

Trends, Major Medical Complications, and Charges Associated
with Surgery for Lumbar Spinal Stenosis in Older AdultsThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: JAMA 2010 (Apr 7); 303 (13): 1259û1265 ~ FULL TEXT
OPEN ACCESS Richard A. Deyo, MD, MPH; Sohail K. Mirza, MD, MPH; Brook I. Martin, MPH;
William Kreuter, MPA; David C. Goodman, MD, MS; Jeffrey G. Jarvik, MD, MPH
Department of Family Medicine,
Mail Code FM, Oregon Health and Science University,
3181 SW Sam Jackson Park Rd,
Portland, OR 97239, USA.
Thanks to ChiroACCESS for the following commentary:
There is a lack of evidence-based support for the efficacy of complex fusion surgeries over conservative surgical decompression for elderly stenosis patients. There is, however, a significant financial incentive to both hospitals and surgeons to perform the complex fusions. Spinal stenosis is the most frequent cause for spinal surgery in the elderly. There has been a slight decrease in these surgeries between 2002 and 2007. However, there has also been an overall 15 fold increase in the more complex spinal fusions (360 degree spine fusions).
Deyo et. al. in yesterdayÆs issue (April 7, 2010) of the Journal of the American Medical Association concludes that ôIt is unclear why more complex operations are increasing. It seems implausible that the number of patients with the most complex spinal pathology increased 15ûfold in just 6 years.ö
The introduction and marketing of new surgical devices and the influence of key opinion leaders may stimulate more invasive surgery, even in the absence of new indicationsàfinancial incentives to hospitals and surgeons for more complex procedures may play a roleàö There is a significant difference in mean hospital costs for simple decompression versus complex surgical fusion. The cost of decompression is $23,724 compared to an average of $80,888 for complex fusion. Despite the much higher cost, there is no evidence of superior outcomes and there is greater morbidity associated with the complex fusion. The surgeon is typically reimbursed only $600 to $800 for simple decompression and approximately ten times more, $6,000 to $8,000 for the complex fusion.
In a JAMA editorial that accompanied this study and was written by Dr. Carragee of Stanford University School of Medicine, the following comment was made ôIn 2007, the final year of data reported in the study by Deyo et al, Consumer Reports rated spinal surgery as number 1 on its list of overused tests and treatments. This was a harsh rebuke given the benefit associated with many common spinal surgeries. However, the findings from the study by Deyo et al should not only remind patients, surgeons, and payors that the efficacy of basic spinal techniques must be assessed carefully against the plethora of unproven but financially attractive alternatives, but also should serve as an important reminder that as currently configured, financial incentives and market forces do not favor this careful assessment before technologies are widely adopted. When applied broadly across medical care in the United States, the result is a formidable economic and social problem.ö
These studies reflect much of what is plaguing our broken health care system. Third party payors, including the government, fail to hold all health professionals to the same evidence-based standards. There is little or no accountability when significantly more expensive surgery, with no evidence of superior effectiveness, is routinely reimbursed. There is discrimination of health care providers and the needs and desires of the patient are secondary to matters of finance and politics.
CONTEXT: In recent decades, the fastest growth in lumbar surgery occurred in older patients with spinal stenosis. Trials indicate that for selected patients, decompressive surgery offers an advantage over nonoperative treatment, but surgeons often recommend more invasive fusion procedures. Comorbidity is common in older patients, so benefits and risks must be carefully weighed in the choice of surgical procedure.
OBJECTIVE: To examine trends in use of different types of stenosis operations and the association of complications and resource use with surgical complexity.
DESIGN, SETTING, AND PATIENTS: Retrospective cohort analysis of Medicare claims for 2002û2007, focusing on 2007 to assess complications and resource use in US hospitals. Operations for Medicare recipients undergoing surgery for lumbar stenosis (n = 32,152 in the first 11 months of 2007) were grouped into 3 gradations of invasiveness: decompression alone, simple fusion (1 or 2 disk levels, single surgical approach), or complex fusion (more than 2 disk levels or combined anterior and posterior approach).
MAIN OUTCOME MEASURES: Rates of the 3 types of surgery, major complications, postoperative mortality, and resource use.
RESULTS: Overall, surgical rates declined slightly from 2002û2007, but the rate of complex fusion procedures increased 15ûfold, from 1.3 to 19.9 per 100,000 beneficiaries. Life-threatening complications increased with increasing surgical invasiveness, from 2.3% among patients having decompression alone to 5.6% among those having complex fusions. After adjustment for age, comorbidity, previous spine surgery, and other features, the odds ratio (OR) of life-threatening complications for complex fusion compared with decompression alone was 2.95 (95% confidence interval [CI], 2.50û3.49). A similar pattern was observed for rehospitalization within 30 days, which occurred for 7.8% of patients undergoing decompression and 13.0% having a complex fusion (adjusted OR, 1.94; 95% CI, 1.74û2.17). Adjusted mean hospital charges for complex fusion procedures were US $80,888 compared with US $23,724 for decompression alone.
CONCLUSIONS: Among Medicare recipients, between 2002 and 2007, the frequency of complex fusion procedures for spinal stenosis increased while the frequency of decompression surgery and simple fusions decreased. In 2007, compared with decompression, simple fusion and complex fusion were associated with increased risk of major complications, 30ûday mortality, and resource use.
From the Full-Text Article:
Background
In planning spine operations, surgeons have wide discretion. For pain-related surgery, consensus on indications for specific procedures (eg decompression alone or decompression plus fusion) is generally lacking [1û3] despite randomized trials for some condition and procedure combinations. [4û10] Furthermore, individual surgeon preferences may outweigh patient and disease characteristics in choosing procedures. [3] Such choices are important, because greater invasiveness is associated with greater complications, health care use, and mortality, [4, 11, 12] but generally similar clinical benefit. [7û10, 12]
Risks of spine surgery are particularly important in older adults, for whom stenosis is the most common surgical indication. Symptomatic lumbar stenosis results from progressive degenerative changes in intervertebral joints and ligamentous structures, leading to spinal canal and neural foraminal narrowing. Diagnosis and treatment require complex judgments integrating data from imaging, clinical findings, and the patientÆs clinical course.
Surgery for spinal stenosis was the fastest-growing type of lumbar surgery in the U.S from 1980 to 2000. [13, 14] Randomized trials indicate that for severely affected patients, decompression without fusion offers greater efficacy than non-surgical treatments. [5, 6] However, assessment of therapeutic safety often requires observational data, because randomized trials may exclude high-risk patients, be too short to identify some risks, or be too small to detect uncommon events. [15]
Better information on surgical complications would help surgeons, referring physicians, and patients weigh benefits and risks, and permit more individualized decision-making. We therefore studied the Medicare population (adults aged 65 and older, who receive federal health insurance coverage) to better define (1) trends in the use of various surgical procedures for lumbar stenosis; (2) how complications vary as a function of age, comorbid conditions, previous surgery, and complexity of the surgical procedure; and (3) healthcare use associated with stenosis surgery, including hospital length of stay, hospital charges, rehospitalization, and postoperative nursing home care.
METHODS
Data Source
We used Part A claims (the Medicare Provider Analysis and Review, or MedPAR database) for the most recent available years (2002û2007) to examine trends in use of various surgical procedures. This database includes 100% of Medicare hospital claims, using surgical procedure codes from the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM). We excluded beneficiaries receiving Social Security Disability Income, those with end-stage renal disease, or those enrolled in a health maintenance organization (HMO). The latter are often excluded from Medicare data analyses as detailed claims may not be available. [16, 17]
These data files have unique patient identifiers that allow linkage among files and identification of repeat hospitalizations. Institutional Review Boards at the University of Washington, Oregon Health and Science University, and Dartmouth College approved the project.
Trends in Surgical Procedures
To examine surgical trends, we selected patients aged 65 years and older with a primary diagnosis of lumbar spinal stenosis (98.2% of cases) or ôspondylogenic compression of lumbar spinal cordö. We included those with a surgical procedure indicating any combination of discectomy, laminectomy, or fusion. We excluded patients if any diagnosis at the index hospitalization indicated cancer, vehicular accident, spinal infection, inflammatory spondyloarthropathies, vertebral fractures or dislocations, or cervical or thoracic spine procedures.
Categorizing Surgical Procedures
We defined 3 broad categories of spine surgery: decompression, simple fusion, or complex fusion. Decompression included any combination of discectomy and laminectomy without fusion. A simple fusion involved a single surgical approach (only codes for anterior fusion, or only for transverse process or posterior fusion techniques), and involved only one or two disc levels (corresponding to the ICD code for fusion involving 2 or 3 vertebrae). Complex fusions involved 360ûdegree spine fusion by single incision (during years this code was available); any combination of anterior with either transverse process or posterior fusion techniques; or any fusion of more than two disc levels. If the number of levels was not coded, cases were classified by approach only (single vs. combined anterior and posterior approach).
Complications
To study complications, we focused on January 1 to December 1, 2007, providing 30 days of postoperative observation for all patients. The index operation was the first operation meeting our eligibility requirements. We selected only patients aged 66 years and older, so that most would have a full prior year of Medicare eligibility to identify recent previous spine surgery, hospitalizations, and comorbid conditions.
Complications in 3 categories were considered: major medical complications, wound complications, and mortality. These may be associated with any surgery, and are not specific for lumbar spine surgery. Major medical complications included procedure codes for cardiopulmonary resuscitation or repeat post-operative endotracheal intubation and mechanical ventilation. They included diagnosis codes for cardiorespiratory arrest, acute myocardial infarction, respiratory failure, pulmonary embolism, bacterial pneumonia, aspiration pneumonia, pneumonia with unknown organism, and stroke, excluding late effects. These complications were chosen because of their major impact on health and more consistent coding, in contrast to minor complications. [18]
Wound complications included hemorrhage, hematoma or seroma complicating a procedure; disruption of operation wound; non-healing surgical wound; postoperative infection; and other infection. We also included patients with a procedure code for ôexcisional debridement of wound, infection or burn,ö or a Diagnosis-Related Group code for wound debridement and skin graft.
Mortality was determined from a file identifying date of death. We calculated mortality within 30 days of hospital discharge, including in-hospital death.
Healthcare utilization
MedPAR includes length of hospital stay and hospital charges, but not professional fees. The file also identifies discharges to a skilled nursing facility. We examined rehospitalizations within 30 days because short-term rehospitalizations are a target for quality improvement, [17] suggesting complications, poor discharge planning, inadequate outpatient follow-up, or other problems.
Measures of Comorbidity
We modified the comorbidity index of Quan and colleagues, [19, 20] We removed codes such as acute myocardial infarction or acute stroke that could represent postoperative complications when recorded at the index hospitalization. However, we used the full index to identify comorbid conditions in any hospitalization during the previous year. We also calculated number of hospitalizations in the year prior to the index hospitalization (excluding those for spine surgery), as a marker of overall disease burden.
Previous spine surgery
We identified patients with previous lumbar surgery in two ways. First, we identified diagnosis or procedure codes suggesting previous surgery, such as postlaminectomy syndrome, or refusion. Second, we searched hospitalizations in the previous year to identify lumbar spine procedures.
Statistical Analysis
Trends in use of surgical procedures were examined using both volume and rates of relevant procedures per 100,000 Medicare beneficiaries, age and sex adjusted by the direct method to the 2002 Medicare population. Charges were adjusted for inflation using the health care component of the consumer price index, adjusting to 2009 dollars.
Proportions of patients with complications, rehospitalizations, or nursing home discharge among subgroups were compared using chi-square analyses for bivariate analyses, and logistic regression for multivariate analyses. In regressions, these events were modeled as a function of age, race, gender, comorbidity, previous spine surgery, secondary diagnoses of spondylolisthesis or scoliosis, and complexity of surgical procedure.
Length of stay and hospital charges were compared among subgroups with t-tests or analysis of variance, then modeled in linear regressions. Regressions were performed using untransformed charges, because mean estimates were similar to those of alternative approaches that better account for skewed data; [21û24] means are often sufficient in large datasets. [22] All significance tests were 2ûsided, with an alpha of 0.05. Statistical analysis was performed with Stata software, version 10 (Statacorp, College Station, Texas).
RESULTS
Surgical Trends
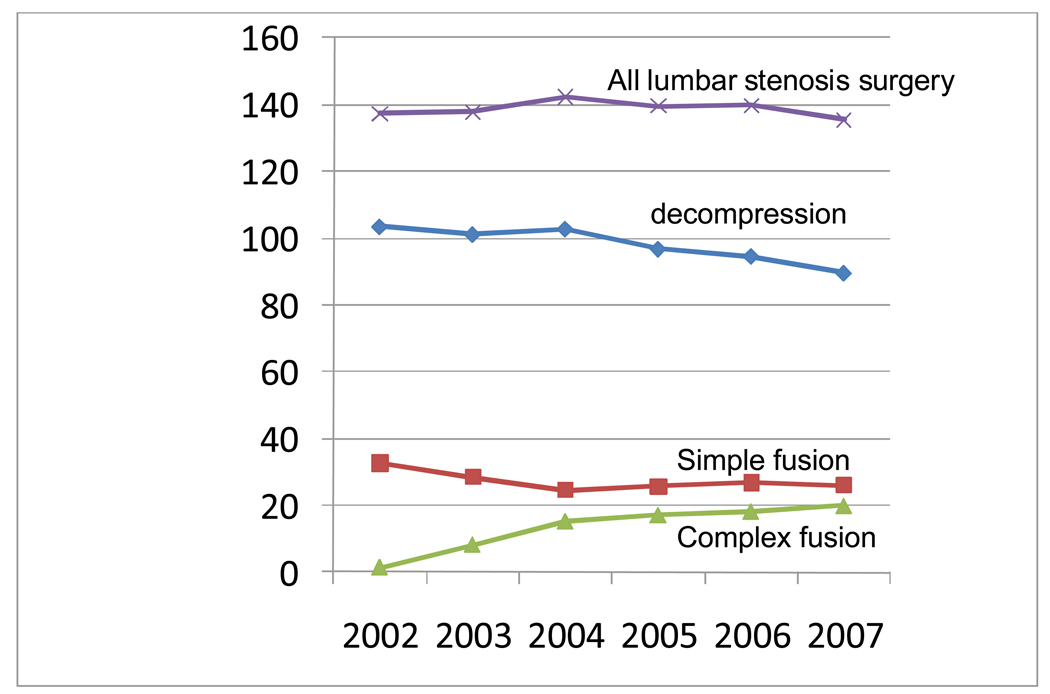
Figure 1 In 2007, there were 37,598 operations for a primary diagnosis of lumbar stenosis among patients meeting our criteria. . The aggregate hospital bill was nearly $1.65 billion (2009 dollars). Over the years 2002û2007, the number of operations and the rate per 100,000 beneficiaries declined slightly (Figure 1). The adjusted rate of lumbar stenosis surgery per 100,000 Medicare beneficiaries was 137.4 in 2002 and 135.5 in 2007.
Rates of decompression surgery and simple fusions declined during these years. However, rates of complex fusion surgery increased from 1.3 per 100,000 (just under 1% of operations) to 19.9 per 100,000 (14.6% of operations), a 15ûfold increase (Figure 1). Correspondingly, although the overall procedure rate fell 1.4%, aggregate hospital charges increased 40% (inflation adjusted).
Complications

Table 1

Table 2 The 2007 study cohort, limited to index procedures among patients aged 66 years and older for 11 months, included 32,152 patients with a mean age of 75.0 years; 54% were women. Among these stenosis patients, 5,915 (18.4%) had a secondary diagnosis of spondylolisthesis and 1,652 (5.1%) had a secondary diagnosis of scoliosis. These secondary diagnoses increased the likelihood of a fusion procedure (Table 1). Patients with scoliosis had the highest percentage of complex fusion procedures. Although patients without spondylolisthesis or scoliosis were less likely to undergo fusion surgery, they accounted for 50% of such procedures.
Major medical complications were reported in 3.1% of patients overall, and wound complications in 1.2%. Mortality was 0.4% within 30 days of discharge. Major medical complications and mortality increased modestly with increasing age and were generally similar for men and women. Major complications and mortality were slightly higher among non-white patients than among Whites (Table 2). Wound complications were not significantly associated with demographic factors.
Major medical complications and mortality rose with increasing comorbidity (p<0.05). For example, major medical complications occurred in 5.3% of patients with a comorbidity score of 3 or greater compared to 2.5% among those with a score of zero. (Table 2). Complication rates were only modestly affected by comorbid diabetes, obesity, or chronic coronary disease. However, complications and mortality were substantially greater among patients with chronic lung diseases than those without. Hospitalizations in the previous year strongly predicted complications and mortality (Table 2).
Operative features were also associated with complications. Previous spine surgery was modestly associated with medical complications or mortality, but strongly associated with wound complications (4.6% vs 1.0% among those without prior surgery, p<0.05). The type of index procedure was associated with major medical complications, wound complications, rehospitalization, and mortality. For example, complex fusion operations were associated with a 5.2% rate of major medical complications compared to 2.1% for decompression alone, and a 30ûday mortality of 0.6% versus 0.3% for decompression (all p<0.05, Table 2). Results were similar considering only patients without spondylolisthesis or scoliosis.
In a sensitivity analysis, we considered patients with any diagnosis of stenosis (primary or secondary), adding 7,561 index operations. Complication estimates were similar and conclusions were unchanged. The most common accompanying diagnoses were spondylolisthesis, scoliosis, and herniated or degenerative disc disease.

Table 3 In multivariate analyses, we focused on the association of surgical procedure with outcomes, adjusting for age, gender, race, comorbidity, previous hospitalizations, spondylolisthesis, scoliosis, and previous back surgery. We combined major medical complications and 30ûday mortality to represent ôlife-threatening complications.ö Age, comorbidity, and previous hospitalizations remained independently associated with life-threatening complications. Complex fusion procedures had an odds ratio of 2.95 (95% CI 2.50, 3.49) for life-threatening complications, compared to decompression alone (Table 3). For wound complications, previous surgery and greater surgical complexity were the greatest risk factors after adjustment.
Health Care Utilization

Table 4 Length of hospitalization varied only modestly by age, race, or gender. However, it rose with increasing comorbidity or previous hospitalizations. Patients with previous lumbar surgery had almost a day longer hospitalization than those undergoing a first operation. Patients having a complex fusion had almost a 2ûday longer stay than those having decompression alone (Table 4).
Mean hospital charges decreased with increasing age, perhaps reflecting less complex surgery in the oldest old. Charges increased modestly with increasing comorbidity and more substantially with previous hospitalizations. The greatest variation occurred with type of surgery. Complex fusion operations resulted in mean hospital charges ($80,888) over 3 times those for decompressions alone ($23,724, Table 4).
Discharges to a skilled nursing facility rose with increasing age, comorbidity, and previous hospitalizations. Among patients aged 80 and older, more than 20% were discharged to a skilled nursing facility. Such discharges also rose with increasing surgical complexity. Approximately 20% of those having any fusion procedure were discharged to a skilled nursing facility, twice the percentage among those having decompression alone.
The likelihood of 30ûday rehospitalization increased steadily with age, comorbidity, and previous hospitalizations. It also rose with increasing surgical complexity (Table 4). Charges and utilization patterns were similar among the subgroup with no scoliosis or spondylolisthesis, or for those with any diagnosis of stenosis (primary or secondary)
In regression models, length of stay, hospital charges, nursing home discharge, and rehospitalization remained significantly associated with type of surgical procedure after adjustment for patient demographic and clinical characteristics (Table 3).
DISCUSSION
Rates of surgery for lumbar stenosis declined slightly from 2002û2007, but use of more complex procedures increased substantially. More complex procedures were associated with greater complications, mortality, hospital charges, and other measures of healthcare utilization, even after adjustment for patient demographic and clinical characteristics. Age was less predictive than comorbidity or type of surgical procedure.
It is unclear why more complex operations are increasing. It seems implausible that the number of patients with the most complex spinal pathology increased 15ûfold in just 6 years. The introduction and marketing of new surgical devices and the influence of key opinion leaders may stimulate more invasive surgery, even in the absence of new indications.14 Surgeons may believe more aggressive intervention produces better outcomes. Improvements in surgical technique, anesthetic technique, and supportive care may make more invasive surgery feasible when risks formerly would have been prohibitive. Financial incentives to hospitals and surgeons for more complex procedures may play a role, as may desires of surgeons to be local innovators.
Geographic variations in spine surgery rates are among the largest observed for surgical procedures, and variations in use of fusion surgery exceed those for decompression alone. [1, 25] Such variations persist despite extensive research in this area, in part because of the difficulty of conducting randomized surgical trials. They suggest a poor consensus on indications for surgery or the choice of particular procedures. Studies among spine surgeons indicate substantial variability in decisions to operate, perform a fusion, or use surgical implants. [2, 3] Our study shows clinically important consequences of these choices.
Evidence for greater efficacy of more complex procedures for lumbar stenosis is lacking. [26] For patients who also have spondylolisthesis or scoliosis, spinal fusion may improve outcomes over decompression alone. [8, 27] However, trials establishing an advantage of surgery over non-surgical care for stenosis alone focused overwhelmingly on decompression without fusion. [5, 6] Some trials for lumbar stenosis suggest equivalent efficacy for decompression alone versus decompression and fusion, in the absence of spondylolisthesis. [7]
It is not surprising that fusion procedures are associated with more complications than decompression alone. Compared to decompression, spine fusion requires more extensive dissection, decortication of bone, and longer operative time, and often involves placement of implants. This study confirms previous findings that fusion is associated with greater complications and post-operative mortality than decompression alone. [11, 28]
For other indications, randomized trials suggest that fusion by a single approach with bone grafting alone, fusion with implants, and combined anterior and posterior fusion with implants have similar efficacy for improving pain and function. [4, 12] For patients with stenosis and degenerative spondylolisthesis, fusions with and without implants have similar clinical outcomes. [8, 29, 30] However, more complex procedures are associated with more complications. [12, 29, 31] Complications also increase with more operated levels, [32] and with revision surgery. [7û10, 33, 34] Our data indicate that these patterns hold true for older patients with spinal stenosis.
Patient demographic and clinical characteristics are generally not matters of choice, but surgeons and patients control the choice of surgical procedure. In the absence of compelling data showing better pain relief or function with more complex surgery, our results may suggest using the least invasive procedure that accomplishes clinical goals. This contrasts with a competing theory, that surgeons should correct every anatomic abnormality, hoping to avoid future symptoms. The theory behind this ôprophylacticö approach remains unproven, and the risk of greater complications from more extensive surgery must be weighed against potential benefits. Thus, it may be prudent to consider whether decompression alone is sufficient; whether stabilizing structures such as facet joints or interspinous ligaments can be preserved; and if a fusion is planned, how much instrumentation and graft material supplementation is needed.
Our study has the advantage of including all Medicare patients having surgery for spinal stenosis, and not selected patients, centers, or surgeons. It includes nearly complete data on repeat hospitalizations and mortality. However, there are also limitations. Diagnoses and procedures may be miscoded, though the data are used for billing and subject to audit. Furthermore, spine operations appear to be generally coded accurately. [35] Surgeons use varying definitions of spine instability, and ICD-9 diagnosis codes may not represent this concept well. The level of detail in ICD-9 spine surgery codes is limited, and information on use of implants is incomplete. Complications may not be consistently recorded, but surgical complications are more reliably coded in large databases than complications from medical therapy. [18] Further, the complications we examined are more consistently coded than minor complications.18 The specificity of claims data for complications is high (unlikely to be coded without a real complication), though sensitivity may be lower (some complications not coded). Thus, we are more likely to underestimate than overestimate complication rates.
Surgeons tailor operations to the nature, extent, and location of an individualÆs pathology, but claims data do not indicate severity or extent of anatomic changes, patient symptoms, or functional status. Nonetheless, studies report substantial variability in surgical decision-making, even for similar patients. [1û3] Further, accounting for coexisting spondylolisthesis or scoliosis did not alter our results.
Another limitation is that we have presented hospital charges rather than actual resource costs or reimbursements, which typically are substantially less than charges. The relationship between costs and charges is complex and varies by hospital and by type of service.
Among Medicare recipients, between 2002 and 2007, the frequency of complex fusion procedures for spinal stenosis increased, while the frequency of decompression surgery and simple fusions decreased. In 2007, compared with decompression, simple fusion and complex fusion were associated with increased risk of major complications, 30ûday mortality, and resource use.
References:
Weinstein JN, Lurie JD, Olson PR, Bronner KK, Fisher ES.
United States trends and regional variations in lumbar spine surgery: 1992û2003.
Spine. 2006;31:2707û2714Irwin ZN, Hilibrand A, Gustavel M, et al.
Variation in surgical decision making for degenerative spinal disorders.
Part I: lumbar spine.
Spine. 2005;30:2208û2213Katz JN, Lipson SJ, Lew RA, et al.
Lumbar laminectomy alone or with instrumented or noninstrumented
arthrodesis in degenerative lumbar spinal stenosis.
Patient selection, costs, and surgical outcomes.
Spine. 1997;22:1123û1131Fritzell P, Hagg O, Wessbert P, Nordwall A
and the Swedish Lumbar Spine Study Group.
2001 Volvo Award winner in clinical studies: lumbar fusion versus
nonsurgical treatment of chronic low back pain: a multicenter
randomized controlled trial from the Swedish Lumbar Spine Study Group.
Spine. 2001;26:2521û2532Weinstein JN, Tosteson TD, Lurie JD, et al.
Surgical versus nonsurgical therapy for lumbar spinal stenosis.
N Engl J Med. 2008;358:794û810Malmivaara A, Slatis P, Heliovaara M, et al.
for the Finnish Lumbar Spinal Research group.
Surgical or nonoperative treatment for lumbar spinal stenosis?
A randomized controlled trial.
Spine. 2007;32:1û8Grob D, Humke T, Dvorak J.
Degenerative lumbar spinal stenosis. Decompression with and without arthrodesis.
J Bone Joint Surg Am. 1995;77:1036û1041Fischgrund JS, Mackay M, Herkowitz HN, et al.
1997 Volvo Award winner in clinical studies.
Degenerative lumbar spondylolisthesis with spinal stenosis: a prospective,
randomized study comparing decompressive laminectomy and arthrodesis
with and without spinal instrumentation.
Spine. 1997;22:2807û2812Moller H, Hedlund R.
Instrumented and noninstrumented posterolateral fusion in
adult spondylolisthesis û a prospective randomized study: part 2.
Spine. 2000;25:1716û1721Bjarke Christensen F, Stender Hansen E, Laursen M, Thomsen K, Bulnger CE.
Long-term functional outcome of pedicle screw instrumentation as a support
for posterolateral spinal fusion: randomized clinical study with a 5-year follow-up.
Spine. 2002;27:1269û1277Ciol MA, Deyo RA, Howell E, Kreif S.
An assessment of surgery for spinal stenosis: time trends,
geographic variations, complications, and reoperations.
J Am Geriatr Soc. 1996;44:285û290Fritzell P, Hagg O, Nordwall A.
Complications in lumbar fusion surgery for chronic low back pain:
comparison of three surgical techniques used in a prospective
randomized study. A report from the Swedish Lumbar Spine Study Group.
Eur Spine J. 2003;12:178û189Taylor VM, Deyo RA, Cherkin DC, Kreuter W.
Low back pain hospitalization: recent United States trends and regional variations.
Spine. 1994;19:1207û1213Deyo RA, Gray DT, Kreuter W, Mirza S, Martin BI.
United States trends in lumbar fusion surgery for degenerative conditions.
Spine. 2005;30:1441û1445Chou R, Helfand M.
Challenges in systematic reviews that assess treatment harms.
Ann Intern Med. 2005;142:1090û1099Schermerhorn ML, OÆMalley AJ, Jhaveri A, Cotterill P, Pomposelli F, Landon EE.
Endovascular vs. open repair of abdominal aortic aneurysms in the Medicare population.
N Engl J Med. 2008;358:464û474Jencks SF, Williams MV, Coleman EA.
Rehospitalizations among patients in the Medicare fee-for-service program.
N Engl J Med. 2009;360:1418û1428Lawthers AG, McCarthy E, Davis RB, Teterson LE, Palmer RH, Iezzoni LI.
Identification of In-hospital complications from claims data:
is it valid?
Medical Care. 2000;38:785û795Quan H, Sundararajan V, Halfon P, et al.
Coding algorithms for defining comorbidities in ICD-9-CM
and ICD-10 administrative data.
Medical Care. 2005;43:1130û1139Deyo RA, Cherkin DC, Ciol MA.
Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases.
J Clin Epidemiol. 1992;45(6):613û619Buntin MB, Zaslavsky AM.
Too much ado about two-part models and transformation?
Comparing methods of modeling Medicare expenditures.
J Health Econ. 2004;23:525û542Lumley T, Diehr P, Emerson S, Chen L.
The importance of the normality assumption in
large public health data sets.
Annu Rev Public Health. 2002;23:151û169Glick HA, Doshi JA, Sonnad SS, Polsky D.
Economic evaluation of clinical trials.
New York: Oxford University Press; 2007Duan N.
Smearing estimate: a nonparametric retransformation method.
J Am Stat Assoc. 1983;78:605û610Deyo RA, Mirza SK.
Trends and variations in the use of spine surgery.
Clin Orthop Rel Res. 2006;443:139û146Deyo RA, Nachemson A, Mirza SK.
Spinal fusion surgery û the case for restraint.
N Engl J Med. 2004;350:722û726Herkowitz HN, Kurz LT.
Degenerative lumbar spondylolisthesis with spinal stenosis:
a prospective study comparing decompression with
decompression and intertransverse process arthrodesis.
J Bone Joint Surg Am. 1991;73:802û808Deyo RA, Ciol MA, Cherkin DC, Loeser JD, Bigos SJ.
Lumbar spinal fusion: a cohort study of complications,
reoperations, and resource use in the Medicare population.
Spine. 1993;18:1463û1470Thomsen K, Christensen FB, Eiskjaer SP, Hansen ES, Fruensgaard S, Bunger CE.
1997 Volvo Award winner in clinical studies.
The effect of pedicle screw instrumentation on functional outcome
and fusion rates in posterolateral lumbar spinal fusion:
a prospective randomized clinical study.
Spine. 1997;22:2813û2822Abdu WA, Lurie JD, Spratt KF, et al.
Degenerative spondylolisthesis: does fusion method influence outcome?
Four-year results of the Spine Patient Outcomes Research Trial.
Spine. 2009 Sept 14; [epub ahead of print]Mirza SK, Deyo RA, Heagerty PJ, Konodi MA, Lee L, Turner J, Goodkin R.
Development of an index to characterize the ôinvasivenessö of spine surgery:
validation by comparison to blood loss and operative time.
Spine. 2008;33:2651û2661Carreon LY, Puno RM, Dimar JR, Glassman SD, Johnson JR.
Perioperative complications of posterior lumbar
decompression and arthrodesis in older adults.
J Bone Joint Surg. 2003:2089û2092. 85-AGlassman SD, Carreon LY, Dimar JR, Campbell MJ, Puno RM, Johnson JR.
clinical outcomes in older patients after posterolateral lumbar fusion.
Spine J. 2007;7:547û551Esses SI, Sachs BL, Dreyzin V.
Complications associated with the technique of pedicle screw fixation.
A selected survey of ABS members.
Spine. 1993;18:2231û2239Cherkin DC, Deyo RA.
Nonsurgical hospitalization for low-back pain:
is it necessary?
Spine. 1993;18:1728û1735.
Return to LOW BACK PAIN
Return to SPINAL STENOSIS
Since 4û10û2010


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |
