

Cervicogenic Headache: An Assessment of the Evidence
on Clinical Diagnosis, Invasive Tests, and TreatmentThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Lancet Neurol. 2009 (Oct); 8 (10): 959–968 ~ FULL TEXT
Nikolai Bogduk, MD, Jayantilal Govind, MBChB
Newcastle Bone and Joint Institute,
Royal Newcastle Centre,
New South Wales, Australia
nbogduk@bigpond.net.auCervicogenic headache is characterised by pain referred to the head from the cervical spine. Although the International Headache Society recognises this type of headache as a distinct disorder, some clinicians remain sceptical. Laboratory and clinical studies have shown that pain from upper cervical joints and muscles can be referred to the head. Clinical diagnostic criteria have not proved valid, but a cervical source of pain can be established by use of fluoroscopically guided, controlled, diagnostic nerve blocks. In this Review, we outline the basic science and clinical evidence for cervicogenic headache and indicate how opposing approaches to its definition and diagnosis affect the evidence for its clinical management. We provide recommendations that enable a pragmatic approach to the diagnosis and management of probable cervicogenic headache, as well as a rigorous approach to the diagnosis and management of definite cervical headache.
From the FULL TEXT Article:
Introduction
Cervicogenic headache is pain referred to the head from a source in the cervical spine. Unlike other types of headache, cervicogenic headache has attracted interest from disciplines other than neurology, in particular manual therapists and interventional pain specialists, who believe that they can find the source of pain among the joints of the cervical spine. Neurologists differ in their acceptance of this disorder. The International Headache Society recognises cervicogenic headache as a distinct disorder [1] and one chapter in a leading headache textbook acknowledges that injuries to upper cervical joints can cause headache after whiplash, [2] although another chapter indicates that this concept is not fully accepted. [3]
In terms of basic sciences, cervicogenic headache is the best understood of the common headaches. The mechanisms are known, and this headache has been induced experimentally in healthy volunteers. In some patients, cervicogenic headache can be relieved temporarily by diagnostic blocks of cervical joints or nerves. However, a matter that remains contentious is how cervicogenic headache should be diagnosed. Some neurologists maintain that this headache can be diagnosed on clinical features; others are not convinced of the validity of such diagnosis. Manual therapists use manual examination of vertebral motion segments, whereas interventional pain specialists use fluoroscopically guided diagnostic blocks.
In this Review, we provide a synopsis of the available evidence on cervicogenic headache. We summarise the basic mechanisms, analyse the evidence on diagnosis and treatment, and provide recommendations on management.
Mechanism of pain referral
Cervicogenic headache is referred pain from the cervical spine. Physiologically, this pain is analogous to pain felt in the shoulders, chest wall, buttocks, or lower limbs that is referred from spinal sources; hence its familiarity to pain specialists.

Figure 1
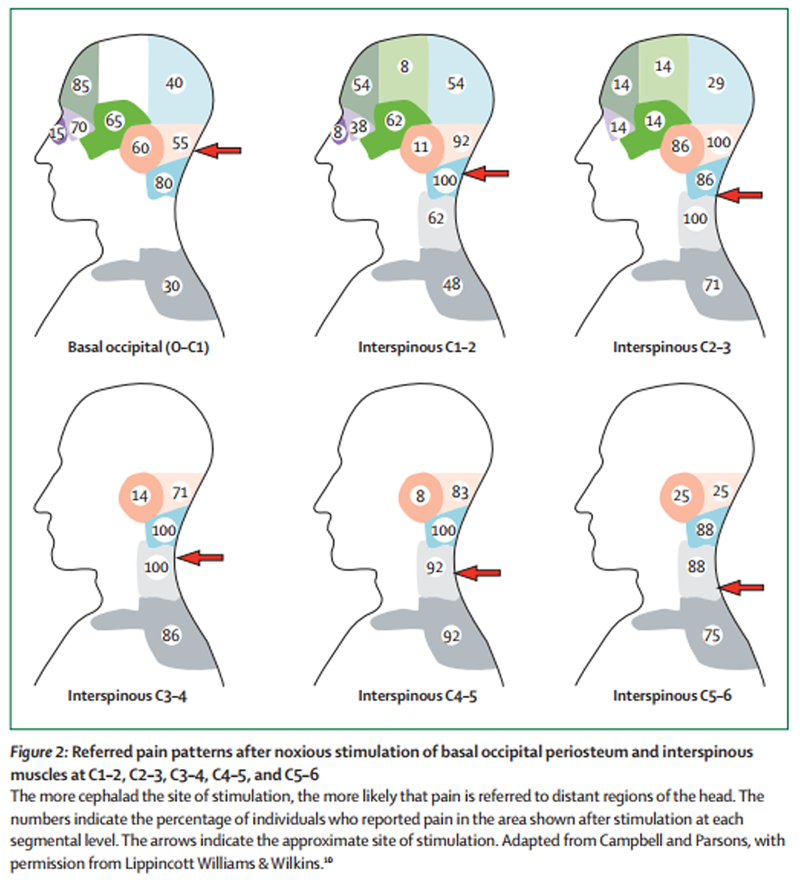
Figure 2
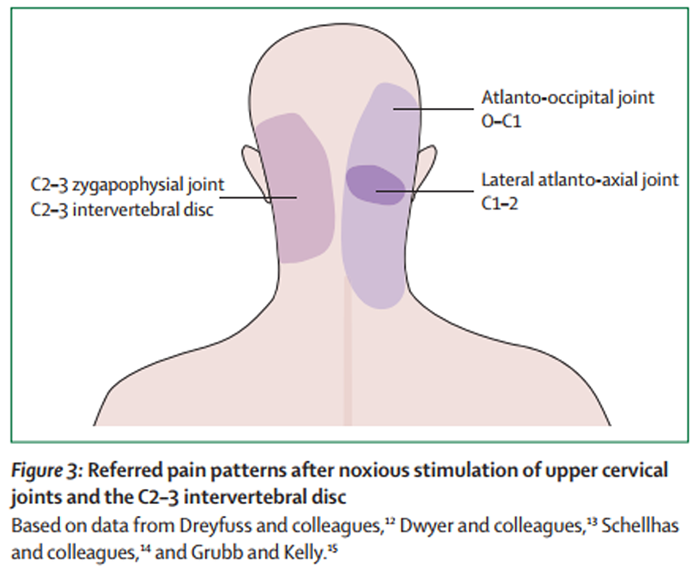
Figure 3
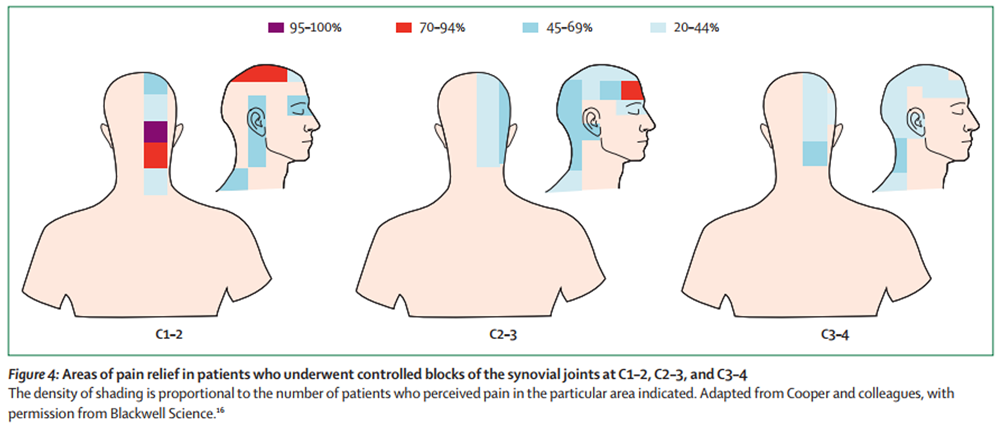
Figure 4 The mechanism underlying the pain involves convergence between cervical and trigeminal afferents in the trigeminocervical nucleus (Figure 1). [4, 5] In this nucleus, nociceptive afferents from the C1, C2, and C3 spinal nerves converge onto second-order neurons that also receive afferents from adjacent cervical nerves and from the first division of the trigeminal nerve (V), via the trigeminal nerve spinal tract. This convergence has been shown anatomically and physiologically in laboratory animals. [5–9] Convergence between cervical afferents allows for upper cervical pain to be referred to regions of the head innervated by cervical nerves (occipital and auricular regions). Convergence with trigeminal afferents allows for referral into the parietal, frontal, and orbital regions.
Such patterns of referral have been elicited in healthy volunteers by experimental, noxious stimulation of cervical structures. Early studies targeted the suboccipital and posterior cervical muscles, [10, 11] and investigators have shown that noxious stimulation of more rostral structures in the cervical spine elicited referred pain in the occipital region and more distant regions, such as the frontal region and orbit. By contrast, stimulation of more caudal structures elicited pain in the neck, which could be referred to the occipital regions, although not to distant regions of the head (Figure 2). Results from later studies have shown that noxious stimulation of the atlanto-occipital and lateral atlanto-axial joints, [12] the C2–3 zygapophysial joint, [13] and the C2–3 intervertebral disc [14, 15] can produce pain in the occipital region (Figure 3).
Complementary studies have mapped the distribution of pain that could be relieved in patients by controlled diagnostic blocks of the lateral atlanto-axial joint or the C2–3 or C3–4 zygapophysial joints. [16] Patients with pain from a particular joint do not have exactly the same distribution of pain, but there are similarities in the distribution. Pain from the lateral atlanto-axial joint (C1–2) tends to be focused on the occipital and suboccipital regions, and tends to be referred to the vertex, orbit, and ear (Figure 4). Pain from the C2–3 zygapophysial joint also occurs in the occipital region and spreads across the parietal region to the frontal region and orbit. Pain from the C3–4 joint can be referred to the head, but is more commonly focused in the upper and lateral cervical region (Figure 4).
These data show that the structures capable of producing referred pain to the head are those innervated by the C1, C2, and C3 nerves. No experimental studies have shown that structures innervated by lower cervical nerves are capable of directly causing headache.
Epidemiology
Estimates of the prevalence of cervicogenic headache differ according to the populations studied and the criteria used to make the diagnosis. When clinical criteria have been used, the prevalence of cervicogenic headache has been estimated to be 1%, 2·5%, [17] or 4·1% [18] in the general population and as high as 17·5% among patients with severe headaches. [17] The prevalence is as high as 53% in patients with headache after whiplash. [19]
Diagnosis
The diagnosis of cervicogenic headache has been driven by two schools of practice. The clinical diagnosis approach arose in Europe and was based on the belief that cervicogenic headache had distinctive clinical features by which it could be diagnosed. The approach of interventional diagnosis by pain medicine arose in Australia and North America and was based on establishing a cervical source of pain in patients with headache by use of controlled diagnostic blocks.
Clinical diagnosis
The first set of clinical diagnostic criteria, published in 1990 [20] and revised in 1998, [21] defined cervicogenic headache as a unilateral headache associated with evidence of cervical involvement through provocation of pain by movement of the neck or by pressing the neck; concurrent pain in the neck, shoulder, and arm; and reduced range of motion of the neck, with or without other features. Results from subsequent studies have shown that these clinical features were either not unique to cervicogenic headache [22–24] or were insufficiently different from features in healthy individuals, [25, 26] thereby precluding them from being valid diagnostic features. [5, 27, 28] Similarly, either there are no radiographic abnormalities in patients said to have cervicogenic headache [29, 30] or the radiographic features overlap with those seen in healthy individuals. [5, 31]
When tested for agreement between observers, the proposed clinical features of cervicogenic headache differed in their reliability. [5, 28] The most reliable features were pain that starts in the neck and radiates to the fronto-temporal region; pain that radiates to the ipsilateral shoulder and arm; and provocation of pain by neck movement. [32, 33] Agreement was poor about other features, such as restricted range of motion and pressure pain on palpation. [5, 28]
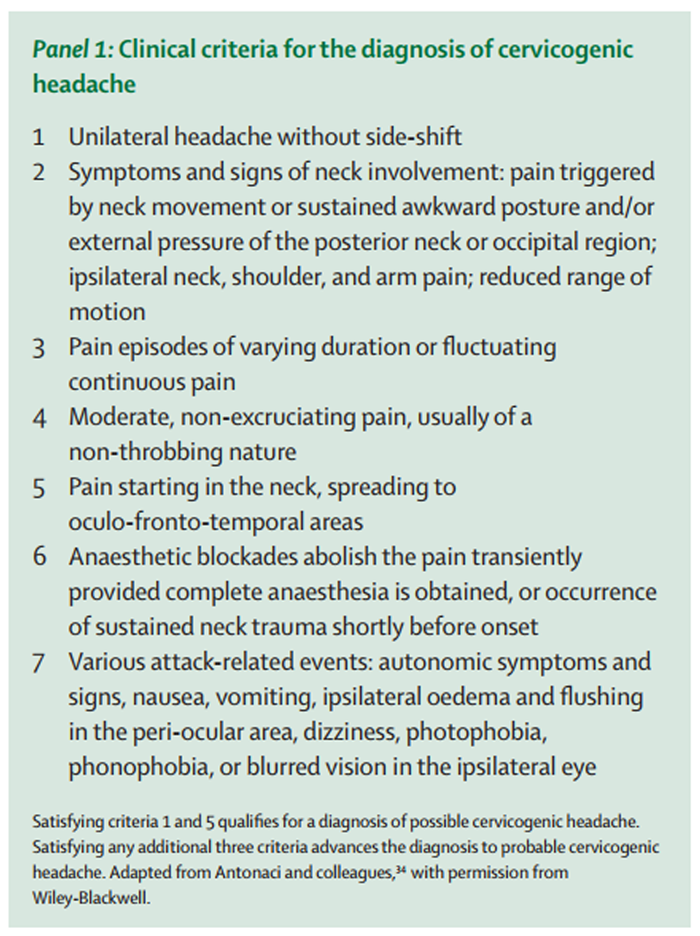
Panel 1 Some investigators have proposed a less emphatic clinical approach to diagnosis. These clinicans reduced the clinical criteria to a list of seven features (Panel 1), [34] and qualified the certainty of diagnosis. These authors proposed that possible cervicogenic headache could be diagnosed if patients had unilateral headache and pain that starts in the neck. If any three additional criteria were fulfilled, the diagnosis was advanced to probable cervicogenic headache. By use of these operational guidelines, the authors felt that they could confidently distinguish cervicogenic headache from migraine. The clinical features most strongly indicative of cervicogenic headache were pain that radiates to the shoulder and arm, varying duration or fluctuating continuous pain, moderate, non-throbbing pain, and history of neck trauma.
Although some investigators have defended the clinical diagnostic criteria for cervicogenic headache, they have examined only their nosological validity (ie, the extent to which the criteria distinguish cervicogenic headache from migraine and tension-type headache). No studies have established that patients who fulfill these diagnostic criteria actually have a cervical source for their pain. Fundamental to the concept of cervicogenic headache is that it constitutes pain referred to the head from a cervical source. Therefore, proving such a source is essential for the diagnosis.

Panel 2 The revised criteria of the International Headache Society [1] reflect the controversy between clinical diagnosis and objective testing for cervicogenic headache (Panel 2). Evidence of a cervical source of pain is required, but the explanatory notes declare that clinical features that have little reliability or validity are not acceptable. With out other evidence, controlled diagnostic blocks become the only means of establishing the diagnosis.
An idiosyncrasy of the International Headache Society criteria is criterion D, which describes resolution of pain 3 months after treatment. This criterion was not designed for diagnosis before treatment, but to promote rigour. The criterion requires that, if a cause is found, relief of pain should ensue if that cause is successfully treated. A corollary of this criterion is that partial or short-lasting relief after treatment does not qualify as a diagnostic criterion.
Interventional diagnosis
Practitioners of interventional pain medicine use fluoroscopically guided, controlled diagnostic blocks to test whether particular structures are the source of pain in patients with suspected cervicogenic headache. Studies have focused on three structures:the lateral atlanto-axial joint can be anaesthetised by use of intra-articular blocks (Figure 5); [35–37]
the C2–3 zygapophysial joint can be blocked by anaesthetising the third occipital nerve where it crosses the joint and supplies it with articular branches; [19, 38] and
the C3–4 zygapophysial joint can be anaesthetised by blocking the medial branches of the C3 and C4 dorsal rami. [38]Complete relief of headache after such blocks, under controlled conditions, provides objective evidence of a cervical source of pain.
The best available studies indicate that the C2–3 zygapophysial joints are the most common source of cervicogenic headache, [16, 19, 39, 40] accounting for about 70% of cases. [13] Although data are not available for the prevalence of lateral atlanto-axial joint as a source of cervicogenic headache, this joint seems to be quite commonly involved. [13, 35] The C3–4 zygapophysial joint has only occasionally been implicated in cervicogenic headache. [16] The C2–3 intervertebral disc can also be a source of this headache, but its prevalence as a causative factor is not known. [41]
In patients whose headaches have been relieved by controlled diagnostic blocks of upper cervical joints, there are no distinctive clinical features. The pain is typically dull and aching in quality. The range of movement of the head might be restricted, but not in any characteristic manner or to any characteristic degree. Tenderness over the C2–3 joint is not a diagnostic feature as this sign has a positive likelihood ratio of only 2·1 to 1. [19]
All studies that have implicated the C2–3 zygapophysial joint as a source of cervicogenic headache have been conducted in patients with a history of trauma. [16, 19, 39, 40] No patients with spontaneous onset of headache have had a cervical source of pain. This observation reinforces one feature in the clinical approach to diagnosis, in which history of neck trauma is an important criterion. A complement to this observation is that no studies that have used controlled diagnostic blocks have shown complete relief of pain in patients with migrainous features, such as photophobia and vomiting. In our experience, such patients do not respond to diagnostic blocks of cervical joints.
Manual diagnosis
Manual therapists contend that they can diagnose cervical sources of headache by manual examination of upper cervical joints. However, this practice has not been validated. An early study compared the diagnosis made by a manual therapist with that made with diagnostic blocks, [42] but the sample size was small and the blocks were not controlled. In a later study, which used a large sample and had controlled diagnostic blocks, manual examination had a high sensitivity but no specificity and, therefore, had no diagnostic validity. [43] Further evidence is required before a scientific basis for manual diagnosis can be confirmed.
Diagnosis through greater occipital nerve blocks
Some investigators use greater occipital nerve blocks as a diagnostic test either for greater occipital neuralgia or for cervicogenic headache. [44] However, there is no clear rationale for this practice. Diagnostic blocks relieve pain from a source innervated by a nerve distal to where it is blocked. Greater occipital nerve blocks are executed near to where the nerve crosses the superior nuchal line. Distal to this point, the nerve supplies only the skin of the occipital region. There are no known disorders of the scalp that might cause persistent pain. Therefore, greater occipital nerve blocks cannot be used to diagnose cervicogenic headache as these blocks do not establish a cervical source of pain. Furthermore, no studies have shown that controlled blocks of the greater occipital nerve consistently produce complete relief of headache. At best, greater occipital nerve blocks have a type of partial, neuromodulatory eff ect on headache mechanisms, whether the headaches have a cervical source or not. Greater occipital nerve blocks relieve pain, temporarily, in substantial proportions of patients with migraine, cluster headache, and hemicrania continua. [45, 46] A positive, greater occipital nerve block, therefore, cannot be a specifi c test for cervicogenic headache. [47]
Differential diagnosis
Several disorders share certain features of cervicogenic headache, such as pain in the neck and head. These disorders can be diffi cult to distinguish, unless (or until) additional features emerge. Other disorders form the differential diagnosis of cervicogenic headache only notionally because they aff ect cervical structures and might cause headache, but these disorders have distinctive features.
The most crucial differential diagnosis of cervicogenic headache is dissecting aneurysms of the vertebral or internal carotid arteries, which can present with neck pain and headache. [48–50] These aneurysms are indicated by the onset of cerebrovascular features, which typically emerge within 1–3 weeks. If this differential diagnosis is not considered, there is a risk of patients being treated with cervical manipulation, with fatal consequences due to aggravation of the aneurysm.
The second most important differential diagnosis is lesions of the posterior cranial fossa, as the dura mater and vessels of the posterior fossa are innervated by upper cervical nerves. These lesions are distinguished by the onset of neurological features or systemic illness. Meningitis of the upper cervical spine can be distinguished from cervicogenic headache by the presence of systemic illness and neck rigidity. Additionally, herpes zoster can produce pain in the occipital region during its prodromal phase; however, the eruption of vesicles distinguishes this disease from cervicogenic headache.
Because the C2 spinal nerve runs behind the lateral atlanto-axial joint and is accompanied by its dural sleeve and a substantial plexus of veins, two distinctive disorders can be confused with cervicogenic headache. [51] First, neck–tongue syndrome occurs when rapid turning of the head subluxates the lateral atlanto-axial joint posteriorly. Tension in the joint capsule causes ipsilateral occipital pain, while compression of the C2 spinal nerve produces numbness of the tongue. [52, 53] This syndrome is distinguished by its precipitating factor and accompanying features. Second, C2 neuralgia can be caused by various disorders. Inflammatory disorders or injuries of the lateral atlanto-axial joint can result in the adjacent nerve becoming incorporated in the fi brotic changes of chronic infl ammation. [54, 55] The C2 spinal nerve can be compromised by a meningioma, [56] neurinoma, [57] anomalous vertebral arteries, [58] and several other vascular anomalies. [54, 58, 59] Nerves affected by vascular abnormalities have several features indicative of neuropathy, such as myelin breakdown, chronic haemorrhage, axon degeneration and regeneration, and increased endoneurial and pericapsular connective tissue. [58] Unlike the dull, aching pain of cervicogenic headache, the features of C2 neuralgia are intermittent, lancinating pain in the occipital region associated with lacrimation and ciliary injection. [54, 58, 59]
Greater occipital neuralgia is an outdated diagnosis, used before the concept of somatic referred pain was widely understood, when physicians believed that any pain in a particular region was due to some affliction of the nerve that ran through that region. Accordingly, pain in the occipital region was attributed to greater occipital neuralgia. However, no pathology of the greater occipital nerve has ever been proven to explain occipital pain. The proposition that the greater occipital nerve could be compressed between the posterior arch of the atlas and the lamina of the axis is incompatible with the anatomy and biomechanics of those vertebrae [60] and was retracted by one of the authors who originally proposed it. [61] Entrapment of the greater occipital nerve where it leaves the posterior neck muscles has not been distinguished from the normal anatomy of the nerve penetrating a fibrous sling. [5, 62] Liberation of the nerve has also not proved to be an eff ective, lasting treatment.5,63 Lancinating pain in the occipital region is more likely to be C2 neuralgia, in which case the pathology aff ects the C2 spinal nerve rather than the greater occipital nerve. Deep aching pain is incompatible with a neuralgia: this pain is more likely to be somatic referred pain from an upper cervical joint. [1, 5]
Another outdated diagnosis is cervical migraine (also known as Barré–Lieou syndrome). [64, 65] In this disorder, headache was purportedly due to irritation of the vertebral nerve, which causes spasm of the vertebral artery. In physiological studies, the vertebral artery is remarkably inert to electrical stimulation of the vertebral nerve [66] and even inert to intra-arterial infusions of vasoactive drugs. [67] These properties rule out the purported mechanism of migraine cervicale. [5]
Lower cervical disorders
Some investigators have studied the proposition that lower cervical disorders can cause headache. These researchers refer to circumstantial evidence that some patients with lower cervical radiculopathy also have headache, of whom various proportions are relieved of headache when the radiculopathy is treated surgically. [68, 69] Although these data imply an association, they do not indicate a direct association between headache and lower cervical disorders. Neuroanatomically, there is no direct link between lower cervical afferents and the trigeminocervical nucleus. Intermediate mechanisms, such as muscle tension and secondary kinematic abnormalities that aff ect upper cervical joints [70] might be involved.
Treatment
Although there have been many treatments suggested for cervicogenic headache, few have been tested and even fewer have been proven successful. Among the determinants of effectiveness are whether the headache was diagnosed clinically or whether a cervical source was proven.
Clinical diagnosis
No drugs are eff ective for cervicogenic headache. Transcutaneous electrical nerve stimulation has been investigated, but not in a controlled study. About 80% of patients obtained at least a 60% decrease in their headache index with this technique, but only at 1 month after treatment. [71]
For manual therapy, most publications are case reports or case series. [72] The few randomised, controlled studies provided follow-up of only 1 or 3 weeks, [73–75] and gave confl icting results. [72] The largest and most recent study showed that treatment with manual therapy, specific exercises, or manual therapy plus exercises was significantly more eff ective at reducing headache frequency and intensity than was no specific care by a general practitioner. [76] Manual therapy alone, however, was not more eff ective than exercises alone, and combining the two interventions did not achieve better outcomes than either intervention alone. About 76% of patients achieved a more than 50% decrease in headache frequency and 35% achieved complete relief at the 7–week follow-up. At 12 months, 72% had a more than 50% decrease in headache frequency, but the proportion that had complete relief was not reported. Corresponding fi gures for decrease in pain intensity were not reported.
In patients with a clinical diagnosis of cervicogenic headache, some investigators have targeted the greater occipital nerve for treatment. In one series, 169 of 180 patients (94%) obtained relief after an injection of 160 mg of depot methylprednisolone and 3–4 mL of 1% lidocaine, but only for a mean duration of 23·5 days (range 10–77 days). [77] In an unrelated study of 50 patients, surgical “liberation” of the nerve initially relieved headache in about 80% of cases, but for a median duration of only about 3–6 months. [63] Excision of the greater occipital nerve provided relief in about 70% of patients, but for a median duration of only 244 days. [78]
Specific diagnosis
In one study, patients were selected for surgery if they fulfilled the clinical criteria for cervicogenic headache and obtained relief of headache from diagnostic blockade of the C2 spinal nerve. [79] Patients underwent decompression and microsurgical neurolysis of the C2 spinal nerve, with excision of scar, and ligamentous and vascular elements that compressed the nerve. 14 of 31 patients were rendered pain-free at a mean follow-up of 16 months. Details on the remaining patients are incomplete, but 51% gained what was called “adequate” relief, and 11% suffered a recurrence.
For patients with pain stemming from the lateral atlanto-axial joints, two options are available. In an observational study, 26 of 32 patients obtained immediate relief after intra-articular injection of steroids. [80] About one in five patients obtained greater than 50% relief from their headache for 3 months, and one in eight obtained complete relief lasting 9 months. However, such outcomes have not been confirmed in a controlled trial and, therefore, cannot yet be attributed to the injection of steroids. It is possible that a placebo effect might have influenced results. The other option is arthrodesis of the joint. The surgical reports attest to success with this procedure, albeit in small numbers of patients, with complete relief of pain lasting over 2 years. [81–83]
For patients with pain stemming from the C2–3 zygapophysial joint, investigators from one study reported that some patients could obtain relief from intra-articular injection of steroids. [84] At 19 months after such injections, two of 18 patients (11%) were free of pain. A further 50% had a reduced frequency of headaches. This study, however, was not controlled and placebo effects were not excluded. Nevertheless, without other alternatives, intra-articular injection of steroids would seem to be a safe and expedient intervention that could benefit some patients.
Radiofrequency neurotomy
The most extensively studied treatment for cervicogenic headache is percutaneous radiofrequency neurotomy. The rationale for this procedure is that if headache can be relieved temporarily by controlled diagnostic blocks of the nerve (or nerves) that innervate a particular cervical joint, then interrupting the pain signal along that nerve, by coagulating it, should provide long-lasting relief. This neurosurgical procedure is particularly applicable for the treatment of headache stemming from the C2–3 zygapophysial joint, in which case the target nerve is the third occipital nerve, which innervates that joint.
Three studies have reported that radiofrequency neurotomy is not eff ective. [85–87] In these studies, patients were selected on the basis of clinical criteria, and neurotomy was done at all levels from C3 to C6. Diagnostic blocks were done in one study, [86] but the results were not used as an indication for treatment. In the first study, only one of 15 patients achieved complete relief of pain; [85] in the second study (n=12), outcomes were no different in patients who received active lesions from those who received sham lesions; [86] and in the third study (n=30), outcomes from neurotomy were no different from those of an injection of local anaesthetic into the greater occipital nerve. [87]
However, there are three potential diffi culties with these studies of radiofrequency neurotomy. First, at no stage was the source of pain established. Second, the technique used for neurotomy has never been validated. Third, neurotomy was done at segmental levels (C3–6) that have rarely, if ever, been implicated as a source of headache. Nerves not proven to mediate the patient’s pain were disrupted by use of techniques not proven to denervate them.
Opposite results were reported in two studies in which the diagnosis was carefully established with controlled diagnostic blocks and meticulous surgical techniques were used. [88, 89] For patients in whom effects of diagnostic blocks indicate that the C2–3 zygapophysial joint is the source of pain, that joint can be denervated percutaneously by radiofrequency neurotomy of the third occipital nerve. The procedure involves placing an electrode parallel and close to the nerve where it crosses the joint, and using the electrode to disrupt the nerve. [90] Under these conditions, complete relief of pain was achieved in 88% of patients, [88, 91] with a median duration of relief of 297 days. [88] For patients in whom headaches recur, relief can be reinstated by repeating the neurotomy. By undertaking repetition as required, some patients have been able to maintain relief of their headache for longer than 2 years. [88] The results of a randomised, placebo-controlled trial indicate that responses to radiofrequency neurotomy are not due to placebo effects (p=0·03). [91] The successful treatment of third occipital headache in this study cannot, therefore, be dismissed as a placebo effect.
A pragmatic clinical approach
The degree to which practitioners might manage cervicogenic headache depends on the facilities available to them. If clinicans can undertake fl uoroscopically guided diagnostic blocks, they can establish a cervical source of pain and thereby fulfill the diagnostic criteria for cervicogenic headache as set by the International Headache Society. [1] If physicians are restricted to clinical diagnosis only, they cannot fulfi l those criteria. Nevertheless, a working diagnosis of possible or probable cervicogenic headache can be established based on the criteria listed in Panel 1. [34]
For probable cervicogenic headache, exercises with or without manual therapy seems to be the best option among conservative therapies. [76] All other treatment strategies are entirely speculative.
If diagnostic blocks or discography can be applied, a source of pain might be established in the lateral atlanto-axial joint, the C2–3 intervertebral disc, or the C2–3 zygapophysial joint. For pain stemming from the lateral atlanto-axial joint, arthrodesis is the only treatment for which there is any evidence of effectiveness. [81–83] Intra-articular injections of steroids are more conservative but their efficacy has not yet been shown. For pain stemming from the C2–3 intervertebral disc, anterior cervical fusion can be effective. [41] No alternative is known for discogenic pain. For pain stemming from the C2–3 zygapophysial joint, intra-articular injection of steroids is a low-risk treatment from which some patients can benefit. [84] However, a placebo effect has not been excluded. The only definitive treatment for headache stemming from the C2–3 zygapophysial joint is radiofrequency neurotomy. [88, 89] However, this procedure has to be done with meticulous accuracy and is indicated only if patients obtain complete relief of headache after controlled blocks of the third occipital nerve. [88, 89]
Conclusions
Neurologists are accustomed to diagnosing headache on the basis of clinical features, supplemented in some cases by medical imaging or other tests. Cervicogenic headache does not lend itself to this approach. As a result, tensions and controversies have arisen within the field. On the one hand, some experts have insisted that cervicogenic headache can be defined by clinical criteria, but the evidence shows otherwise. Consequently, owing to the absence of valid clinical criteria, some neurologists have doubted the diagnosis or consider it to be too often carelessly applied. On the other hand, pain specialists have developed invasive techniques whereby the diagnosis can be established objectively. But fluoroscopically guided, controlled diagnostic blocks are not among the conventional armamentarium of neurologists, nor are they widely available.
One of two developments is required for cervicogenic headache to become more commonly accepted. Pain specialists, or interventional radiologists, could collaborate with neurologists to add further studies to the published works showing the effects of diagnostic blocks in patients with suspected cervicogenic headache and the effects of target-specific interventions. Such collaboration would be in line with the call to reduce iatrogenic discomfort resulting from incorrect diagnosis and inappropriate treatment of headaches in general. [92] Alternatively, physicians intent on clinical diagnosis need to develop criteria that are not only reliable but also valid for a cervical source of pain. Doing so would allow elimination of the footnotes in the criteria of the International Headache Society, which currently prohibit clinical diagnosis. [1]
References:
International Headache Society.
The International Classification of Headache Disorders, 2nd edn.
Cephalalgia 2004; 24 (suppl 1): 115–16.Kasch H, Ramadan NM.
Headache attributed to whiplash injury.
In: Olesen J, Goadsby PJ, Ramadan NM, Tfelt-Hansen P, Welch KMA, eds. The Headaches, 3rd edn.
Philadelphia: Lippincott Williams & Wilkins, 2006: 879–83.Gobel H, Edmeads JG.
Disorder of the skull and cervical spine.
In: Olesen J, Goadsby PJ, Ramadan NM, Tfelt-Hansen P, Welch KMA, eds.
The Headaches, 3rd edn.
Philadelphia: Lippincott Williams & Wilkins, 2006: 1003–11.Bogduk N.
The neck and headaches.
Neurol Clin N Am 2004; 22: 151–71.Bogduk N, Bartsch T.
Cervicogenic headache. In: Silberstein SD, Lipton RB, Dodick DW, eds.
Wolff ’s Headache, 8th edn.
New York: Oxford University Press, 2008: 551–70.Bogduk N.
Cervicogenic headache: anatomic basis and pathophysiologic mechanisms.
Curr Pain Headache Rep 2001; 5: 382–86.Bartsch T, Goadsby PJ.
Stimulation of the greater occipital nerve induces increased central excitability of dural afferent input.
Brain 2002; 125: 1496–1509.Bartsch T, Goadsby PJ.
Increased responses in trigeminocervical nociceptive neurons to cervical input after stimulation of the dura mater.
Brain 2003; 126: 1801–13.Goadsby PJ, Ratsch T.
On the functional neuroanatomy of neck pain.
Cephalalgia 2008; 28 (suppl 1): 1–7.Campbell DG, Parsons CM.
Referred head pain and its concomitants.
J Nerv Ment Dis 1944; 99: 544–51.Feinstein B, Langton JBK, Jameson RM, Schiller F.
Experiments on referred pain from deep somatic tissues.
J Bone Joint Surg 1954; 36A: 981–97.Dreyfuss P, Michaelsen M, Fletcher D.
Atlanto-occipital and lateral atlanto-axial joint pain patterns.
Spine 1994; 19: 1125–31.Dwyer A, Aprill C, Bogduk N.
Cervical zygapophysial joint pain patterns I: a study in normal volunteers.
Spine 1990; 15: 453–57.Schellhas KP, Smith MD, Gundry CR, Pollei SR.
Cervical discogenic pain: prospective correlation of magnetic resonance imaging and discography in asymptomatic subjects and pain sufferers.
Spine 1996; 21: 300–12.Grubb SA, Kelly CK.
Cervical discography: clinical implications from 12 years of experience.
Spine 2000; 25: 1382–89.Cooper G, Bailey B, Bogduk N.
Cervical zygapophysial joint pain maps.
Pain Medicine 2007; 8: 344–53.Evers S.
Comparison of cervicogenic headache with migraine.
Cephalalgia 2008; 28 (suppl 1): 16–17.Sjaastad O.
Cervicogenic headache: comparison with migraine without aura; Vĺgĺ study.
Cephalalgia 2008; 28 (suppl 1): 18–20.Lord S, Barnsley L, Wallis B, Bogduk N.
Third occipital headache: a prevalence study.
J Neurol Neurosurg Psychiatr 1994; 57: 1187–90.Sjaastad O, Fredriksen TA, Pfaffenrath V.
Cervicogenic headache: diagnostic criteria.
Headache 1990; 30: 725–26.Sjaastad O, Fredriksen TA, Pfaffenrath V.
Cervicogenic headache: diagnostic criteria.
Headache 1998; 38: 442–45.Leone M, D’Amico D, Frediani F, Torri W, Sjaastad O, Bussone G.
Clinical considerations on side-locked unilaterality in long-lasting primary headaches.
Headache 1993; 33: 381–84.D’Amico D, Leone M, Bussone G.
Side-locked unilaterality and pain localization in long-lasting headaches: migraine, tension-type headache, and cervicogenic headache.
Headache 1994; 34: 526–30.Leone M, D’Amico D, Moschiano F, Frainotti M, Filippini G, Bussone G.
Possible identification of cervicogenic headache amongst patients with migraine: an analysis of 374 headaches.
Headache 1995; 35: 461–64.Bovim G.
Cervicogenic headache, migraine, and tension-type headache. Pressure-pain threshold measurements.
Pain 1992; 51: 169–73.Jull G, Barrett C, Magee R, Ho P.
Further clinical clarification of the muscle dysfunction in cervical headache.
Cephalalgia 1999; 19: 179–85.Leone M, D’Amico D, Grazzi L, Attanasio A, Bussone G.
Cervicogenic headache: a critical review of the current diagnostic criteria.
Pain 1998; 78: 1–5.Bogduk N.
Distinguishing primary headache disorders from cervicogenic headache: clinical and therapeutic implications.
Headache Currents 2005; 2: 27–36.Fredriksen TA, Fougner R, Tengerund A, Sjaastad O.
Cervicogenic headache. Radiological investigations concerning head/neck.
Cephalalgia 1989; 9: 139–46.Pfaffenrath V, Dandekar R, Pollman W.
Cervicogenic headache—the clinical picture, radiological findings and hypotheses on its pathophysiology.
Headache 1987; 27: 495–99.Zwart JA.
Neck mobility in different headache disorders.
Headache 1997; 37: 6–11.van Suijlekom JA, de Vet HCW, van den Berg SGM, Weber WEJ.
Interobserver reliability of diagnostic criteria for cervicogenic headache.
Cephalalgia 1999; 19: 817–23.van Suijlekom HA, de Vet HCW, van den Berg SGM, Weber WEJ.
Interobserver reliability in physical examination of the cervical spine in patients with headache.
Headache 2000; 40: 581–86.Antonaci F, Ghirmai S, Bono S, Sandrini G, Nappi G.
Cervicogenic headache: evaluation of the original diagnostic criteria.
Cephalalgia 2001; 21: 573–83.McCormick CC.
Arthrography of the atlanto-axial (C1–C2) joints: technique and results.
J Intervent Radiol 1987; 2: 9–13.Busch E, Wilson PR.
Atlanto-occipital and atlanto-axial injections in the treatment of headache and neck pain.
Reg Anesth 1989; 14 (suppl 2): 45.Aprill C, Axinn MJ, Bogduk N.
Occipital headaches stemming from the lateral atlanto-axial (C1–2) joint.
Cephalalgia 2002; 22: 15–22.International Spine Intervention Society.
Cervical medial branch blocks.
In: Bogduk N, ed. Practice Guidelines for Spinal Diagnostic and Treatment Procedures.
San Francisco: International Spinal Intervention Society, 2004: 112–37.Bogduk N, Marsland A.
On the concept of third occipital headache.
J Neurol Neurosurg Psychiatr 1986; 49: 775–80.Bogduk N, Marsland A.
The cervical zygapophysial joints as a source of neck pain.
Spine 1988; 13: 610–17.Schoff erman J, Garges K, Goldthwaite N, Kosetler M, Libby E.
Upper cervical anterior diskectomy and fusion improves discogenic cervical headaches.
Spine 2002; 27: 2240–44.Jull G, Bogduk N, Marsland A.
The Accuracy of Manual Diagnosis for Cervical Zygapophysial Joint Pain Syndromes
Med J Aust 1988 (Mar 7); 148 (5): 233–236King W, Lau P, Lees R, Bogduk N.
The validity of manual examination in assessing patients with neck pain.
Spine J 2007; 7: 22–26.Bovim G, Berg R, Dale LG.
Cervicogenic headache: anaesthetic blockades of cervical nerves (C2–C5) and facet joint (C2/C3).
Pain 1992; 49: 315–20.Afridi SK, Shields KG, Bhola R, Goadsby PJ.
Greater occipital nerve injection in primary headache syndromes—prolonged effects from a single injection.
Pain 2006; 122: 126–29.Goadsby PJ, Lipton RB, Ferrari MD.
Migraine—current understanding and treatment.
N Engl J Med 2002; 346: 257–70.Leone M, Cecchini AP, Mea E, Tullo V, Bussone G.
Epidemiology of fi xed unilateral headaches.
Cephalalgia 2008; 28 (suppl 1): 8–11.de Sousa JE, Halfon MJ, Bonardo P, Reisin RC, Fernández Pardal MM.
Different pain patterns in patients with vertebral artery dissections.
Neurology 2005; 64: 925–26.Bin Saeed A, Shuaib A, Al Sulaiti G, Emery D.
Vertebral Artery Dissection: Warning Symptoms, Clinical Features and Prognosis in 26 Patients
Canadian Journal of Neurological Sciences 2000 (Nov); 27 (4): 292–296 .Campos CR, Calderaro M, Scaff M, Conforto AB.
Primary headaches and painful spontaneous cervical artery dissection.
J Headache Pain 2007; 8: 180–84.Bogduk N.
Local anaesthetic blocks of the second cervical ganglion: a technique with application in occipital headache.
Cephalalgia 1981; 1: 41–50.Lance JW, Anthony M.
Neck tongue syndrome on sudden turning of the head.
J Neurol Neurosurg Psychiatr 1980; 43: 97–101.Bogduk N.
An anatomical basis for neck tongue syndrome.
J Neurol Neurosurg Psychiatr 1981; 44: 202–08.Jansen J, Markakis E, Rama B, Hildebrandt J.
Hemicranial attacks or permanent hemicrania—a sequel of upper cervical root compression.
Cephalalgia 1989; 9: 123–30.Poletti CE, Sweet WH.
Entrapment of the C2 root and ganglion by the atlanto-epistrophic ligament: clinical syndrome and surgical anatomy.
Neurosurgery 1990; 27: 288–91.Kuritzky, A.
Cluster headache-like pain caused by an upper cervical meningioma.
Cephalalgia 1984; 4: 185–86.Sharma RR, Parekh HC, Prabhu S, Gurusinghe NT, Bertolis G.
Compression of the C-2 root by a rare anomalous ectatic vertebral artery.
J Neurosurg 1993; 78: 669–72.Jansen J, Bardosi A, Hildebrandt J, Lucke A.
Cervicogenic, hemicranial attacks associated with vascular irritation or compression of the cervical nerve root C2. Clinical manifestations and morphological findings.
Pain 1989; 39: 203–12.Hildebrandt J, Jansen J.
Vascular compression of the C2 and C3 roots—yet another cause of chronic intermittent hemicrania?
Cephalalgia 1984; 4: 167–70.Hunter CR, Mayfield FH.
Role of the upper cervical roots in the production of pain in the head.
Am J Surg 1949; 78: 743–49.Mayfi eld FH.
Symposium on cervical trauma. Neurosurgical aspects.
Clin Neurosurg 1955; 2: 83–90.Bogduk N.
The anatomy of occipital neuralgia.
Clin Exp Neurol 1980; 17: 167–84.Bovim G, Fredriksen TA, Stolt-Nielsen A, Sjaastad O.
Neurolysis of the greater occipital nerve in cervicogenic headache. A follow up study.
Headache 1992; 32: 175–79.Barre N.
Sur un syndrome sympathique cervicale posterieure et sa cause frequente: l’arthrite cervicale.
Revue du Neurologie 1926; 33: 1246–48.Tamura T.
Cranial symptoms after cervical injury. Aetiology and treatment of the Barre-Lieou syndrome.
J Bone Joint Surg 1989; 71B: 283–87.Bogduk N, Lambert G, Duckworth JW.
The anatomy and physiology of the vertebral nerve in relation to cervical migraine.
Cephalalgia 1981; 1: 1–14.Lambert GA, Duckworth JW, Bogduk N, Lance JW.
Low pharmacological responsiveness of the vertebro-basilar circulation in macaca nemestrina monkeys.
Eur J Pharmacol 1984; 102: 451–58.Jansen J.
Surgical treatment of cervicogenic headache.
Cephalalgia 2008; 28 (suppl 1): 41–44.Diener HC, Kaminski M, Stappert G, Stolke D, Schoch B.
Lower cervical disc prolapse may cause cervicogenic headache: prospective study in patients undergoing surgery.
Cephalalgia 2007; 27: 1050–54.Amevo B, Aprill C, Bogduk N.
Abnormal instantaneous axes of rotation in patients with neck pain.
Spine 1992; 17: 748–56.Farina S, Granella F, Malferrari G, Manzoni C.
Headache and cervical spine disorders: classification and treatment with transcutaneous electrical nerve stimulation.
Headache 1986; 26: 431–33.Haldeman S, Dagenais S.
Cervicogenic Headaches: A Critical Review
Spine J 2001 (Jan); 1 (1): 31–46Nilsson N..
A Randomized Controlled Trial of the Effect of Spinal Manipulation
in the Treatment of Cervicogenic Headache
J Manipulative Physiol Ther. 1995 (Sep); 18 (7): 435—440Nillson N, Christensen HW, Hartvigsen J.
The Effect of Spinal Manipulation in the Treatment of Cervicogenic Headache
J Manipulative Physiol Ther 1997 (Jun); 20 (5): 326–330Vernon HT.
Spinal Manipulation and Headaches of Cervical Origin
J Manipulative Physiol Ther 1989 (Dec); 12 (6): 455–468Jull G, Trott P, Potter H, et al.
A Randomized Controlled Trial of Exercise and
Manipulative Therapy for Cervicogenic Headache
Spine (Phila Pa 1976) 2002 (Sep 1); 27 (17): 1835—1843Anthony M.
Cervicogenic headache: prevalence and response to local steroid therapy.
Clin Exp Rheumatol 2000; 18 (suppl 19): S59–64.Anthony M.
Headache and the greater occipital nerve.
Clin Neurol Neurosurg 1992; 94: 297–301.Pikus HJ, Phillips JM.
Characteristics of patients successfully treated for cervicogenic headache by surgical decompression of the second cervical root.
Headache 1995; 35: 621–29.Narouze SN, Casanova J, Mekhail N.
The longitudinal effectiveness of lateral atlantoaxial intra-articular steroid injection in the treatment of cervicogenic headache.
Pain Med 2007; 8: 184–88.Joseph B, Kumar B.
Gallie’s fusion for atlantoaxial arthrosis with occipital neuralgia.
Spine 1994; 19: 454–55.Ghanayem AJ, Leventhal M, Bohlman HH.
Osteoarthrosis of the atlanto-axial joints—long-term follow-up after treatment with arthrodesis.
J Bone Joint Surg 1996; 78A: 1300–07.Schaeren S, Jeanneret B.
Atlantoaxial osteoarthritis: case series and review of the literature.
Eur Spine J 2005; 14: 501–06.Slipman CW, Lipetz JS, Plastara CT, Jackson HW, Yang ST, Meyer AM.
Therapeutic zygapophyseal joint injections for headache emanating from the C2–3 joint.
Am J Phys Med Rehabil 2001; 80: 182–88.van Suijlekom HA, van Kleef M, Barendse GAM, Sluijter ME, Sjaastad O, Weber WEJ.
Radiofrequency cervical zygapophyseal joint neurotomy for cervicogenic headaches: a prospective study of 15 patients.
Funct Neurol 1998; 13: 297–303.Stovner LJ, Kolstad F, Helde G.
Radiofrequency denervation of facet joints C2–C6 in cervicogenic headache: a randomised, double-blind, sham-controlled study.
Cephalalgia 2004; 24: 821–30.Haspeslagh SR, van Suijlekom HA, Lame IE, Kessels AG, van Kleef M, Weber WE.
Randomised controlled trial of cervical radiofrequency lesions as a treatment for cervicogenic headache.
BMC Anesthesiol 2006; 6: 1.Govind J, King W, Bailey B, Bogduk N.
Radiofrequency neurotomy for the treatment of third occipital headache.
J Neurol Neurosurg Psychiatr 2003; 74: 88–93.Barnsley L.
Percutaneous radiofrequency neurotomy for chronic neck pain: outcomes in a series of consecutive patients.
Pain Med 2005; 6: 282–86.International Spine Intervention Society.
Percutaneous radiofrequency cervical medial branch neurotomy.
In: Bogduk N, ed. Practice Guidelines for Spinal Diagnostic and Treatment Procedures.
San Francisco: International Spine Intervention Society, 2004: 249–84.Lord SM, Barnsley L, Wallis BJ, McDonald GJ, Bogduk N.
Percutaneous radio-frequency neurotomy for chronic cervical zygapophysial-joint pain.
N Engl J Med 1996; 335: 1721–26.Lipton RB, Silberstein SD, Saper JR, Bigal ME, Goadsby PJ.
Why headache treatment fails.
Neurology 2003; 60: 1064–70
Return to CERVICOGENIC HEADACHE
Since 7-20-2017


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |