major cause: preterm birth complications (3·1%), neonatal encephalopathy (birth asphyxia and birth trauma; 2·0%), sepsis and other infectious disorders of the newborn baby (1·8%), and other neonatal disorders (1·2%).
Several diseases within the NCD group caused more than 15 million DALYs in 2010. All neoplasms accounted for 7·6% of global DALYs. Of the 28 categories of cancer included in the analysis, four caused more than 15 million DALYs each: stomach cancer (0·7%), other neoplasms (0·7%), liver cancer (0·8%), and trachea, bronchus, and lung cancers (1·3%). Cardiovascular and circulatory diseases ac counted for 11·8% of global DALYs; the major diseases within this group are ischaemic heart disease (5·2%), haemorrhagic stroke (2·5%), ischaemic stroke (1·6%), and hypertensive heart disease (0·6%). The larger bur den of haemorrhagic stroke compared with ischaemic stroke is mostly a function of the younger average age of death for this form of stroke and consequently more YLLs per death. Chronic respiratory diseases as a group accounted for 4·7% of global DALYs, with chronic obstructive pulmonary disease (COPD) making up twothirds of the total and asthma nearly a fi fth of the total. Surprisingly, cirrhosis accounted for 1·2% of global DALYs with a nearly equal share related to hepatitis B, hepatitis C, and alcohol. 3·0% of global DALYs were from neurological disorders; of which a quarter were due to epilepsy and nearly a third were from migraine.
While in some regions dementias were a major cause, at the global level they accounted for 11·3 million DALYs. Mental and behavioural disorders accounted for 7·4% of DALYs; within this large grouping fi ve different diseases caused more than 15 million DALYs each. In order of importance, the main causes were major depressive disorder (2·5%), anxiety disorders (1·1%), drug use disorders (0·8%), alcohol use disorders (0·7%), and schizophrenia (0·6%). Nearly 5·0% of all DALYs were from diseases in the diabetes, urogenital, blood, and endo crine group: the most important diseases were diabetes mellitus (1·9%), chronic kidney diseases (0·8%), and the group of haemo globinopathies and haemolytic anaemias (0·6%). Musculoskeletal disorders accounted for 6·8% of total DALYs. Of this large total, low back pain accounted for nearly half, neck pain a fi fth, and osteo arthritis about 10·0%. A further 5·1% of the GBD was due to causes in the category other NCDs; roughly 30% of which was due each to congenital anomalies, skin diseases, and sense organ diseases.
Injuries collectively caused 11·2% of DALYs with many different injuries making important contributions. The largest was road injuries, which accounted for 27% of the injury total. Within road injuries, nearly equal shares were due to pedestrian injuries, injuries sustained by occupants of three or more wheeled vehicles, and the rest of road injuries. The next most important injury was selfharm (1·5%) followed by falls (1·4%) and inter personal violence (1·0%). Drowning and fi res each accounted for just over 19 million DALYs.
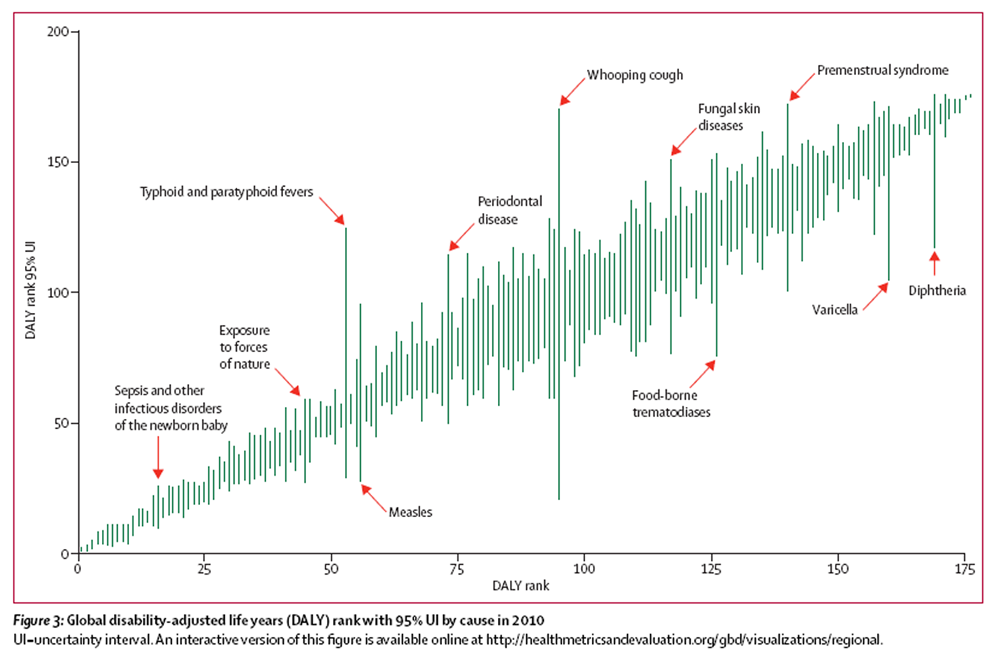
Figure 3 An important innovation in the GBD 2010 is the quantifi cation of uncertainty from all sources entering the estimation of DALYs. Figure 3 shows how the first and second ranked disorders, ischaemic heart disease and lower respiratory infections, have nearly overlapping uncertainty distributions but do not overlap with any of the lower ranked causes. There are many examples of causes with similar ranks where the uncertainty intervals are up to tenfold wider for one compared with another cause. The largest rank uncertainty intervals are for whooping cough (149 ranks), typhoid and paratyphoid fevers (96 ranks), food-borne trematodiasis (77 ranks), fungal skin diseases (75 ranks), premenstrual syndrome (71 ranks), and acute hepatitis E (68 ranks).

Table 2 Global DALYs decreased slightly from 2·503 billion in 1990 to 2·490 billion in 2010 (Table 2). The nearly constant volume of DALYs is due to a near balancing of two key forces: nearly 40% growth in DALYs due to increases in population numbers, ageing of the population, and declines of more than 35% due to changes in age-specific and sex-specific rates. The story is more complicated when we examine the balance of these demographic and epidemiological forces at the level of broad cause groups. Communicable, maternal, neonatal, and nutritional disorders have de clined by more than 25% because population growth has been more than compensated by large drops in expected DALYs due to population ageing and over 50% reductions in DALYs due to declining agespecific and sex-specific rates. For NCDs, both population growth and ageing of the world’s population is driving up DALYs; these factors alone would have increased NCD DALYs by nearly 50%. Declines in age-specific and sex-specific rates overall have meant that this group increased by 25%. Injury DALYs have increased more modestly, driven to a large extent by growth in population numbers and modest declines in rates.
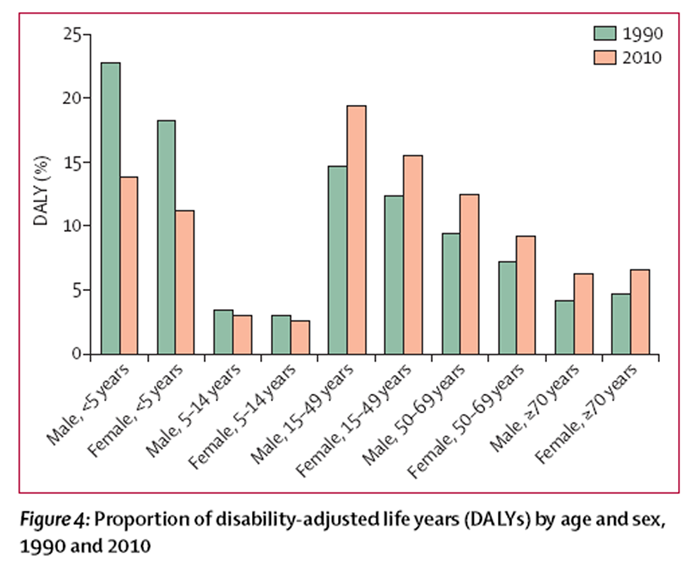
Figure 4 These demographic and epidemiological changes have changed the age distribution of burden. In 1990, 41% of DALYs were due to deaths and disability in children younger than 5 years of age but by 2010, that had decreased to 25% (Figure 4). Over the same interval, the burden of disease in the reproductive age-groups (15–49 years) increased from 27·0% to 35·0% of total DALYs. The shift to burden at older ages is also evident in the age groups 50–69 years and 70 years or older. The slight predominance of burden of 54·4% in male individuals in 1990 increased to 55·0% in 2010 (figure 4). Male burden is higher than female burden in all age groups except in the age groups 75–79 years in 1990 and 80 years or older in 1990 and 2010.
In 1990, 23·3% of DALYs were from YLDs. From 1990 to 2010, YLLs decreased from 1·919 billion to 1·713 billion, and YLDs increased 583 million to 777 million, so that by 2010, YLDs accounted for 31·2% of global DALYs, reflecting the relative increase of non-fatal versus fatal loss of healthy life years. As a consequence of these substantial structural changes in the burden of disease from younger to older ages and from YLLs to YLDs, the broad composition of the burden of disease has shifted from communicable, maternal, neonatal and nutritional disorders to NCDs and injuries. In 1990, 47% of DALYs were caused by communicable, maternal, neonatal and nutritional disorders, 43% from NCDs, and 10% from injuries. By 2010, this had shifted to 35% caused by communicable, maternal, neonatal, and nutritional disorders, 54% by NCDs, and 11% by injuries. The main changes from 1990 to 2010 are the reductions in infectious diseases, mostly among children, an increase in the HIV/AIDS and tuberculosis category, and increases in a diverse set of NCD and injury categories. Maternal disorders declined from 0·9% of DALYs in 1990 to 0·6% in 2010. From 1990 to 2010, mental and behavioural disorders increased from 5·4% to 7·4% and musculoskeletal disorders increased from 4·7% to 6·8%. Neurological disorders including dementia increased from 1·9% to 3·0% over the two decades. Increases in cardio vascular diseases were modest from 9·6% to 11·8%. Uninten tional injuries including transport injuries increased from 7·6% to 8·1% in 2010.
Across the 1000 draws of the entire study results, each cause has been ordered in terms of total DALYs (figure 5). Causes in the figure are ordered by their mean rank across the 1000 draws. The order based on the mean rank across draws is not the same as the order based on the mean value of DALYs shown in table 1. For example, in 2010, malaria caused slightly more DALYs than HIV/ AIDS in table 1; HIV/AIDS, however, in this figure ranks fi fth and malaria seventh because across the 1000 draws HIV/AIDS is more likely to rank higher in the list than malaria because of the much greater uncertainty around the number of DALYs due to malaria. Causes in figure 5 are connected with lines to show changes in ranks over the two decades. Causes that moved into or dropped out of the top 25 ranks between 1990 and 2010 are listed at the bottom. Mean of the ranks of a disease across the 1000 draws of DALYs, the 95% UI in ranks, and the rank of the global mean value are shown for 1990 and 2010. The final column on the right-hand side provides the percentage change in the number of global DALYs for each cause from 1990 to 2010. As a general observation, most of the NCDs are rising in the rank list and most but not all communicable, maternal, neonatal, and nutritional disorders are declining. Notable exceptions are the stable and increasing ranks for malaria and HIV/AIDS, respectively. Among NCDs at a global level, only COPD and congenital anomalies have declined. Given that lung cancer is increasing from 24th to 22nd, the decline in COPD is worth noting. The decline is driven by the reduction of other determinants of COPD such as household air pollution in India and China, despite increasing cumulative exposure to tobacco.
In 1990, the leading cause of burden was lower respiratory infections, for which 81·4% of the total burden occurred among children younger than 5 years of age. The second leading cause was diarrhoeal diseases with 82·0% also occurring in children younger than 5 years of age. In 2010, these two causes remained among the top four causes of burden, but the absolute number of DALYs from these disorders has declined over the two decades by more than 40% in both cases. For the two cardiovascular causes now in the first and third slots, ischaemic heart disease and stroke, most of this burden is from YLLs not from YLDs, 93·2% and 95·7%, respectively. Both causes are increasing in absolute terms because of demographic changes; ischaemic heart disease increased by 29% and stroke by 19% over the two decades. HIV/AIDS went from the 33rd largest cause of burden in 1990 to the fifth largest cause in 2010. This rank is despite major declines in HIV/AIDS mortality since 2005. Malaria remains at seventh rank from 1990 to 2010, although uncertainty around malaria burden is large, spanning from the third to the eleventh rank. Of the malaria burden, 22·6% occurs in adults over age 15 years, a previously unrecognised cause of adult disease burden. [94] Measles dropped from the 16th to the 56th cause.
Some causes not included in the top 25 list have changed substantially over the period 1990 to 2010. The 15 causes with the largest increases include two causes of blindness or low vision: glaucoma and macular degeneration. Age-sex specific prevalence rates for these disorders have not increased; the rise in burden is completely due to the increase in the world population in the oldest age groups. Two major neurological disorders concentrated in older age-groups are also in the list of top increases—dementia and Parkinson’s disease. Atrial fibril lation, peripheral vascular disease, and benign prostatic hyperplasia also increased substantially over the two decades. Not surprisingly, in view of the time course of the epidemic, HIV/AIDS increased by 351%. Kidney cancer is the one cancer to be included in this list of top increases. Conversely, the largest declines have occurred for several of the infectious diseases including measles, tetanus, rabies, whooping cough, diarrhoeal diseases, lower respiratory infections, syphilis, leishmaniasis, and ascariasis. Large upward trends in exposure to forces of nature and downward trends in collective violence reflect the stochastic nature year by year in these causes.
East, southeast, and south Asia made up 52·7% of the global burden in 1990, declining to 48·3% in 2010 (table 3). The absolute number of DALYs has also declined in western and central Europe, central Asia, and Andean Latin America. Tropical Latin America, North Africa and Middle East, and eastern sub-Saharan Africa, have barely changed over the interval although in all three regions a substantial change in the age-structure and cause composition has occurred. Other regions have seen increases in the number of DALYs. The largest increases have been in western, southern, and central sub-Saharan Africa. The increase in the Caribbean is largely related to the Haiti earthquake in 2010 because of the increase in the death rate and the fact that Haiti accounts for 26·3% of the Caribbean population. Most high-income regions have also seen modest increases in the number of DALYs. DALYs per 1000 confirm that these increases are largely driven by population growth; only in three regions, namely the Caribbean, southern sub-Saharan Africa, and eastern Europe, did the rate of DALYs per 1000 increase substantially over the 20-year period as a proportion of population. Declines in DALYs per 1000 have generally been larger in developing country regions than in high-income country regions. These declines are partly due to the effect of population ageing lowering DALYs per 1000 from communicable, maternal, neonatal, and nutritional disorders, which are highest in the young age groups.
The share of burden from non-fatal health outcomes has generally increased from 1990 to 2010 in nearly all regions (figure 6); declines in southern sub-Saharan Africa can be related to the large HIV-related increase in mortality and in the Caribbean due to mortality from the 2010 Haiti earthquake. Figure 6, in which the regions are ordered by the mean age of death, shows that in general the share of burden from disability increased with the demographic and epidemiological transition. In 2010, the fraction of DALYs due to YLDs varied widely, from 55% in Australasia to 15% in central sub-Saharan Africa. Australasia had a higher ratio than high-income Asia Pacific; both had low mortality levels but higher YLD rates prevailed in Australasia. In eastern Europe, the fraction due to YLDs has not increased noticeably from 1990 to 2010 because of the rise in adult mortality in the region over this period, especially for men.
The global shift in the burden of disease from communicable, maternal, neonatal, and nutritional disorders to NCDs and injuries masks enormous epidemiological heterogeneity in the leading causes of burden in different regions. In the regions with an advanced demographic and epi demiological transition (high-income Asia Pacific, western Europe, Australasia, high-income North America, and central Europe), communicable, maternal, neonatal, and nutritional disorders account for less than 7% of DALYs (figure 7). Cancer and cardiovascular diseases account for a further 36% of DALYs. Mental and behavioural disorders account for 11% and musculo skeletal disorders account for 13%. Injuries make up about 11%. At the other end of the epidemiological transition, in eastern, western, and central sub-Saharan Africa communicable, maternal, neonatal, and nutritional disorders account for 67–71% of DALYs. A middle group of regions have a transitional volume of burden due to com municable, maternal, neonatal, and nutri tional disorders. Comparison of 1990 and 2010 shows the most profound shifts in these tran sitional regions, moving from a profile with substantial burden from infectious diseases predom inantly in chil dren and neonatal causes, to a much greater dominance of injuries, musculoskeletal disorders, mental and behavioural disorders, as well as cancers and cardio vascular diseases. The great rise in HIV/AIDS and tuberculosis is also evident in southern and eastern sub-Saharan Africa. In 2010, deaths from the Haiti earth quake accounted for the substantial change in cause composition in the Caribbean from 1990 to 2010.
Although a strong tendency exists for the cancer DALY rate to increase with the demographic and epidemiological transition, there is notable variation. Oceania and the Caribbean seem to have higher than expected rates and central Latin America, lower rates (figure 8). Lung, colon and rectum, breast, and pancreatic cancers are associated with DALY rates that are generally higher in the high-income regions, while cervical cancer is lower. Liver, stomach, leukaemia, and skin cancers show strong geographic variation. Among high-income regions, Asia Pacific has a substantially different pattern with more stomach and liver cancer, and less breast cancer. Oceania has relatively high rates of liver, leukaemia, and cervical cancers.
The GBD study provides results for a set of diseases that are much smaller in magnitude at the global level but are important causes of burden in communities at risk. The neglected tropical diseases excluding malaria make up 1·0% of global DALYs (figure 9). Rates of neglected tropical diseases vary across regions by 961 fold. The highest rates were in central sub-Saharan Africa, largely because of the combination of schistosomiasis, onchocerciasis, African trypanosomiasis, and hookworm. Globally, leishmaniasis, schistosomiasis, hook worm, lymphatic filariasis, and foodborne trematodiases are the dominant causes in this grouping. In view of the focal nature of the transmission of many of these diseases, the regional pattern varies substantially. As most of these diseases cause limited mortality, the neglected tropical diseases highlight why quantification of the disability from diseases is important.
The order of causes in figure 10 follows the global ranking of burden shown in figure 5. All causes that appear in the top 25 in any region are included in figure 10. The cells in the figure have been colour coded to help identify different patterns in each region. Eight causes appear as the leading cause in at least one region. Ischaemic heart disease is ranked first in seven of 21 regions. Lower respiratory infections are ranked first in Andean Latin America, south Asia, and Oceania. Malaria is ranked first in two regions: western and central sub-Saharan Africa. HIV/AIDS is ranked first in eastern and southern sub-Saharan Africa. Interpersonal violence is ranked first in central Latin America and ranked second in tropical Latin America. Due to the Haiti earthquake in 2010, forces of nature ranks first for the Caribbean. Low back pain is a top ten cause in 15 regions. Falls are a top ten cause in three regions. A total of 33 causes appear in the top ten in at least one region. This extended list includes disorders such as chronic kidney diseases, drug use disorders, cirrhosis, dementia, meningitis, liver cancer, stomach cancer, and colon and rectum cancers.
Discussion
The GBD 2010 estimates that the number of DALYs for the world in 1990 was 2·503 billion, having decreased by 0·5% in 2010. Relatively small changes in the number of DALYs have occurred because the increase in global population has been largely balanced by a decrease in age-sex-specific DALY rates. The differential effect of population growth, population ageing, and changes in age-sex-specific rates have led to striking changes in the profile of burden in every dimension. Over two decades, the burden has shifted substantially from communicable, maternal, neonatal, and nutritional disorders towards NCDs. A much larger fraction of the burden is now caused by disability rather than premature mortality. Burden has shifted away from death of children younger than 5 years of age to death and disability in the reproductive age groups; nonetheless, a quarter of the burden is still caused by disease and injury in children younger than 5 years of age. Because of the richer dataset, improved methods, and more extensive cause list, our results for 1990 to 2010 supersede and replace previous GBD studies; comparisons with previous studies to assess change over time would not be valid.
On top of a general pattern of the demographic and epidemiological transition associated with both mortality decline and fertility decline, substantial regional heterogeneity exists in the diseases and injuries that cause burden. HIV/AIDS is one vivid example that has come to be the dominant cause of burden in eastern and southern sub-Saharan Africa. Interpersonal violence is a leading cause in central Latin America (rank 1) and tropical Latin America (rank 2), and in southern sub- Saharan Africa (rank 5); the pattern of interpersonal violence across regions is unrelated to metrics of the epidemiological and demo graphic transition. Self-harm is a top ten cause of burden in high-income Asia Pacific (rank 5), eastern Europe (rank 6) and central Europe (rank 11). Cirrhosis is an important cause in central Asia (rank 9), central Europe (rank 10), eastern Europe (rank 11), and central Latin America (rank 12). Drug use disorders are especially important in Australasia (rank 11) and high-income North America (rank 11). Site-specific cancers show substantial regional heterogeneity. The leading cancer across regions ranges from lung to liver to stomach and colon and rectum.
Some diseases show a strong relation between prevalence and mortality with age. As the number of indi viduals aged 75 years and older in the world increased from 119 million in 1990 to 206 million in 2010, it has driven up the burden of these diseases substantially. The most notable diseases include the various causes of blindness and low vision but also several neurological disorders. The rise of dementia and Parkinson’s disease is almost entirely attributable to population ageing be cause age-specific rates have remained constant. In view of the global shifts in fertility and declines in age-specific mortality, we can expect the numbers of individuals with age-related disorders to increase sub stantially in coming decades. This shift in numbers of people with certain disorders will have sub stantial implications for health-service planning.
At least partly viewed through the lens of the Millennium Development Goals (MDGs), the world has paid increased attention to the mortality of children younger than 5 years of age, maternal mortality, HIV/ AIDS, tuberculosis, and malaria. Collectively the MDGrelated causes of burden account for 742 million DALYs in 2010, or 29·8% of the total burden of disease — this burden includes YLLs from all causes in children younger than 5 years of age and DALYs from maternal disorders, HIV/AIDS, tuberculosis, and malaria. Progress has clearly been made. In 1990 these disorders accounted for 1096 million DALYs or 43·8% of the total burden. Although we are unlikely to achieve most of the health-related MDG targets by 2015, the burden of these disorders has declined by nearly 32·0% from 1990 to 2010 and will probably decline further by 2015 in view of current trends. More than two-thirds of global DALYs now arise from disorders not targeted in the MDGs. As 2015 nears and the world is discussing goals for the post- MDG period, addressing the leading, and often largely preventable, causes of the non-MDG health spectrum, especially NCDs and injuries, should be given greater priority than hitherto. When examined at a regional level (figure 11), the issue is even starker. In 2010, the fraction of the burden of disease that is related to disorders targeted in the MDGs ranges from 68·9% in western sub-Saharan Africa to 1·7% in high-income Asia Pacific. In fi ve of 21 regions, the burden of MDG-related disorders exceeds a third of regional DALYs: the four sub- Saharan Africa regions, and south Asia. This regional hetero geneity shows how it will be important for post-2015 development goals to reflect the widely diff ering disorders across regions in setting targets.
The findings from this study have implications for health system investment decisions, including health manpower needs and the content of medical education. Many systems are already grappling with the challenges posed by rising numbers of cardiovascular events and cancers; these findings also highlight the importance of health-care professionals who will service the specialties of trauma, rehabilitation, mental health, musculoskeletal disorders, and diabetes. More generally, the shifting burden of disease driven by population ageing and differential rates of decline in age-specific rates that are greater for communicable, maternal, neonatal, and nutritional disorders than for NCDs also has implications for any health system’s capital investments. These invest ments will often be used over decades so that they need to reflect future burden. Within professions, the shifting burden should also be reflected in the content of education for health professionals. The pace of demo graphic and epidemiological change is fast enough that a forwardlooking assessment of the burden should be incorporated in the reform of health professional education on a region-by-region basis. [95]
The burden of musculoskeletal disorders is much larger than in previous GBD assessments. In the 2004 revision of the GBD study, this group of disorders was estimated to account for 2·0%, compared with 6·8% in this study. This much higher share relates to three factors. First, there has been a much more comprehensive and systematic assessment of the epidemiological data. These data show that low back pain, neck pain, osteoarthritis, and other musculoskeletal disorders are extremely common in nearly all populations. Second, the disability weights assigned to these disorders — which cause pain, discomfort, lack of mobility, anxiety, and sleeplessness — in the population-based surveys are higher than those based on the judgment of health-care professionals. Finally, in previous assessments that focused on incidence, the duration of symptoms was probably systematically underestimated. These disorders account for a substantial number of health-care visits and cost in populations with access to medical care. [96, 97] The burden is likely to grow steadily because of rising rates with age, little change over time, and an ageing world population. In view of the epidemiological pattern and associated costs, health-care systems will need to develop a coherent policy for dealing with musculoskeletal disorders. Prioritisation of research on the most effective and affordable strategies is urgently needed to deal with these disorders.
A key fi nding of the GBD 1990 and 2000 studies was the large unrecognised burden of mental illness in developed and developing countries—8·5% of DALYs in the GBD 1990 study and 10·1% in the GBD 2000 study. These results were reported for DALYs with discounting and ageweighting. Age-weighting assigns maximum value to young and middle-aged adults in whom the prevalence of mental illness is high. Without age-weighting and discounting, the burden of mental illness in the GBD 1990 study was 5·7%. Despite the switch in the GBD 2010 study to a base case for DALY computation of no discounting and no age-weighting, mental and behavioural disorders account for 7·4% of global DALYs in 2010. This study has expanded the set of disorders carefully assessed to include many disorders previously crudely estimated in a residual category. Newly added disorders include all anxiety disorders com pared with only three in the earlier studies, childhood disorders, and eating disorders. Some disorders such as major depressive disorder have a higher prevalence than pre viously estimated. Using consistent defi nitions in this study over time for the mental and behavioural disorders, the number of DALYs for this group increased by 38% from 1990 to 2010. The drivers of the increase are the combination of population growth, shift in age structure towards the age groups at highest risk, and relatively stable age-specific prevalence rates—although notable fluctuations exist in drug use disorders over time. We can expect that the absolute number and share of burden attributable to mental and behavioural disorders, already substantial, will probably steadily increase in the future. Despite increased global attention to mental health in the past decade, [17] practical strategies for managing these disorders in low-income and middle-income countries are urgently needed.
Road injury accounts for 75·5 million DALYs in 2010, up from 56·7 million in 1990. To put road injury in context, it accounts for 53% more burden than tuberculosis. Road injury shows a classic inverted U-shaped pattern with the largest DALY rates and highest rank as a cause of burden in regions that are upper low-income or middle-income. Nevertheless, even in the demographically and epidemiologically advanced regions, road injury is in the top 16 causes. The distribution of road injury by specific subcause is also important for policy: in seven developing regions more than 40% of road injury deaths are in pedestrians including all sub-Saharan African regions, south and east Asia, and Andean Latin America. Motorised two-wheel vehicles account for more than 20% of road injury deaths in southeast and east Asia and tropical Latin America. The local patterns of road injury and publications on road safety [98, 99] argue that most road injury is preventable. Some high-income countries such as Australia have been able to reduce the death rate from road injuries by 43·7% since 1990, providing a population level dem onstration that many deaths are preventable. Various global initiatives on road safety have been launched [100, 101] but they remain relatively weakly funded and are yet to have a demonstrable effect on the rising burden from road injury globally. Continued attention from both the health sector and the transport sector will be needed to address this growing challenge.
Interpersonal violence in 2010 ranks 27th across causes at the global level; in view of the fact that 81% of the DALYs due to interpersonal violence are in male individuals, it is the 21st ranked cause in male individuals and 49th in female individuals. This global figure masks enormous inter-regional variation in the extent of interpersonal violence. Three regions—central and tropical Latin America and southern sub-Saharan Africa—have violence as a top five cause of burden. Ecological analyses of the root causes of violence [102–106] have helped elucidate risk factors for different forms of violence and have been useful for exploring the determinants of within-country variation. [107] Few studies, however, help to explain why violence is such a dominant factor in population health in specific countries and regions. More robust research on this topic, as well as its relation to social and political changes and drug markets, would be a valuable addition to the public health literature. Increased links between public health researchers and social scientists working in this complex field could make this research more productive. A key challenge for this area, nevertheless, will be proposing, testing, and assessing effective policy interventions that stem from increased understanding of the broader determinants.
Among the top cancer causes of DALYs, liver cancer and pancreatic cancer DALYs have increased the most. Stomach cancer is declining, and lung, colon and rectum, breast, and brain cancer increased by about 35% from 1990 to 2010. For smaller causes, kidney cancer, pros tate cancer, liver cancer secondary to hepatitis C, and non-melanoma skin cancer are the only causes of cancer that have increased by more than 50% from 1990 to 2010. Many researchers might have expected the burden of liver cancer secondary to hepatitis B to decrease because of expansion of hepatitis B vaccine coverage. Burden, how ever, has increased from 1990 to 2010 by over 45%. The increase can be understood in terms of population growth in areas with substantial prevalence of hepatitis B and the long lag between childhood immunisation for hepatitis B and reductions in adult liver cancer deaths some 30–50 years later. Increases are also in part related to other causes of liver cancer, including hepatitis C and alcohol.
The downward trend in COPD rates in east Asia and upward trend in lung cancer in this region need explanation. In addition to tobacco con sumption, other factors probably contribute to levels and trends in COPD possibly including exposure to particulates from biomass and coal fuels. [108–110] Relations might exist between exposure to respiratory infections as a child and adult COPD that can also alter secular trends. [111] Historical analysis, for example, for the UK and Australia also suggests that cause of death declined from 1900 to 1940 then increased until the 1980s from rising tobacco consumption. [112, 113] The same set of determinants that account for the downward trend in high-income coun tries could be occurring in Asia.
In a study covering 291 diseases and injuries, 1160 sequelae, 20 age groups, both sexes, and 21 regions, many limitations reflect the availability, representativeness, and broad quality of the data. It is beyond the scope of this summary article to describe all the specific limitations associated with data availability, efforts to enhance quality and comparability of data, and model specification for estimation. More detail is provided in accompanying articles on all-cause mortality, causes of death, disability weights, and YLDs. [3, 11, 90, 92] Future causespecifi c publications will also provide a forum for exploring disease or injury-specific limitations. More generally, we have tried for the first time to quantify uncertainty as a way to inform the user of the strength of the evidence on the burden of a given disease or injury. Relative uncertainty varies widely across causes. For example, only four studies are available on onchocercal skin disease but for epilepsy there are 353 studies. For some causes of death like ischaemic heart disease, the models have very small prediction error whereas for others like dengue or rabies it is very large. The width of the UIs is a useful guide to where the limitations of the analysis are greatest. Of course, we might be missing sources of uncertainty for some disease and injury sequelae. As with any systematic analysis, selection bias in data collection can lead to systematic biases in the results that are not reflected in the statistical UIs. In computing UIs, we assumed that uncertainty distributions for YLLs and YLDs were independent; this assumption, however, could be incorrect.
Countries with poor data on mortality and causes of death might be more likely to have poor data on the prevalence of sequelae. Empirical information to establish the cor relation, however, is extremely limited. If data quality between causes of death and prevalence are correlated, our UIs could be underestimated. For the YLD com ponent, disability weights play a crucial role; to the extent that lay descriptions used in the measurement of disability weights do not reflect the average experience of an individual with a sequela then the YLDs could be overestimated or underestimated. The shift in burden towards YLDs from 1990 to 2010 is not a function of the disability weights because the same weights are used for computing 1990 and 2010 burden. However, since the disability weights for this study are on average somewhat lower than the GBD 1990 disability weights, this shift would have seemed greater with the older, larger weights. For the first time in the GBD study, we have taken into account comorbidity. These corrections have reduced the number of YLDs that would be estimated without taking into account comorbidity. Because of limitations of data, we have only been able to take into account independent comorbidity within an age-sex group.
The heterogeneity across the 21 regions in the burden of disease highlights how important it will be to make estimates at the national level. Two strategies exist for national estimation: using the information collected in the GBD 2010 to report national burden results, and national burden of disease studies that start with collection and analysis of all local sources. Both strategies are useful. The wealth of data on causes of death and within-region variation in prevalence of disease and injury sequelae can be used to generate informed national estimates building on the GBD 2010 results. These estimates can be immediately useful for enriching a range of national policy debates but can also serve as an informative starting point for an in-depth national burden of disease study. Many countries will also be keenly interested in estimating disease burden for subpopulations on the basis of geography, ethnic group, and socioeconomic status. Capacity and methods to undertake this type of analysis need to be created or strengthened through appropriate training. The new generation methods and standardisation of approaches will make it easier than in the past to undertake comparable, comprehensive, and consistent national assessments.
The results of the GBD study show a truism known to everyone trained in clinical practice that also applies to population health: that individuals and communities suff er from a wide range of disorders. Clinical subspecialties have emerged in modern medicine to deal with some of this complexity at the level of individual patients. One of the fundamental challenges for the global health system and for national health systems is responding to the diversity of urgent health needs for communities. The GBD study provides quantifi cation of this diversity and reminds us that the organised social response to health problems must deal with a wide array of medical and public health priorities for action. Regular updating of the GBD study is an important way that the world can track many different health problems without the risk of a limited set of temporary priorities capturing all of our attention. Regular updates would provide a mechanism both to assess the latest evidence but also to promote accountability of health systems for achieving reduc tions in the burden of disease. Furthermore, despite this complexity and diversity, important health challenges are readily identifi able for which tech nologies and knowledge exist to substantially reduce or eliminate their impact on burden of disease rankings. The sustained com mit ment of governments, donors, and the public health community to do so is crucial, on the basis of the essential health in telligence that regular burden of disease updates can provide.
Acknowledgments
We would like to thank the countless individuals who have contributed to the Global Burden of Disease 2010 study in various capacities. We would like to specifi cally acknowledge the important contribution to this work from multiple staff members of the World Health Organization. We also wish to express our gratitude to the following organisations that hosted consultations during the fi nal stages of the analytic process, providing valuable feedback about the results and the data to improve the study’s fi ndings overall: Pan American Health Organization; Eastern Mediterranean Regional Offi ce of WHO; UNAIDS; Ministry of Health, Brazil; China Centers for Disease Control; and the University of Zambia. We would like to thank Lori M Newman, Jördis Ott, Poul Erik Petersen, Shekhar Saxena, and Gretchen A Stevens for their collaboration and input into the analyses and estimates. Finally, we would like to acknowledge the extensive support from all staff members at the Institute for Health Metrics and Evaluation and specifi cally thank: James Bullard, Andrew Ernst, and Serkan Yalcin for their tireless support of the computational infrastructure required to produce the results; Linda A Ettinger for her expert administrative support in order to facilitate communication and coordination amongst the authors; Peter Speyer, Abigail McLain, Katherine Leach-Kemon, and Eden Stork for their persistent and valuable work to gain access to and catalog as much data as possible to inform the estimates; and Erin C Mullany for her systematic eff orts in organising drafts of papers, formatting correspondence with expert groups, and preparing the fi nal manuscript. The following individuals would like to acknowledge various forms of institutional support. J P Abraham, B Bartels, and P Yeh recognise the support of the World Bank Global Road Safety Facility and Department of Global Health & Population, Harvard School of Public Health, and the World Health Organization Violence and Injury Prevention. B Bikbov acknowledges support from the Moscow State University of Medicine and Dentistry, Moscow, Russia; Academician V I Shumakov Federal Research Center of Transplantology and Artifi cial Organs, Moscow, Russia; International Society of Nephrology. R Bourne acknowledges the Vision & Eye Research Unit, Postgraduate Medical Institute, Anglia Ruskin University, Cambridge, UK. S Brooker is supported by a Wellcome Trust Senior Fellowship in Basic Biomedical Science (098045). T S Brugha received funding from the Department of Health London, for the National Health Service Information Centre, by the University of Leicester. R Buchbinder is partially funded by an Australian National Health and Medical Research Council (NHMRC) Practitioner Fellowship, Monash University, and Cabrini Health. P Burney and D Jarvis acknowledge the Chronic Respiratory Disease group received funding from the BUPA Foundation. They had no role in study design, data collection and analysis, interpretation of data, decision to publish, or preparation of the manuscript. C Cella, M Cortinovis, F Gaspari, V Miglioli, and N Perico, on behalf of the entire Genitourinary Expert Group, acknowledge the International Society of Nephrology (ISN). H Chen acknowledges that his participation in this study was in part supported by the intramural research program of the NIH, the National Institute of Environmental Health Sciences. L E Coff eng, and W A Stolk received fi nancial support from the Africa Programme for Onchocerciasis Control (WHO/APOC) for their work on onchocerciasis. B C Cowie received institutional support from the Victorian Infectious Diseases Reference Laboratory, Melbourne, Australia. M Cross and L March acknowledge the University of Sydney (USYD); Institute of Bone and Joint Research, University of Sydney, Department of Rheumatology, Royal North Shore Hospital, St Leonards NSW 2065 Australia. N Dahodwala was supported by NIH grant K23 AG034236 and the Parkinson Council while working on this project. L Degenhardt is supported by an Australian NHMRC Senior Research Fellowship and funding to support her work for illicit drug dependence was provided by the Australian National Drug and Alcohol Research Centre of the University of New South Wales, Australia. R Dellavalle was supported by the US Department of Veterans Aff airs while contributing to this study. S Derrett acknowledges the Health Research Council of New Zealand and the University of Otago for their support. V Feigin and R Krishnamurthi were supported by the National Institute for Stroke and Applied Neurosciences, AUT University. E Fevre acknowledges the Wellcome Trust for grant 085308. W Hall was supported by an NHMRC Australia Fellowship. R Havmoeller was supported by a grant from the Swedish Research Council (#2011-1071). D Hoy was supported by the Bill and Melinda Gates Foundation and the Australian National Health and Medical Research Council. K H Jacobsen was supported by the World Health Organization for her work on hepatitis A. N Kawakami notes that the collection of data ultimately used in this study was supported by the following grants: The World Mental Health Japan (WMH-J) is supported by the Grant for Research on Psychiatric and Neurological Diseases and Mental Health (H13-SHOGAI-023, H14-TOKUBETSU-026, H16-KOKORO-013) from the Japan Ministry of Health, Labour, and Welfare. He would like to thank staff members, fi eld coordinators, and interviewers of the WMH Japan 2002–2004 Survey. L L Laslett is supported by an Australian Government Australian Postgraduate Award. She also notes that the TasOAC study, the results of which were used in this research, was supported by the National Health and Medical Research Council of Australia; Arthritis Foundation of Australia; Tasmanian Community Fund; Masonic Centenary Medical Research Foundation, Royal Hobart Hospital Research Foundation, and University of Tasmania Institutional Research Grants Scheme. R Malekzadeh received funding from a research grant of Tehran University of Medical Sciences to do the related studies. R Matzopoulos acknowledges the two institutions that support his research work: South African Medical Research Council Burden of Disease Research Unit; and the University of Cape Town School of Public Health and Family Medicine. T Merriman acknowledges the Health Research Council of New Zealand. K Naidoo was supported by the Brien Holden Vision Institute. P Nelson was supported by the National Drug and Alcohol Research Centre (UNSW, Australia). R G Nelson acknowledges his research was supported in part by the Intramural Research Program of the National Institute of Diabetes and Digestive and Kidney Diseases. C Olives was funded in part by the Biostatistics, Epidemiologic and Bioinformatic Training in Environmental Health Training Grant (ES015459). D Ozgediz acknowledges the staff and collaborators at the Mulago Hospital and Makerere University in Kampala, Uganda. K Pesudovs received institutional support from Flinders University. R Room’s position at the University of Melbourne and Turning Point Alcohol and Drug Centre is funded by the Foundation for Alcohol Research and Education and the Victorian Department of Health. J A Salomon received support from the Burke Global Health Fellowship while working on this study. U Sampson received funding support from the Harold Amos Medical Faculty Development Award of the Robert Wood Johnson Foundation and the Vanderbilt Clinical and Translational Scholars Award. L Sanchez-Riera acknowledges the Spanish Society of Rheumatology (Sociedad Espańola de Reumatología). M Segui-Gomez’s participation was partly supported by funds from the European Center for Injury Prevention, Universidad de Navarra. E Smith acknowledges the Department of Health and Ageing, Commonwealth Government of Australia, Institute of Bone and Joint Research (IBJR), University of Sydney (USYD). G D Thurston was supported in part by Center grant ES00260 from the National Institute of Environmental Health Sciences. D J Weatherall was supported by the Wellcome Trust UK, the Medical Research Council UK and the Anthony Cerami and Ann Dunne Research Trust.
References:
Murray CJ, Salomon JA, Mathers CD, Lopez AD. Summary measures of population health: concepts, ethics, measurement and applications. Geneva: WHO, 2002.
Mathers CD, Sadana R, Salomon JA, Murray CJ, Lopez AD. Healthy life expectancy in 191 countries, 1999. Lancet 2001; 357: 1685–91.
Salomon JA, Vos T, Hogan DR, et al. Common values in assessing health outcomes from disease and injury: disability weights measurement study for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2129–43.
Chamie M. What does morbidity have to do with disability? Disabil Rehabil 1995; 17: 323–37.
Grosse SD, Lollar DJ, Campbell VA, Chamie M. Disability and disability-adjusted life years: not the same. Public Health Rep 2009; 124: 197–202.
Leonardi M, Bickenbach J, Ustun TB, Kostanjsek N, Chatterji S, and the MHADIE Consortium. The defi nition of disability: what is in a name? Lancet 2006; 368: 1219–21.
Thomas C. How is disability understood? An examination of sociological approaches. Disabil Soc 2004; 19: 569–83.
Murray C, Evans D. Quantifying individual levels of health: definitions, concepts, and measurement issues. In: Health systems performance assessment: debates, methods and empiricism. Geneva: World Health Organization, 2003: 301–18.
Murray CJ, Lopez AD. The Global Burden of Disease: a comprehensive assessment of mortality and disability from diseases, injuries, and risk factors in 1990 and projected to 2020. Boston: Harvard School of Public Health on behalf of the World Health Organization and the World Bank, 1996.
Murray CJ, Lopez AD. Global Health Statistics: A compendium of incidence, prevalence and mortality estimates for over 200 conditions. Boston: Harvard School of Public Health on behalf of the World Health Organization and the World Bank, 1996.
Murray CJ, Lopez AD. Mortality by cause for eight regions of the world: Global Burden of Disease study. Lancet 1997; 349: 1269–76.
Murray CJ, Lopez AD. Regional patterns of disability-free life expectancy and disability-adjusted life expectancy: Global Burden of Disease Study. Lancet 1997; 349: 1347–52.
Murray CJ, Lopez AD. Global mortality, disability, and the contribution of risk factors: Global Burden of Disease Study. Lancet 1997; 349: 1436–42.
Murray CJ, Lopez AD. Alternative projections of mortality and disability by cause 1990-2020: Global Burden of Disease Study. Lancet 1997; 349: 1498–504.
World Health Organization. The world health report 1999— making a diff erence. http://www.who.int/whr/1999/en/whr99_ en.pdf (accessed July 9, 2012).
World Health Organization. The world health report 2000— health systems: improving performance. http://www.who.int/ whr/2000/en/whr00_en.pdf (accessed July 9, 2012).
World Health Organization. The world health report 2001—mental health: new understanding, new hope. http://www.who.int/ whr/2001/en/whr01_en.pdf (accessed June 25, 2012).
World Health Organization. The world health report 2002— reducing risks, promoting healthy life. http://www.who.int/ whr/2002/en/whr02_en.pdf (accessed July 9, 2012).
World Health Organization. The world health report 2004— changing history http://www.who.int/whr/2002/en/whr02_en.pdf (accessed July 9, 2012).
Lopez AD, Ahmad OB, Guillot M, et al. World mortality in 2000: life tables for 191 countries. Geneva: World Health Organization, 2002.
World Health Organization. Regional burden of disease estimates for 2004. http://www.who.int/healthinfo/global_burden_disease/ estimates_regional/en/index.html (accessed July 5, 2012).
World Bank. World Development Report 1993. Investing in health: world development indicators. Oxford: Oxford University Press, 1993.
Mathers CD, Vos ET, Stevenson CE, Begg SJ. The burden of disease and injury in Australia. Bull World Health Organ 2001; 79: 1076–84.
Mathers CD, Vos ET, Stevenson CE, Begg SJ. The Australian Burden of Disease Study: measuring the loss of health from diseases, injuries and risk factors. Med J Aust 2000; 172: 592–96.
Begg SJ, Vos T, Barker B, Stanley L, Lopez AD. The burden of disease and injury in Australia in the new millennium: measuring health loss from diseases, injuries and risk factors. Med J Aust 2008; 188: 36–40.
Gadelha AMJ, Leite I da C, Valente JG, Schramm JM de A, Portela MC, Campos MR. Projeto Carga de Doenca: relatorio fi nal do projeto—estimativa da carga de doenca do Brasilů, 1998. http:// www.scribd.com/doc/2350704/Projeto-Carga-de-Doenca-Fiocruz (accessed June 26, 2012).
Concha Barrientos M, Aguilera Sanhueza X, Salas Vergara J. Estudio de Carga de Enfermedad, Informe fi nal. Estudio Prioridades de Inversión en Salud. República de Chile: Ministerio de Salud, 1996.
Ministerio de Salud. Estudio de carga de enfermedad y carga atribuible, Chile. Informe Final. Chile: Ministerio de Salud, 2007.
Escobar M, Gallardo H, Giraldo G, Londońo J, Rodríguez J. La carga de la enfermedad en Colombia. Santa Fé: Ministerio de Salud de Bogotá, 1994.
Rodríguez J, Gallardo H. Carga Global de Enfermedad. Colombia, 1985–1995. República de Colombia: Ministerio de Salud, Imprenta Nacional, 2000.
Rodríguez J, Peńaloza E, Acosta N. Carga de Enfermedad Colombia. Resultados alcanzados. Bogotá: Pontifi ca Universidad Javeriana. Centro de Proyectos para el Desarrollo, 2008.
Ministerio de Salud de Costa Rica. Medición de la carga de enfermedad en Costa Rica, 2005. En: Programa de Desarrollo del Sector Salud. Componente Fortalecimiento Institucional del Ministerio de Salud. Costa Rica: Ministerio de Salud, 2008.
Seuc AH, Domínguez E. Evolution of disease mortality burden in Cuba: 1990-2005. Cad Saude Publica 2010; 26: 615–23.
Lozada P, Aguinaga L, Páez R, Olmedo C, Pozo A. El Peso de la Enfermedad en el Ecuador. Quito: Partnerships for Health Reform, 1999.
Fouad D, El-Essawy OM. Egypt disability-adjusted life years and life expectation models and human development. EcoMod2007 Conference, 2007. http://www.ecomod.net/sites/default/fi les/ document-conference/ecomod2007/18.doc (accessed July 27, 2012).
Lai T, Habicht J, Kiivet R-A. Measuring burden of disease in Estonia to support public health policy. Eur J Public Health 2009; 19: 541–47.
Highlights on health in Estonia 2005. Estonia, WHO Country Offi ce: World Health Organization, 2006.
Lapostolle A, Lefranc A, Gremy I, Spira A. Sensitivity analysis in summary measure of population health in France. Eur J Public Health 2008; 18: 195–200.
Hyder AA, Rotllant G, Morrow RH. Measuring the burden of disease: healthy life-years. Am J Public Health 1998; 88: 196–202.
NCMH Background Papers—Burden of Disease in India. New Delhi: Ministry of Health & Family Welfare, 2005.
Kosen S, Sidharta Y. Estimation of national burden of disease in Indonesia: a challenge of epidemiological transition. Jakarta: Health Services Research and Development Center, National Institute of Health Research and Development, Ministry of Health, 1998.
Mariotti S, D’Errigo P, Mastroeni S, Freeman K. Years of life lost due to premature mortality in Italy. Eur J Epidemiol 2003; 18: 513–21.
Ikeda S, Tabata K. Estimation of disability-adjusted life years (DALYs) in Japan using a simplifi ed method. J Health Care Soc 1998; 8: 83–99.
Fukuda Y, Hasegawa T, Yatsuya H, Tabata K. Disease burden and disability-adjusted life years (DALYs) in Japan. J Health Welfare Stat 1999; 46: 28–33.
Yoon S-J, Bae S-C, Lee S-I, et al. Measuring the burden of disease in Korea. J Korean Med Sci 2007; 22: 518–23.
Oh IH, Yoon SJ, Kim EJ. The burden of disease in Korea. J Korean Med Assoc 2011; 54: 646–52.
Bowie C. The burden of disease in Malawi. Malawi Med J 2007; 18: 104–10.
Yusoff AF, Mustafa AN, Kaur GK, et al. Malaysian Burden of Disease and Injury Study. http://espace.uq.edu.au/view/UQ:172329 (accessed June 26, 2012).
Stevens G, Dias RH, Thomas KJA, et al. Characterizing the epidemiological transition in Mexico: national and subnational burden of diseases, injuries, and risk factors. PLoS Med 2008; 5: e125.
Lozano R, Gómez-Dantés H, Franco M, Rodríguez A. Carga de la enfermedad en municipios urbanos marginados, México 2004–2008, BID, Observatorio de la Salud para América Latina y el Caribe. México: Funsalud, 2009.
Lozano R. El peso de la enfermedad en México: avances y desafíos. In: Observatorio de la Salud. Necesidades, servicios y políticas. México: Funsalud, 1997: 23–61.
Lozano R, Murray CJL, Frenk J, Bobadilla J-L. Burden of disease assessment and health system reform: results of a study in Mexico. J Int Dev 1995; 7: 555–63.
Dgedge M, Novoa A, Macassa G, et al. The burden of disease in Maputo City, Mozambique: registered and autopsied deaths in 1994. Bull World Health Organ 2001; 79: 546–52.
Melse JM, Essink-Bot ML, Kramers PG, Hoeymans N, and the Dutch Burden of Disease Group. A national burden of disease calculation: Dutch disability-adjusted life-years. Am J Public Health 2000; 90: 1241–47.
Ministry of Health. The burden of disease and injury in New Zealand—Public Health Intelligence Occasional Bulletin No. 1. New Zealand: Ministry of Health, 2001.
Hyder AA, Morrow RH. Applying burden of disease methods in developing countries: a case study from Pakistan. Am J Public Health 2000; 90: 1235–40.
Velásquez Valdivia A. Análisis del Estudio de Carga de Enfermedad en el Perú-MINSA 2004 y Propuesta Metodológica para el Ajuste con Datos Nacionales de Morbilidad. Lima: Promoviendo alianzas y estrategias, Abt Associates Inc, 2006.
de Salud M. Estudio de Carga de Enfermedad en el Perú, 2004. Lima, Perú: DGE, 2006.
Jankovi? S, Vlajinac H, Bjegovi? V, et al. The burden of disease and injury in Serbia. Eur J Public Health 2007; 17: 80–85.
Phua HP, Chua AV, Ma S, Heng D, Chew SK. Singapore’s burden of disease and injury 2004. Singapore Med J 2009; 50: 468–78.
Bradshaw D, Groenewald P, Laubscher R, et al. Initial burden of disease estimates for South Africa, 2000. S Afr Med J 2003; 93: 682–88.
Norman R, Bradshaw D, Schneider M, Pieterse D, Groenewald P. Revised burden of disease estimates for the comparative risk factor assessment, South Africa 2000. South African Medical Research Council, 2006.
Gčnova-Maleras R, Alvarez-Martín E, Morant-Ginestar C, Fernández de Larrea-Baz N, Catalá-López F. Measuring the burden of disease and injury in Spain using disability-adjusted life years: an updated and policy-oriented overview. Public Health 2012; published online Oct 9. DOI:S0033-3506(12)00318-6.10.1016/j.puhe.2012.08.012.
Catalá López F, Alvarez Martín E, Gčnova Maleras R, Morant Ginestar C. Relationship between research funding in the Spanish National Health System and the burden of disease. Rev Esp Salud Publica 2009; 83: 137–51.
Peterson S, Backlund I, Diderichsen F. Sjukdomsbördan i Sverige: en svensk DALY-kalkyl [Burden of disease in Sweden—a Swedish DALY calculation]. Stockholm: National Public Health Institute, 1998.
Bundhamcharoen K, Odton P, Phulkerd S, Tangcharoensathien V. Burden of disease in Thailand: changes in health gap between 1999 and 2004. BMC Public Health 2011; 11: 53.
Bundhamcharoen K, Teerawattananon Y, Vos T, Begg S. Thai Working Group on Burden of Disease and Injuries: burden of disease and injuries in Thailand: priority setting for policy. Thainland: Ministry of Public Health, 2002.
The Thai Working Group on Burden of Disease and Injury. Burden of disease for priority of health problems in Thailand 1999. J Health Sci 2004; 13: 239–56.
Samutharuk B, Vecharuk P, Kongkamnerd R, Sitthiamorn C. Measurement of disease burden on Thai society: guideline of health problem prioritisation. Nonthaburi: Health System Research Institute, 1999.
Ministry of Health. Baskent University. Burden of disease fi nal report. Ankara: Refi k Saydam Hygiene Center Presidency Refi k Saydam School of Public Health Directorate, 2004.
Green S, Miles R. The burden of disease and illness in the UK: a preliminary assessment to inform the development of UK health research and development priorities. Oxford: Healthcare Associates, 2007.
Michaud CM, McKenna MT, Begg S, et al. The burden of disease and injury in the United States 1996. Popul Health Metr 2006; 4: 11.
McKenna MT, Michaud CM, Murray CJL, Marks JS. Assessing the burden of disease in the United States using disability-adjusted life years. Am J Prev Med 2005; 28: 415–23.
Viet Nam Burden of Disease and Injury Study 2008. Viet Nam burden of disease and injury study 2008. Hanoi: Medical Publishing House, 2011.
Chapman G, Hansen KS, Jelsma J, et al. The burden of disease in Zimbabwe in 1997 as measured by disability-adjusted life years lost. Trop Med Int Health 2006; 11: 660–71.
Schopper D, Pereira J, Torres A, et al. Estimating the burden of disease in one Swiss canton: what do disability adjusted life years (DALY) tell us? Int J Epidemiol 2000; 29: 871–77.
Kominski GF, Simon PA, Ho A, Luck J, Lim Y-W, Fielding JE. Assessing the burden of disease and injury in Los Angeles County using disability-adjusted life years. Public Health Rep 2002; 117: 185–91.
Zhou S-C, Cai L, Wan C-H, Lv Y-L, Fang P-Q. Assessing the disease burden of Yi people by years of life lost in Shilin county of Yunnan province, China. BMC Public Health 2009; 9: 188.
Friedman C, McKenna MT, Ahmed F, et al. Assessing the burden of disease among an employed population: implications for employer-sponsored prevention programs. J Occup Environ Med 2004; 46: 3–9.
Hsairi M, Fekih H, Fakhfakh R, Kassis M, Achour N, Dammak J. Années de vie perdues et transition épidémiologique dans le gouvernorat de Sfax (Tunisie). Sante Publique 2003; 15: 25–37.
Dodhia H, Phillips K. Measuring burden of disease in two inner London boroughs using Disability Adjusted Life Years. J Public Health (Oxf) 2008; 30: 313–21.
Prince M, Patel V, Saxena S, et al. No health without mental health. Lancet 2007; 370: 859–77.
Polinder S, Haagsma JA, Lyons RA, et al. Measuring the population burden of fatal and nonfatal injury. Epidemiol Rev 2012; 34: 17–31.
Mathers CD, Ezzati M, Lopez AD. Measuring the burden of neglected tropical diseases: the global burden of disease framework. PLoS Negl Trop Dis 2007; 1: e114.
Mathur P, Shah B. Research priorities for prevention and control of noncommunicable diseases in India. Indian J Community Med 2011; 36 (suppl 1): S72–77.
Murray CJL, Ezzati M, Flaxman AD, et al. The Global Burden of Disease Study 2010: design, defi nitions, and metrics. Lancet 2012; 380: 2063–66.
Lopez AD, Mathers CD, Ezzati M, Jamison DT, Murray CJ, eds. Global burden of disease and risk factors. Washington, DC: Oxford University Press and The World Bank, 2006.
Naghavi M, Makela S, Foreman K, O’Brien J, Pourmalek F, Lozano R. Algorithms for enhancing public health utility of national causes-of-death data. Popul Health Metr 2010; 8: 9.
Ahern RM, Lozano R, Naghavi M, Foreman K, Gakidou E, Murray CJ. Improving the public health utility of global cardiovascular mortality data: the rise of ischemic heart disease. Popul Health Metr 2011; 9: 8.
Wang H, Dwyer-Lindgren L, Lofgren KT, et al. Age-specifi c and sex-specific mortality in 187 countries, 1970–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2071–94.
King G, Tomz M, Wittenberg J. Making the most of statistical analyses: improving interpretation and presentation. Am J Pol Sci 2000; 44: 347–61.
Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2095–128.
Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLD) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012; 380: 2163–96.
Murray CJL, Rosenfeld LC, Lim SS, et al. Global malaria mortality between 1980 and 2010: a systematic analysis. Lancet 2012; 379: 413–31.
Frenk J, Chen L, Bhutta ZA, et al. Health professionals for a new century: transforming education to strengthen health systems in an interdependent world. Lancet 2010; 376: 1923–58.
Yelin E, Callahan LF, Group TNADW, and the National Arthritis Data Work Groups. The economic cost and social and psychological impact of musculoskeletal conditions. Arthritis Rheum 1995; 38: 1351–62.
Coyte PC, Asche CV, Croxford R, Chan B. The economic cost of musculoskeletal disorders in Canada. Arthritis Care Res 1998; 11: 315–25.
Peden M, Scurfi eld R, Sleet D, et al. World report on road traffi c injury prevention. Geneva: World Health Organization, 2004.
Hyder AA, Allen KA, Di Pietro G, et al. Addressing the implementation gap in global road safety: exploring features of an eff ective response and introducing a 10-country program. Am J Public Health 2012; 102: 1061–67.
World Health Organization, and the United Nations Road Safety Collaboration. http://www.who.int/roadsafety/en/ (accessed June 28, 2012).
United Nations. The United Nations and Road Safety. http://www. un.org/en/roadsafety/background.shtml (accessed June 28).
Reiss A, Roth J, eds. Violence in families: understanding and preventing violence. Panel on the understanding and control of violent behavior. Washington, DC: National Academy Press, 1993.
Carp FM. Elder abuse in the family: an interdisciplinary model for research. New York: Springer, 2000.
Schiamberg LB, Gans D. An ecological framework for contextual risk factors in elder abuse by adult children. J Elder Abuse Negl 1999; 11: 79–103.
Heise LL. Violence against women: an integrated, ecological framework. Violence Against Women 1998; 4: 262–90.
Chalk RA, King P. Violence in families: assessing prevention and treatment programs. Washington, DC: National Academies Press, 1998.
World Health Organization. Violence prevention: the evidence, 2010 http://www.who.int/violence_injury_prevention/violence/ 4th_milestones_meeting/publications/en/ (accessed July 21, 2012).
Kurmi OP, Semple S, Simkhada P, Smith WCS, Ayres JG. COPD and chronic bronchitis risk of indoor air pollution from solid fuel: a systematic review and meta-analysis. Thorax 2010; 65: 221–28.
Hu G, Zhou Y, Tian J, et al. Risk of COPD from exposure to biomass smoke: a metaanalysis. Chest 2010; 138: 20–31.
Marks G, Burney P. Diseases of the respiratory system. In: The health of adult britain 1841–1994. London: HM Stationery Offi ce, 1997.
Hurley SF, Matthews JP. The Quit Benefi ts Model: a Markov model for assessing the health benefi ts and health care cost savings of quitting smoking. Cost Eff Resour Alloc 2007; 5: 2.
Logan WPD. Mortality in England and Wales from 1848 to 1947. Popul Stud 1950; 4: 132–78.
Adair T, Hoy D, Dettrick Z, Lopez AD. 100 years of Chronic Obstructive Pulmonary Disease (COPD) mortality in Australia: the role of tobacco consumption. Int J Tuberc Lung Dis 2012; 16: 1699–705.

Return to GLOBAL BURDEN OF DISEASE
Since 11-19-2018


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |