

A Hypothesis of Chronic Back Pain:
Ligament Subfailure Injuries Lead to Muscle Control DysfunctionThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: European Spine Journal 2006 (May); 15 (5): 668–676 ~ FULL TEXT
OPEN ACCESS Manohar M. Panjabi
Biomechanics Research Laboratory,
Department of Orthopaedics and Rehabilitation,
Yale University School of Medicine,
New Haven, CT 06520-8071, USA.
manohar.panjabi@yale.edu

Clinical reports and research studies have documented the behavior of chronic low back and neck pain patients. A few hypotheses have attempted to explain these varied clinical and research findings. A new hypothesis, based upon the concept that subfailure injuries of ligaments (spinal ligaments, disc annulus and facet capsules) may cause chronic back pain due to muscle control dysfunction, is presented. The hypothesis has the following sequential steps. Single trauma or cumulative microtrauma causes subfailure injuries of the ligaments and embedded mechanoreceptors. The injured mechanoreceptors generate corrupted transducer signals, which lead to corrupted muscle response pattern produced by the neuromuscular control unit.
Muscle coordination and individual muscle force characteristics, i.e. onset, magnitude, and shut-off, are disrupted. This results in abnormal stresses and strains in the ligaments, mechanoreceptors and muscles, and excessive loading of the facet joints. Due to inherently poor healing of spinal ligaments, accelerated degeneration of disc and facet joints may occur. The abnormal conditions may persist, and, over time, may lead to chronic back pain via inflammation of neural tissues. The hypothesis explains many of the clinical observations and research findings about the back pain patients. The hypothesis may help in a better understanding of chronic low back and neck pain patients, and in improved clinical management.
From the Full-Text Article:
Introduction
Low back pain is an important societal problem with significant costs. Up to 70–85% of the population in industrialized societies experience low back pain at least once in their lifetime, with point prevalence of about 30%. [1, 24] The total cost of low back pain has been estimated to exceed 50 billion dollars per year in the USA. [17] Although neck pain due to whiplash-associated disorder is less common and less costly, awareness of this disorder, diagnosis and treatment are equally baffling. [63] The term “back pain” as used here does not include back pain due to known infections, tumor, systemic disease, fractures or fracture dislocations. [73] Further, the term used here refers generally to the entire spine but in particular to the cervical and lumbar regions.
Back pain is complex. The exact cause of most back (low back and neck) pain remains unproven. [72] The multi-factorial nature of back pain is well recognized with respect to its causes, diagnosis, chronicity, disability and treatment. [73] Abnormal mechanics of the spinal column has been hypothesized to lead to back pain via nociceptive sensors. [72]
The path from abnormal mechanics to nociceptive sensation may go viainflammation [8, 11],
biochemical and nutritional changes [6],
immunological factors [44], and
changes in the structure and material of the endplates [6] and discs [40, 41], and neural structures, such as nerve ingrowth into diseased intervertebral disc. [15, 16]The abnormal mechanics of the spine may be due to degenerative changes of the spinal column [18] and/or injury of the ligaments. [43] Most likely, the initiating event is some kind of trauma involving the spine. It may be a single trauma due to an accident or microtrauma caused by repetitive motion over a long time. It is also possible that spinal muscles will fire in an uncoordinated way in response to sudden fear of injury, such as when one misjudges the depth of a step. All these events may cause spinal ligament injury. Adverse psycho–social factors may also play an important role in transforming the back pain into disability. [3]
The research literature on chronic back pain is vast. However, there are some important and common observations. Chronic low back pain patients have delayed muscle response when asked to perform a task [65] or when the spine is suddenly loaded [35], or in anticipation of raising an arm to horizontal position [20], and also delayed muscle shut-off after the external challenge has been withdrawn. [52] Further, they show poorer spinal posture control and balance, especially during complex tasks, when compared to subjects without back pain. [10, 33, 53] The findings in neck pain patients are similar, although the number of studies is fewer. Patients with whiplash-associated disorders have disrupted neck motion [2, 4, 14, 27, 34, 49, 51] and less efficient muscle control. [14, 19, 22, 31, 34]
A few hypotheses have attempted to explain the clinical observations and research findings in back pain patients. As the nociceptive sensors are present in most components of the spinal column, the hypotheses have focused on disruption of the spinal column and its components, such as spinal column degeneration [25], injury and clinical instability [47, 73]; facet joint injury [13], and inferior facet-tip impingement on the lamina [77], and Schmorl’s nodes. [29] Others have focused on spinal muscles. The pain adaptation [32] and pain–spasm–pain [54] hypotheses were evaluated in a recent review article. [69] The evidence was mixed, and authors suggested that other models, such as spinal instability [46, 47], may be explored. The role played by the injury to the mechanoreceptors embedded in the ligaments of the spinal column has not been explored by any hypothesis.
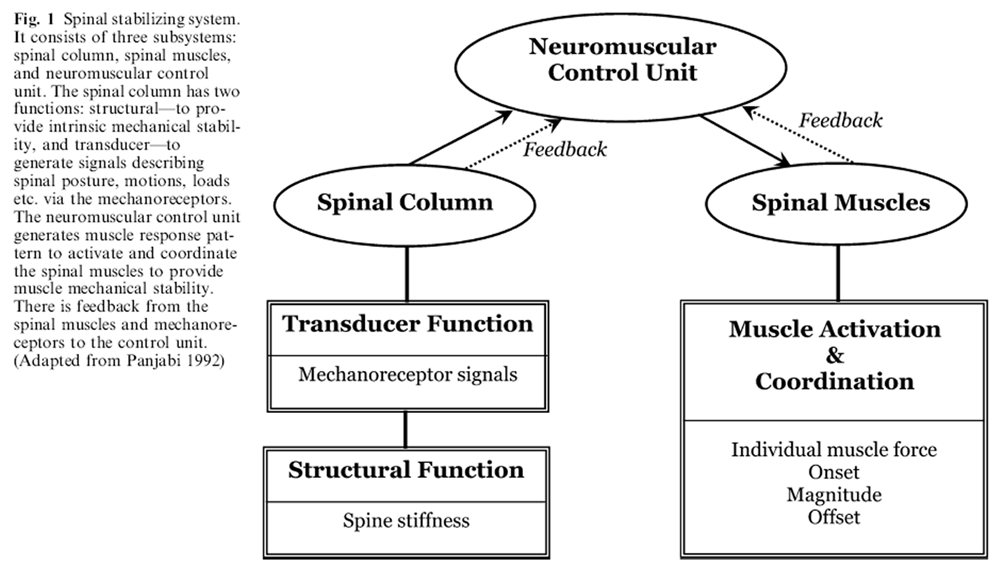
Figure 1 The spinal column, consisting of ligaments (spinal ligaments, discs annulus and facet capsules) and vertebrae, is one of the three subsystems of the spinal stabilizing system. [46] The other two are the spinal muscles and neuromuscular control unit, Figure 1. The spinal column has two functions: structural and transducer. The structural function provides stiffness to the spine. The transducer function provides the information needed to precisely characterize the spinal posture, vertebral motions, spinal loads etc. to the neuromuscular control unit via innumerable mechanoreceptors present in the spinal column ligaments [26, 58], facet capsules [11, 36, 76] and the disc annulus. [26] These mechanical transducers provide information to the neuromuscular control unit which helps to generate muscular spinal stability via the spinal muscle system and neuromuscular control unit. [46]
The criterion used by the neuromuscular unit is hypothesized to be the need for adequate and overall mechanical stability of the spine. If the structural function is compromised, due to injury or degeneration, then the muscular stability is increased to compensate the loss. What happens if the transducer function of the ligaments of the spinal column is compromised? This has not been explored. There is evidence from animal studies that the stimulation of the ligaments of the spine (disc and facets [21], and ligaments [59, 62]) results in spinal muscle firing. The mechanoreceptor-muscle firing relationships are modulated by several factors, such as ligament fatigue [61], static flexed posture [60], and cumulative microtrauma. [75]
The observations from animal studies just mentioned, together with the possibility of transducer dysfunction in back pain patients, form the basis of a new back pain hypothesis. The purpose is to describe the hypothesis, use the hypothesis to explain the various important research findings, and suggest possible treatment options.
The hypothesis
The hypothesis consists of the following sequential steps:
Single trauma or cumulative microtrauma causes subfailure injury of the spinal ligaments
and injury to the mechanoreceptors embedded in the ligaments.When the injured spine performs a task or it is challenged by an external load, the transducer
signals generated by the mechanoreceptors are corrupted.Neuromuscular control unit has difficulty in interpreting the corrupted transducer signals because
there is spatial and temporal mismatch between the normally expected and the corrupted signals received.The muscle response pattern generated by the neuromuscular control unit is corrupted, affecting
the spatial and temporal coordination and activation of each spinal muscle.The corrupted muscle response pattern leads to corrupted feedback to the control unit via tendon
organs of muscles and injured mechanoreceptors, further corrupting the muscle response pattern.The corrupted muscle response pattern produces high stresses and strains in spinal components
leading to further subfailure injury of the spinal ligaments, mechanoreceptors and muscles, and
overload of facet joints.The abnormal stresses and strains produce inflammation of spinal tissues, which have abundant
supply of nociceptive sensors and neural structures.Consequently, over time, chronic back pain may develop.
The subfailure injury of the spinal ligament is defined as an injury caused by stretching of the tissue beyond its physiological limit, but less than its failure point. [48]
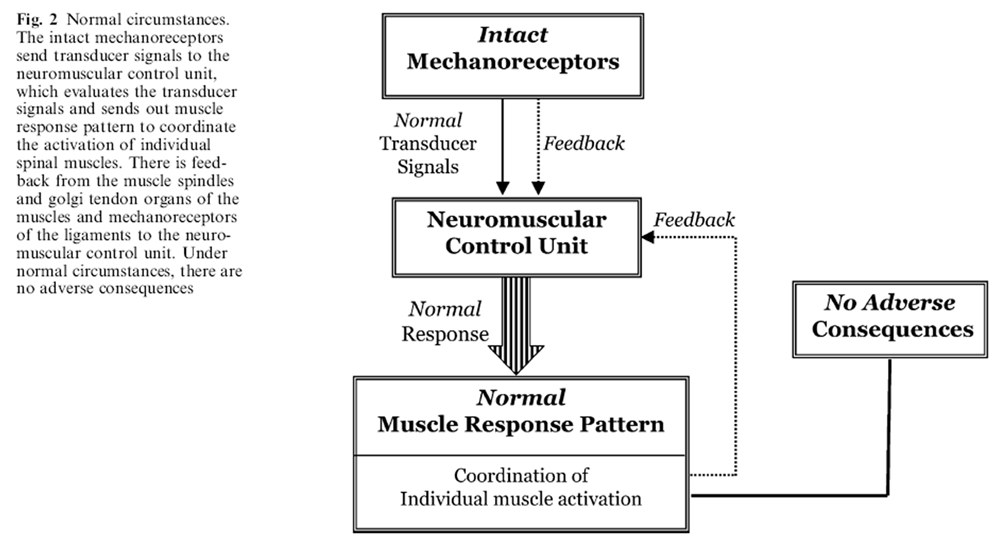
Figure 2 Under normal circumstances, to perform a task or to respond to an external challenge, the mechanoreceptors generate a complex and redundant set of transducer signals describing vertebral position, spinal motion, spinal load, and so forth, at each spinal level (Figure 2). The signals are transmitted to the neuromuscular control unit for interpretation and action. The neuromuscular control unit evaluates the signals and produces a normal muscle response pattern, based upon several factors, including the need for spinal stability, postural control, balance, minimal stress/stain in various spinal components, and so forth. This is achieved via feedback from the muscle spindles and golgi tendon organs of the muscles as well as the mechanoreceptors of the ligaments. The muscle response pattern includes all the information needed to dynamically orchestrate the muscles: to choose the individual muscles needed, and to activate each muscle in a defined sequence with respect to its onset, activation level and shut-off. The entire dynamic procedure is relatively quick, non-injurious and leads to no adverse consequences.
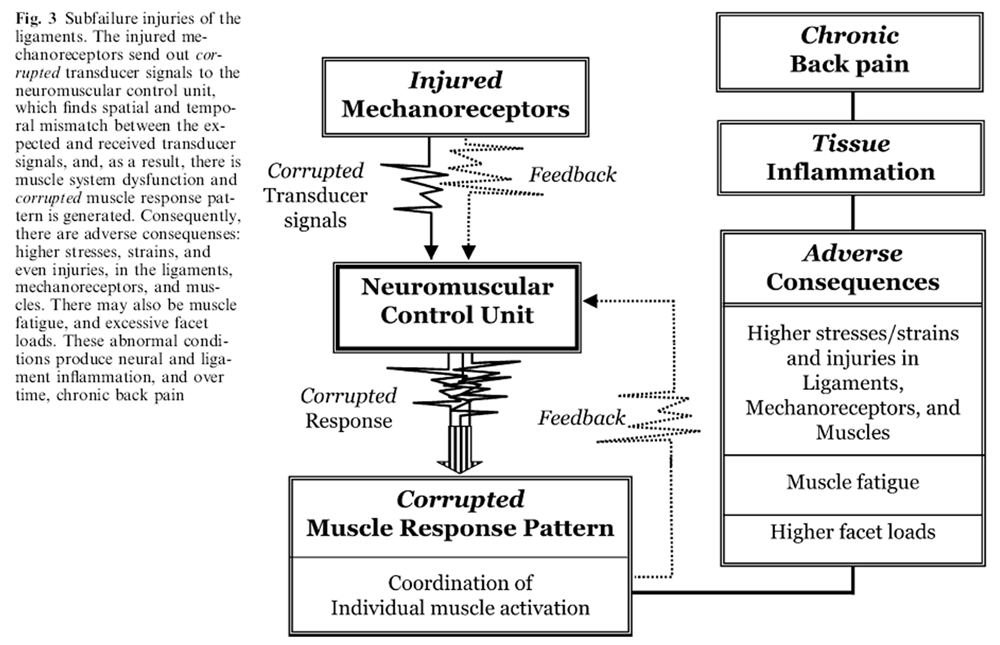
Figure 3 The injured spine behaves differently (Figure 3). The subfailure injuries of the ligaments disrupt and/or injure the embedded mechanoreceptors. When the spine performs a routine task or responds to an external challenge, the disrupted/injured mechanoreceptors produce corrupted transducer signals, describing vertebral position, motion, spinal loads etc. for each spinal level. There is loss of spatial and temporal integrity of the transducer signals received from multiple redundant mechanoreceptors distributed through the spinal column. The neuromuscular control unit, not affected by the injury itself, senses a mismatch between the normally expected and the received transducer signals, and, therefore, has difficulty in choosing the appropriate muscle response pattern. However, it must act.
Consequently, the neuromuscular control unit produces a corrupted muscle response pattern, which is the closest match it can determine to the corrupted transducer signals. The corrupted muscle response pattern affects the choice of the spinal muscles to activate, and the individual muscle activation: force onset, intensity and shut-off. The orchestration of the various spinal muscles responsible for spinal stability, posture and motion is disrupted. Additionally, the feedback to the neuromuscular control unit and mechanoreceptors is also negatively affected, further corrupting the muscle response pattern.
This has several adverse effects. Higher stresses, and strains and injuries may develop in the spinal ligaments, and mechanoreceptors. The facet joints may be overloaded, and the spinal muscles may fatigue or be injured. Over time, these injurious stresses and strains can initiate inflammation of neural tissues [12], and accelerate disc [40] and facet joint [9] degeneration. Thus, a vicious cycle is set up, leading to chronic dysfunction of the entire spinal system, resulting in back pain.
Discussion
The underlying concept of the spinal instability hypothesis was the need for adequate spinal stability provided by vertebrae and ligaments of the spinal column, and augmented by the spinal muscles under the neuromuscular control. [46, 47] In the present hypothesis, the focus is on the disruption of the mechanoreceptors due to ligament injury leading to corrupted transducer signals and muscle response pattern, and overall system dysfunction. What follows is an attempt, using the new hypothesis, to explain some of the observations concerning low back and neck pain patients, and to suggest treatment options.
Delayed muscle response is a common observation in low back pain patients. When low back pain patients were challenged by a sudden external load, the delayed muscle onset was observed [35], and delayed muscle shut-off was seen when the load was removed. [52] Similarly, the anticipatory response of the transverse abdominis was delayed. [20] These findings can be explained by the hypothesis. An individual with intact spinal system, when challenged by a sudden change in its load or posture, will produce a quick and normal muscle response pattern, specific to the challenge (Fig. 2).
However, when the neuromuscular control unit receives corrupted transducer signals, it may take a longer time to choose a muscle response pattern that most closely matches the corrupted transducer signals, taking into account a multitude of factors such as spinal stability, postural balance, tissue overload and so forth (Fig. 3). Additional factors, such as muscle fatigue, complexity of the task, mental distraction, and so forth, may further decrease the efficiency of the neuromuscular control unit leading to the delayed muscle system response.
Balance and postural control are deficient in low back pain patients. [10, 33, 53] The balance and postural control includes a three-step process: generation of transducer signals by the mechanoreceptors; selection of appropriate muscle response pattern by the neuromuscular control unit based up mechanoreceptor signals; and feedback from the mechanoreceptors and muscle spindles and golgi tendon organs (Fig. 2). Therefore, subfailure injuries of the ligaments disrupt all the three steps involving the mechanoreceptors thereby resulting in poor balance and postural control.
Re-positioning error has been consistently found in both low back pain [7, 38, 42] and whiplash [19, 31] patients. The error occurs when the patient is asked, starting from an initial posture, to first bend or twist the spine to a certain posture, and then to return to the initial posture. Based upon the hypothesis presented, this is to be expected. The muscle response pattern generated to bring back the trunk or head to the initial posture makes use of the mechanoreceptor transducer signals, in the three-step process described above. With the ligament injury in back pain patients, the corrupted mechanoreceptor information and the corrupted muscle response pattern will both lead to the re-positioning error.
Among chronic whiplash patients, decreased neck motion has been observed in most studies [2, 4, 14, 34, 49, 51]. These were active motion studies in which the subject was encouraged to produce the motion. However, when the subject was relaxed and the motion was produced passively by the examiner, the motion was found to be increased in the whiplash patients compared to the control group. [27] How can one explain these contrasting findings? In the active motion studies, corrupted muscle response pattern (generated due to corrupted mechanoreceptor signals) applies higher muscle forces on the cervical spine. Such forces stiffen the spine and reduce the motion [50, 68, 74]. In the relaxed passive motion studies, care was taken to decrease the influence of muscle guarding, pain and lack of motivation by relaxing the neck and shoulder muscles with application of vapor coolant, and then letting the examiner move the patient’s head into maximum flexion. Thus, when the abnormal muscle forces were minimized in the passive examination, the intrinsic injury of the spinal column was exhibited as the increased motion.
Muscle spasm is commonly observed in both low back pain [5, 30] and whiplash patients [39, 55, 67]. Muscle coordination may be thought of as an orchestrated activation of various spinal muscles to stabilize the spinal column and accomplish a certain task. The orchestration consists of activation of individual muscles with respect to the onset, magnitude of the force generated, and offset. With the injury of the ligaments, the mechanoreceptors generate corrupted transducer signals, and therefore, there is a mismatch between the expected and the received corrupted transducer signals. The neuromuscular control unit senses the mismatch and may fire simultaneously both the agonist and antagonist muscles at its command to temporarily stabilize the spine and minimize the intervertebral motions, corrupted transducer signals, and pain. If the situation does not improve with time, then the muscle action may become chronic. Such simultaneous firing of agonistic and antagonist muscles has been observed in low back pain patients.
Greater variability has been observed in almost all parameters measured in low back [28, 33, 37, 42, 53] and whiplash [14, 34] patients. The new hypothesis can explain this increased variability. The subfailure injuries of ligaments are incomplete injuries, which may range between tearing of a few fibers to a nearly complete rupture of a ligament. Importantly, a complex joint, such as a functional spinal unit, includes many ligament structures. This collection of ligament structures may encompass a wide range of injuries, each structure with different injury severity, depending upon the magnitude and mode of the trauma. The density of the mechanoreceptors imbedded in the various ligament structures may also vary. The result of all these numerous variations can produce a wide spectrum of corrupted muscle response patterns for seemingly similar injury-causing events. Further, each low back pain patient is unique, for example with respect to the anatomy, mechanical properties of ligaments, and muscle response to the trauma, adding further to the muscle response pattern variability.
There are limitations to the hypothesis. Back pain is a complex multifactorial problem, and a single hypothesis cannot explain each and every clinical and research observation, and there may also be alternative explanations, such as instability [46, 47], and/or pain [32, 54]. It is recognized that the pain is a subjective experience. Besides affecting the muscle system via the corrupted mechanoreceptor signals, ligament injury may also result in muscle atrophy and weakness due to disuse, thus directly affecting the spinal system function. Additionally, muscle injury, fatigue, atrophy, and so forth may aggravate the spinal system dysfunction. As the muscles participate in the feedback loop via the mechanoreceptors in the form of muscle spindles and golgi tendon organs (Fig. 3), their disruption could further corrupt the muscle response pattern. However, an injured muscle may heal relatively quickly due to abundant blood supply, and, therefore, may not be the main cause of chronic back pain. In contrast, the ligament injuries heal poorly and, therefore, may lead to tissue degeneration over time [40, 41].
Thus, the ligament injuries are more likely to be the major cause of the chronic back pain. The corrupted transducer signals may be the result not only of the ligament injury, but also due to ligament fatigue and viscoelastic creep stretch [61], but such an effect is often reversible given sufficient rest, and, therefore, may not always lead to chronic back pain. The clinical and research studies presented constitute only a small, but an important and quite representative sample, of the vast literature available on the subject of back pain. It is recognized that there may be other studies whose explanation may or may not fit the new hypothesis. In general, hypotheses and models are extremely difficult, if not impossible, to fully validate. [45] They can only attempt to explain the available findings, and may be used to predict outcomes in specific situations.
Can the system adapt to the subfailure injury of the mechanoreceptors? A minor subfailure injury is probably repaired or compensated with no long-term consequences. A mild subfailure injury, on the other hand, may be successfully compensated in the short-term by temporarily modifying the chosen muscle response pattern. However, the modification may be difficult to maintain overtime, as it is likely to produce excessive tissue loads and muscle fatigue. Lapses in the maintenance of the modified muscle response pattern may occur from time to time. Could this be the mechanism for recurrent episodes of back pain that many patients experience? [57, 71] On the other hand, if the corrupted muscle response pattern becomes permanent, then it may result in abnormal posture, disturbed intervertebral motion pattern, altered gait, and, in general, a less efficient system to perform every day spinal functions.
One can speculate as to the possible treatment options based upon the hypothesis. The incoming corrupted transducer data may never become normal, even though the ligaments, incorporating the injured mechanoreceptors, may heal/scar over time. After breaking the cascade of injury, inflammation, and pain by suitable drug treatment, the patient may be encouraged to re-train the neuromuscular control unit to produce an altered muscle response pattern that is suited to both the corrupted transducer signals and activities of daily living. The criterion for the altered muscle response pattern may be the reduction of stresses and strains of the ligaments, loads on facet joints, and muscle forces, which may reduce the back pain. A set of tasks may be designed for this purpose. The tasks may be repeated and varied. Improvement in the efficiency of the neuromuscular control unit may develop over time, with concomitant relief of back pain. Several clinical studies have incorporated these and similar ideas.
Re-training exercises involving muscle control have shown promising results in both chronic low back pain [22, 23, 70], and neck pain [56, 64, 66] patients, compared to traditional therapies. More research is needed in this area. I hope that the presentation of this hypothesis will stimulate discussion among clinicians and researchers in biomechanics to evaluate the usefulness of the hypothesis towards better understanding of back pain, development of more precise diagnostic methods, and design of more efficient treatments for back pain patients.
Conclusions
A new hypothesis of chronic back pain based upon muscle system dysfunction due to ligament injuries is described. Subfailure injuries of the ligaments and embedded mechanoreceptors generate corrupted mechanoreceptor signals. Consequently, the neuromuscular control unit produces corrupted muscle response pattern, resulting in excessive loading and, possibly, injuries of the spinal structures, including additional injuries of the mechanoreceptors. The hypothesis accounts for many of the common and important experimental observations and clinical findings seen in low back pain and whiplash patients. In the low back pain patients, it explains findings of delayed muscle response, poor balance, inefficient postural control, greater error in re-positioning the trunk, muscle spasm, and greater variability in the tasks performed. In the whiplash patients, both the decreased motion in active testing and increased motion in passive-relaxed testing are explained. The hypothesis proposes that the dysfunction of the muscle system over time may lead to chronic back pain via additional mechanoreceptor injury, and neural tissue inflammation.
Acknowledgements
I would like to express my sincere thanks to Elena Gimenez, Paul Ivancic, Kim Reid, and James Yue for their valuable advice, and assistance. I also want to thank the anonymous reviewers for their thoughtful suggestions to improve the manuscript. Further, I would like to acknowledge the grant support over many years from the National Institutes of Health.
References:
Andersson GB.
The epidemilogy of spinal disorders. 2nd edn.
Philadelphia: Lippincott-Raven; 1997.Antonaci F, Bulgheroni M, Ghirmai S, et al.
3D kinematic analysis and clinical evaluation of neck movements in patients with whiplash injury.
Cephalalgia. 2002;22:533–542Bigos SJ, Spengler DM, Martin NA, et al.
Back injuries in industry: a retrospective study. III. Employee-related factors.
Spine. 1986;11:252–256Bonelli A, Donati P, Maltoni G, et al.
Neck motion evaluation after whiplash: a radiographic and kinematic protocol.
Ital J Anat Embryol. 2000;105:51–62Borenstein DG, Korn S.
Efficacy of a low-dose regimen of cyclobenzaprine hydrochloride in acute skeletal muscle spasm: results of two placebo-controlled trials.
Clin Ther. 2003;25:1056–1073Brown MF, Hukkanen MV, McCarthy ID, et al.
Sensory and sympathetic innervation of the vertebral endplate in patients with degenerative disc disease.
J Bone Joint Surg Br. 1997;79:147–153Brumagne S, Cordo P, Lysens R, et al.
The role of paraspinal muscle spindles in lumbosacral position sense in individuals with and without low back pain.
Spine. 2000;25:989–994Burke JG, Watson RW, McCormack D, et al.
Intervertebral discs which cause low back pain secrete high levels of proinflammatory mediators.
J Bone Joint Surg Br. 2002;84:196–201Butler D, Trafimow JH, Andersson GB, et al.
Discs degenerate before facets.
Spine. 1990;15:111–113Byl NN, Sinnott PL.
Variations in balance and body sway in middle-aged adults: subjects with healthy backs compared with subjects with low-back dysfunction.
Spine. 1991;16:325–330Cavanaugh Mechanisms of low back. 1997;pain:a.
Cornefjord M, Olmarker K, Otani K, et al.
Nucleus pulposus-induced nerve root injury: effects of diclofenac and ketoprofen.
Eur Spine J. 2002;11:57–61Farfan HF, Sullivan JD.
The relation of facet orientation to intervertebral disc failure.
Can J Surg. 1967;10:179–185Feipel V, Rondelet B, LePallec JP, et al.
The use of disharmonic motion curves in problems of the cervical spine.
Int Orthop. 1999;23:205–209Freemont AJ, Peacock TE, Goupille P, et al.
Nerve ingrowth into diseased intervertebral disc in chronic back pain.
Lancet. 1997;350:178–181Freemont AJ, Watkins A, Le Maitre C, et al.
Nerve growth factor expression and innervation of the painful intervertebral disc.
J Pathol. 2002;197:286–292Frymoyer JW, Cats-Baril WL.
An overview of the incidences and costs of low back pain.
Orthop Clin North Am. 1991;22:263–271Fujiwara A, Tamai K, An HS, et al.
The relationship between disc degeneration, facet joint osteoarthritis, and stability of the degenerative lumbar spine.
J Spinal Disord. 2000;13:444–450Heikkila H, Astrom PG.
Cervicocephalic kinesthetic sensibility in patients with whiplash injury.
Scand J Rehabil Med. 1996;28:133–138Hodges PW, Richardson CA.
Inefficient muscular stabilization of the lumbar spine associated with low back pain. A motor control evaluation of transversus abdominis.
Spine. 1996;21:2640–2650Indahl A, Kaigle AM, Reikeras O, et al.
Interaction between the porcine lumbar intervertebral disc, zygapophysial joints, and paraspinal muscles.
Spine. 1997;22:2834–2840Jull GA, Richardson CA.
Motor control problems in patients with spinal pain: a new direction for therapeutic exercise.
J Manipulative Physiol Ther. 2000;23:115–117Kankaanpaa M, Taimela S, Airaksinen O, et al.
The efficacy of active rehabilitation in chronic low back pain. Effect on pain intensity, self-experienced disability, and lumbar fatigability.
Spine. 1999;24:1034–1042Kelsey JL, White AA.,
III Epidemiology and impact of low-back pain.
Spine. 1980;5:133–142Kirkaldy-Willis WH, Wedge JH, Yong-Hing K, et al.
Pathology and pathogenesis of lumbar spondylosis and stenosis.
Spine. 1978;3:319–328Kojima Y, Maeda T, Arai R, et al.
Nerve supply to the posterior longitudinal ligament and the intervertebral disc of the rat vertebral column as studied by acetylcholinesterase histochemistry. I. Distribution in the lumbar region.
J Anat. 1990;169:237–246Kristjansson E, Leivseth G, Brinckmann P, et al.
Increased sagittal plane segmental motion in the lower cervical spine in women with chronic whiplash-associated disorders, Grades I-II: a case-control study using a new measurement protocol.
Spine. 2003;28:2215–2221Lariviere C, Gagnon D, Loisel P.
The comparison of trunk muscles EMG activation between subjects with and without chronic low back pain during flexion-extension and lateral bending tasks.
J Electromyogr Kinesiol. 2000;10:79–91Lipson SJ, Fox DA, Sosman JL.
Symptomatic intravertebral disc herniation (Schmorl’s node) in the cervical spine.
Ann Rheum Dis. 1985;44:857–859Long DM, BenDebba M, Torgerson WS, et al.
Persistent back pain and sciatica in the United States: patient characteristics.
J Spinal Disord. 1996;9:40–58Loudon JK, Ruhl M, Field E.
Ability to reproduce head position after whiplash injury.
Spine. 1997;22:865–868Lund JP, Donga R, Widmer CG, et al.
The pain-adaptation model: a discussion of the relationship between chronic musculoskeletal pain and motor activity.
Can J Physiol Pharmacol. 1991;69:683–694Luoto S, Aalto H, Taimela S, et al.
One-footed and externally disturbed two-footed postural control in patients with chronic low back pain and healthy control subjects. A controlled study with follow-up (discussion 9–90)
Spine. 1998;23:2081–2089Madeleine P, Prietzel H, Svarrer H, et al.
Quantitative posturography in altered sensory conditions: a way to assess balance instability in patients with chronic whiplash injury.
Arch Phys Med Rehabil. 2004;85:432–438Magnusson ML, Aleksiev A, Wilder DG, et al.
European Spine Society–the AcroMed Prize for Spinal Research 1995. Unexpected load and asymmetric posture as etiologic factors in low back pain.
Eur Spine J. 1996;5:23–35McLain RF.
Mechanoreceptor endings in human cervical facet joints.
Spine. 1994;19:495–501Newcomer KL, Jacobson TD, Gabriel DA, et al.
Muscle activation patterns in subjects with and without low back pain.
Arch Phys Med Rehabil. 2002;83:816–821Newcomer KL, Laskowski ER, Yu B, et al.
Differences in repositioning error among patients with low back pain compared with control subjects.
Spine. 2000;25:2488–2493Norris SH, Watt I.
The prognosis of neck injuries resulting from rear-end vehicle collisions.
J Bone Joint Surg Br. 1983;65:608–611Osti OL, Vernon-Roberts B, Fraser RD.
1990 Volvo Award in experimental studies. Anulus tears and intervertebral disc degeneration. An experimental study using an animal model.
Spine. 1990;15:762–767Osti OL, Vernon-Roberts B, Moore R, et al.
Annular tears and disc degeneration in the lumbar spine. A post-mortem study of 135 discs.
J Bone Joint Surg Br. 1992;74:678–682O’Sullivan PB, Burnett A, Floyd AN, et al.
Lumbar repositioning deficit in a specific low back pain population.
Spine. 2003;28:1074–1079Oxland TR, Crisco JJ, III, Panjabi MM, et al.
The effect of injury on rotational coupling at the lumbosacral joint. A biomechanical investigation.
Spine. 1992;17:74–80Palmgren T, Gronblad M, Virri J, et al.
Immunohistochemical demonstration of sensory and autonomic nerve terminals in herniated lumbar disc tissue.
Spine. 1996;21:1301–1306Panjabi M.
Validation of mathematical models.
J Biomech. 1979;12:238Panjabi MM.
The stabilizing system of the spine. Part I. Function, dysfunction, adaptation, and enhancement (discussion 97)
J Spinal Disord. 1992;5:383–389Panjabi MM.
The stabilizing system of the spine. Part II. Neutral zone and instability hypothesis (discussion 7)
J Spinal Disord. 1992;5:390–396Panjabi MM, Yoldas E, Oxland TR, et al.
Subfailure injury of the rabbit anterior cruciate ligament.
J Orthop Res. 1996;14:216–222Patijn J, Wilmink J, ter Linden FH, et al.
CT study of craniovertebral rotation in whiplash injury.
Eur Spine J. 2001;10:38–43Patwardhan AG, Havey RM, Ghanayem AJ, et al.
Load-carrying capacity of the human cervical spine in compression is increased under a follower load.
Spine. 2000;25:1548–1554Puglisi F, Ridi R, Cecchi F, et al.
Segmental vertebral motion in the assessment of neck range of motion in whiplash patients.
Int J Legal Med. 2004;118:235–239Radebold A, Cholewicki J, Panjabi MM, et al.
Muscle response pattern to sudden trunk loading in healthy individuals and in patients with chronic low back pain.
Spine. 2000;25:947–954Radebold A, Cholewicki J, Polzhofer GK, et al.
Impaired postural control of the lumbar spine is associated with delayed muscle response times in patients with chronic idiopathic low back pain.
Spine. 2001;26:724–730Roland MO.
A critical review of the evidence for a pain-spasm-pain cycle in spinal disorders.
Clin Biomech (Bristol, Avon) 1986;1:102–109Ronnen HR, Korte PJ, Brink PR, et al.
Acute whiplash injury: is there a role for MR imaging?–a prospective study of 100 patients.
Radiology. 1996;201:93–96Rosenfeld M, Seferiadis A, Carlsson J, et al.
Active intervention in patients with whiplash-associated disorders improves long-term prognosis: a randomized controlled clinical trial.
Spine. 2003;28:2491–2498Salminen JJ, Erkintalo MO, Pentti J, et al.
Recurrent low back pain and early disc degeneration in the young.
Spine. 1999;24:1316–1321Sekine M, Yamashita T, Takebayashi T, et al.
Mechanosensitive afferent units in the lumbar posterior longitudinal ligament.
Spine. 2001;26:1516–1521Solomonow M, Zhou B, Baratta RV, et al.
Neuromuscular disorders associated with static lumbar flexion: a feline model.
J Electromyogr Kinesiol. 2002;12:81–90Solomonow M, Zhou BH, Baratta RV, et al.
Biomechanics and electromyography of a cumulative lumbar disorder: response to static flexion.
Clin Biomech (Bristol, Avon) 2003;18:890–898Solomonow M, Zhou BH, Baratta RV, et al.
Biomechanics of increased exposure to lumbar injury caused by cyclic loading: Part 1. Loss of reflexive muscular stabilization.
Spine. 1999;24:2426–2434Solomonow M, Zhou BH, Harris M, et al.
The ligamento-muscular stabilizing system of the spine.
Spine. 1998;23:2552–2562Spitzer WO, Skovron ML, Salmi LR, Cassidy JD, Duranceau J, Suissa S, Zeiss E.
Scientific Monograph of the Quebec Task Force on Whiplash-Associated Disorders
Redefining Whiplash and its Management
Spine (Phila Pa 1976). 1995 (Apr 15); 20 (8 Suppl): S1-S73Taimela S, Diederich C, Hubsch M, et al.
The role of physical exercise and inactivity in pain recurrence and absenteeism from work after active outpatient rehabilitation for recurrent or chronic low back pain: a follow-up study.
Spine. 2000;25:1809–1816Taimela S, Osterman K, Alaranta H, et al.
Long psychomotor reaction time in patients with chronic low-back pain: preliminary report.
Arch Phys Med Rehabil. 1993;74:1161–1164Taimela S, Takala EP, Asklof T, et al.
Active treatment of chronic neck pain: a prospective randomized intervention.
Spine. 2000;25:1021–1027Tarsy D.
Comparison of acute- and delayed-onset posttraumatic cervical dystonia.
Mov Disord. 1998;13:481–485Tawackoli W, Marco R, Liebschner MA.
The effect of compressive axial preload on the flexibility of the thoracolumbar spine.
Spine. 2004;29:988–993Dieen JH, Selen LP, Cholewicki J.
Trunk muscle activation in low-back pain patients, an analysis of the literature.
J Electromyogr Kinesiol. 2003;13:333–351Vezina MJ, Hubley-Kozey CL.
Muscle activation in therapeutic exercises to improve trunk stability.
Arch Phys Med Rehabil. 2000;81:1370–1379Wasiak R, Pransky G, Verma S, et al.
Recurrence of low back pain: definition-sensitivity analysis using administrative data.
Spine. 2003;28:2283–2291White AA, III, Gordon SL.
Symposium on idiopathic low back pained.
St. Louis: C.V. Mosby; 1982.White AA, III, Panjabi MM.
Clinical Biomechanics of the Spine. 2nd edn.
Philadelphia: Lippincott; 1990.Wilke HJ, Wolf S, Claes LE, et al.
Stability increase of the lumbar spine with different muscle groups. A biomechanical in vitro study.
Spine. 1995;20:192–198Williams M, Solomonow M, Zhou BH, et al.
Multifidus spasms elicited by prolonged lumbar flexion.
Spine. 2000;25:2916–2924Yamashita T, Cavanaugh JM, el-Bohy AA, et al.
Mechanosensitive afferent units in the lumbar facet joint.
J Bone Joint Surg Am. 1990;72:865–870Yang KH, King AI.
Mechanism of facet load transmission as a hypothesis for low-back pain.
Spine. 1984;9:557–565
Return to SUBLUXATION
Return to ABOUT SPINAL ADJUSTING
Return to NON-MUSCULOSKELETAL DYSFUNCTION
Since 7-07-2018


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |