

Introducing the External Link Model for
Studying Spine Fixation and Misalignment:
Part 1 — Need, Rationale, and ApplicationsThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: J Manipulative Physiol Ther 2007 (Mar); 30 (3): 239–245 ~ FULL TEXT
OPEN ACCESS Charles N.R. Henderson, DC, PhD, Gregory D. Cramer, DC, PhD,
Qiang Zhang, MD, DC, James W. DeVocht, DC, PhD, Jaeson T. Fournier, DC, MPH
Charles N.R. Henderson, DC, PhD,
Palmer Center for Chiropractic Research,
741 Brady, Davenport, IA 52803.
The Abstract: This is the first article in a series introducing a new animal model, the External Link Model that we propose will allow researchers to produce and study spine lesions with the cardinal biomechanical features of the chiropractic subluxation: fixation (hypomobility) and misalignment.
From the FULL TEXT Article
Introduction
This is the first article of a series that introduces a novel animal model, the External Link Model (ELM), that we propose will allow researchers to produce and study an experimental mimic of the chiropractic subluxation. A mimic of the chiropractic subluxation would permit a rigorous examination of the theoretical construct that provides a primary rationale for spinal manipulation and defines the profession in state and federal statutes. [1–3] The term subluxation is currently the focus of a heated debate in the chiropractic profession. Subluxation is a revered historical icon to many in the chiropractic profession and an anachronistic obstacle to others. [3–5] After a century of philosophical commentary and entrepreneurial explanation, many within the chiropractic profession are convinced that subluxation has progressed from theoretical construct to fact. However, there is still no validated criterion for identifying a subluxation. [6]
The current debate over the relevance of subluxation, as well as its role in defining the chiropratic identity, enjoins examination of this foundational theoretical construct. [5, 6] In this series of articles, we take a ‘proof of concept’ approach. The subluxation is operationally defined by 2 cardinal biomechanical features that are common to its many definitions, fixation (hypomobility) and malposition. [7, 8] Consequently, fixation and malposition were induced experimentally using the ELM to produce a subluxation mimic. Because the ELM is a long-term survival model, with the rats studied for many months after experimental spine fixation, we believe that it may be used to study the putative chronic effects of subluxation as well as the effects of various therapeutic interventions.
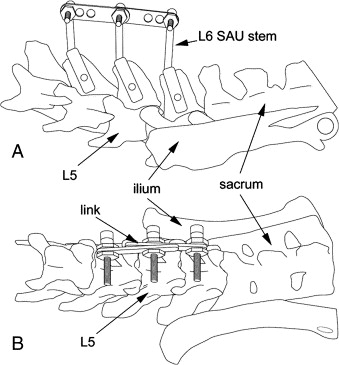
As the name suggests, spine fixation and misalignment are produced in the ELM by means of an external link system attached to spine segments (Fig 1, Fig 2). The external links may be removed after a variable link period without stressing the animal but allowing normal forces to be reintroduced to the motion segments.
This article explains the need for the model, its biomechanical rationale, and its current application in research related to chiropractic theory. Future articles will report the biomechanical characterization of the ELM, describe the implant procedure, list required materials, discuss new refinements, and present data on practicality issues such as failure rate, cost, ease of use, and reproducibility.
Figure 1. This figure shows the ELM with
3 link configurations.
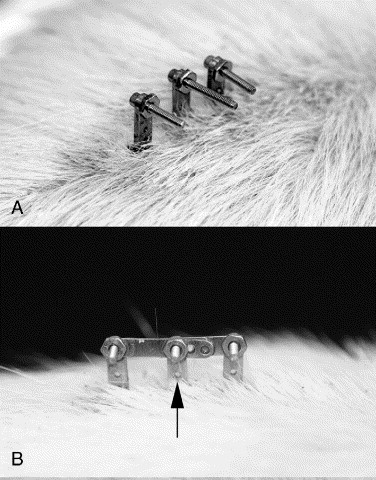
Figure 2. Diagram of SAUs linked in neutral
on the rat spine.
Discussion
The Need for a Model
After the first federally subsidized scientific workshop on spinal manipulation, The Research Status of Spinal Manipulative Therapy (1975), [9] General Chairman Murray Goldstein commented, “The lack of a relevant and reproducible animal model may be one important obstacle to clarification of these issues. …Thus, subluxation remains a hypothesis yet to be evaluated experimentally.” Unfortunately, this ‘important obstacle’ remained in place for more than a quarter century following this conference.
In 1996, an historic chiropractic conference was convened. The National Workshop to Develop the Chiropractic Research Agenda was funded through a contract between the US Health Resources and Services Administration Bureau of Health Professions and Palmer College of Chiropractic. [10] Workgroups reported on the current status of chiropractic clinical, educational, outcomes, basic, and health services research. The basic research workgroup report contained these comments: “Studies to address these kinds of questions [autonomic effects of adjustment] are hampered by the absence of a reliable animal model.” and “If the subluxation consists, at least in part, of fixation of the Z-joints [zygapophysial joints], then an animal model that mimics such fixation is clearly needed.” [11]
Chiropractic researchers have long appreciated the need for an animal model of subluxation because such a model would permit evaluation of the effects predicted by the widely held conceptual model of this lesion. [12] In addition, a number of important clinical questions might be directly evaluated with an animal model. One such question is, “Is there a ‘time window’ when spinal manipulation is most effective?” Others include, “What are the effects of different chiropractic therapeutic techniques given various states of the subject?” “What is the effect of repeated manipulation with differing states of the subject?” “Can subluxation alter visceral function?”
Studies conducted with animal models must be followed by human studies; however, without baseline biological data provided by animal models, expensive clinical trials are difficult to justify to the institutional review boards that serve as gatekeepers to all clinical research activities. [13] More important, the absence of basic research evidence often prompts the larger scientific and health care communities to treat lightly the results of clinical studies. This point was succinctly made by White and Panjabi [14]: “The concept of chiropractic subluxation remains a hypothesis yet to be evaluated experimentally. We believe that this has been one of the most frustrating aspects of certain views of the pathology that is purported to be altered with spinal manipulative therapy. When one is correcting a ‘subluxation’ that cannot be perceived by independent scientific observers, it is difficult to convince those observers that the treatment is effective.”
This is particularly true for studies examining chiropractic treatment of visceral complaints. In his review of animal models used in subluxation research, Vernon [15] noted the great need for basic science research if the chiropractic profession wants to persuade the larger health care community that chiropractic manipulation has a place in the treatment of nonmusculoskeletal conditions. He stated [15]:
It is my opinion that dozens of clinical trials can occur in such areas as the chiropractic treatment of asthma, dysmenorrhea, hypertension, etc, but they will all be relegated to insignificance and be dismissed as large-scale exercises in the placebo effect if a credible, valid biologic mechanism that links dysfunction in the spinal column with dysfunction in organ systems cannon be provided. This is the task that only the profession's basic scientists, in their pursuit of the scientific basis of subluxation, can accomplish.
Vernon presented 8 reasons for using animal models in research:(1) test theories derived from conceptual models;
(2) provide data to support clinical experience;
(3) high level of experimental control;
(4) prospective, therefore can explore cause and effect relationships;
(5) explore ‘treatment’ effects when lesion is reversed,
(6) explore physiologic components of subluxation;
(7) chronic experiments may allow for exploration of behavioral effects;
(8) animal studies are the ‘Holy Grail’ of clinical science.
Vernon stated that there are essentially 2 ways to model the spinal subluxation. First, investigators may create the obligate precipitating elements of the subluxation, vertebral fixation and misalignment, and monitor the effects, [15] “…this approach retains the highest fidelity to the natural circumstances and achieves the highest level of prospective validity, because, …the disorder was created first and its putative effects were studied thereafter.” Vernon also observed that this approach is by far the more difficult of the two, requiring a reproducible and biomechanically well-characterized model. He commented further that one or more ‘first-order’ effects (eg, nerve root compression or joint pain) must be shown by an animal model before ‘second-order’ effects (eg, changes in peripheral reflexes or nerve conduction velocities) can be studied and logically correlated with the theoretical subluxation model. He considered this to be a difficult task and noted that it was never really achieved in any of the studies he reviewed. The second approach Vernon suggested was to accept the proposition that vertebral misalignment creates nerve interference as presented above and simply begin the experiment by modeling the effects of intervertebral foramen encroachment or altered somatic afferent input (for Vernon—spinal joint pain). He stated a preference for this alternative approach because he considered it to be easier, both experimentally and logically, stating, [15] “When sufficient data have been gathered from studies like this, then investigators can return to the problem of whether the misalignment really produces the proximate effects."
The ELM introduced here takes the first approach, which Vernon asserted. We believe that the ELM will enable researchers to directly examine and develop subluxation theory, a foundational concept for much of chiropractic practice.
Previous Studies of the Subluxation with Intact Animal Models
There have been relatively few attempts to model the chiropractic subluxation. The earliest recorded attempt to produce a small animal model of the chiropractic subluxation was reported by Cleveland. [16] Cleveland described a transcutaneous spinous fixation system that was similar to that presented here. His spinous attachment device consisted of a compression clamp with a needle that pierced the spinous process of a vertebra (Fig 3). He reported 2 case studies in rabbits in which a misalignment of T12 was produced, [16] “…the 12th dorsal vertebra was subluxated posterior and right.” Cleveland observed numerous pathological and physiological changes over a 2–month period but did not report quantitative data and did not follow up with additional studies. After Cleveland, several attempts were made to produce a small animal model of the chiropractic subluxation. These have been reviewed by Vernon, [15] and more recently by Henderson. [12] It is ironic that the first model attempted in early subluxation research may offer the greatest promise as a research tool today. The ELM features a refined mechanical fixation device and implantation procedure, but the outstanding features of this intact animal model (minimal trauma, external fixation, and reversibility) are native to the idea first presented by Cleveland in 1965. [16]
Figure 3. Spinous attachment units
in the Cleveland model of spine fixation
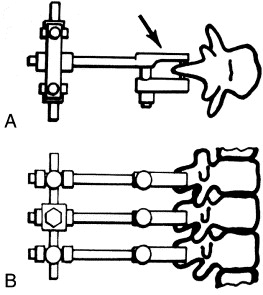
Why Use Rats?
The rat is arguably one of the most widely used animals in research. There are many reasons for this, some of which favored the rat as the subject for the ELM. The rat is inexpensive, readily available from laboratory animal breeders, and is easily handled and maintained. Several strains of rats (eg, Sprague-Dawley) are available with well-characterized genotypes, an important property for scientific study. Two additional attributes are especially valuable to the ELM. A recent systematic comparison of genomes across 13 vertebrates (including humans, chimps, cats, dogs, mice, rats, and chickens) reported that the rat genome is closer to the human than either dogs or cats, [17] “Analysis of transposable element insertions highlights the variation in genome dynamics among these species and confirms the placement of rodents as a sister group to the primates.” In addition, the rat is robust, demonstrating a high resistance to infection. The necessary transcutaneous nature of the model's external link system presents a potential channel for infection. Therefore, the rat's well-known resistance is a particularly valuable asset for this model. Finally, a very large body of research already exists in which the rat has been used to study joint anatomy, physiology, biochemistry, pathology, and biomechanics. [18–23] Using the rat in the ELM permits integration of this previous work.
One limitation imposed by using rats is that the rat spine does not permit ready fixation of the thoracic or cervical regions because the spinous processes in these areas are too small for attachment of our implant devices, spinous attachment units (SAUs). We originally considered using rabbits, thinking the rabbit spinous processes would be larger. However, we found that rabbit spinous processes were not much larger than those of large rats (450 g), and the caudal lumbar processes in rabbits were rounded, making SAU attachment more problematic. We also examined guinea pig spines, but these had only vestigial spinous processes and were not suitable for implanting SAUs.
Quadruped Versus Biped
It is widely accepted that quadruped spines are subjected to different loads than upright human spines. So, how can anything be learned about the biomechanics of the human, a biped, from studying rats, which are quadrupeds? This is an important question, and so, it will be considered from multiple perspectives.
First, numerous scientific articles actually counter the view that the biomechanical difference between the biped spine and the quadruped spine is so great. Both experimental and theoretical studies have shown striking similarities in the structure and mechanical loading of quadruped and biped spines. [24–28] In addition, even in the most disparate biological systems, fundamental features are conserved and may be studied. Rat organs and body systems are actually quite similar to humans; and, as noted previously, rats have well characterized genotypes that are surprisingly similar to humans. [29, 30] From these similarities, we have learned a great deal about fundamental human physiology.
There are also important differences between animals and humans; and investigators must be careful to interpret data with this in mind. It is recognized that many insights into human physiology were achieved precisely because researchers exploited these differences in the laboratory. The giant squid axon nicely illustrates this point. The anatomy and physiology of the giant squid is dramatically different from that of a human. These axons can be up to 1 mm in diameter, being 100 to 1000 times larger than mammalian axons! This remarkable size permitted researchers to attain the first intracellular recordings of action potentials from nerve cells and enabled the first experimental measurements of ionic currents; thereby revealing how the nervous system conveys information. The great size of the axon also permitted researchers to extrude the cytoplasm by pressing it out with a small roller, much like toothpaste in a tube, and measure its ionic composition. Researchers were subsequently able to replace the axoplasm with artificial solutions and observe resulting changes in membrane potential. Moreover, some giant squid nerve cells form very large synaptic contacts with other giant nerve cells, allowing extraordinary access to these important neurological structures. From this series of experiments, the fundamental principles of how the nervous system conveys and integrates information in all animals, including humans, were discovered. Hodgkin and Huxley were awarded the Nobel Prize in Medicine (1963) for their body of work, much of it done with the giant squid axon.
Finally, it must be acknowledged that, despite the biomechanical and physiological similarities highlighted above, researchers have noted that the differences between biped and quadruped animals may have functionally significant consequences. One of the most dramatic examples of this difference may be found in experimentally induced scoliosis. Machida et al [31] reported that pinealectomized biped rats, but not pinealectomized quadruped rats, develop scoliosis. Although normally quadruped, rats frequently engage in periods of bipedal posture. Machida and other researchers have induced longer periods of bipedal posture and locomotion by raising biped rats. In these rats, the forelimbs and tail were surgically amputated soon after birth, and food and water were kept in an elevated position to encourage bipedal activity. Apparently, some unique feature(s) of bipedal posture combined with melatonin deficiency produces scoliosis in rats. Machida's study and the work of several other investigators show that something about biped rats predisposes them to pathophysiological changes that are greater, or materially different, than those observed in quadruped rats. [32–34] So, returning to the question of what we can learn from the study of quadruped rats: we can learn a good deal about fundamental biomechanical and physiological mechanisms. However, because the chiropractic subluxation is a lesion attributed to perturbed spinal biomechanics, it is reasonable to expect that bipedal posture may also play an important role in some aspects of its pathophysiology. Consequently, we are currently examining the ELM with behaviorally induced biped rats. [35] In behaviorally induced biped rats, bipedal activity is encouraged by placing food and water in a progressively elevated position as the animals grow, but the forelimbs and tail are never amputated.
We believe that the ELM represents a powerful new tool for examining the mechanism and consequences of intervertebral fixation and misalignment, and, with the controlled application of specific treatment interventions, its subsequent treatment. In addition to the rat, this model may be applied in various animal species to benefit from the potentially significant differences between animals such as size, posture, and modes of locomotion.
Current and Future Applications of the ELM
The ELM has been used recently to study the degenerative effects [36] of spine fixation and misalignment as well as biomechanical, [37, 38] neurophysiological, [39] and behavioral consequences. Our recently published work on degenerative spine changes following experimental fixation showed one way this model may be used to explore the biological effects of subluxation. [36] We observed progressive degenerative changes in zygapophysial joints (Z-joints) of experimental animals compared with SAU bearing control animals that were never linked. As expected, Z-joint degeneration was greater for animals that had been experimentally linked for longer periods. However, although spontaneous improvement in Z-joint articular surface degeneration and osteophyte formation appeared to occur in some rats after the external links were removed, when present, this improvement was associated with a link-time threshold. Some rats linked for 1 to 4 weeks, and then observed after the links were removed for various periods, showed less articular surface degeneration. Similarly, osteophytes were less frequent and smaller in some rats linked for 4 to 8 weeks and then observed for weeks after link removal. However, it was noted that all rats linked for more than 4 weeks showed progressive articular surface degeneration, and all rats linked for more than 8 weeks developed more (or larger) osteophytes during the subsequent unlinked period.
The second article in this series will report changes in spine stiffness associated with various linked and unlinked periods. [38] The model shows changes in spine stiffness and intervertebral position that are consistent with subluxation theory and clinical chiropractic experience. Two interesting questions are currently under study: (1) How correlated are spine stiffness and degenerative Z-joint changes? and (2) Can therapeutic intervention stop or even reverse increased spine stiffness and/or degenerative spine changes? We are currently using the ELM to examine the putative treatment effects of low-velocity variable-amplitude (flexion-distraction mobilization) and instrument-assisted high velocity low-amplitude (thrust) spinal manipulation on spine biomechanics and degeneration, spinal cord neuropeptide production, and behavior. [35, 40]
We recently reported preliminary morphologic evidence drawn from this model that vertebral hypomobility can modify neural circuitry within the superficial dorsal horn of the spinal cord. [39] In a small pilot study, the number of synapses as well as symmetry and type of postsynaptic profile showed interesting trends when compared across experimentally fixed and SAU bearing control animals. Future studies are needed to assess the statistical and biological significance of these observations that should be highly relevant to contemporary chiropractic theory. [7]
The ELM is also being used to study behavioral changes in the rat associated with spine subluxation and subsequent spinal manipulation treatment. In our laboratories, we use a form of open-field behavior testing. Open-field testing is a frequently used behavioral assessment that is responsive to a wide spectrum of physical and emotional stressors in animals and has been extensively studied in rats. [41–43] In epidemiological terms, open-field behavior is an ‘animal-centered’ outcome, analogous to patient-centered outcomes widely used in contemporary clinical trials. [44] We consider this animal-centered outcome to be a nonspecific indicator of quality of life or well being for the rat. Data collection has recently been completed on a large ELM study (n = 90), in which such behavioral data were collected. The results of this behavior study will be reported in future publications.
Lastly, the ELM may be used to study visceral changes associated with spine subluxation. A tissue bank has been established at the Palmer College of Chiropractic, which contains organs harvested from both control and spine-fixated animals. These tissues may be made available to researchers interested in studying the visceral effects of spine subluxation.
Because the ELM is a long-term survival model, we believe it may be used to study the putative chronic effects of spine fixation and misalignment as well as the effects of various therapeutic interventions. Consequently, this model should enable basic science researchers to directly examine and develop subluxation theory, a foundational concept for much of chiropractic practice.
Practical Applications
The ELM has been used recently to study the degenerative effects of spine fixation and misalignment as well as biomechanical, neurophysiological, and behavioral consequences.
Because the ELM is a long-term survival model, it may be used to study the putative chronic effects of spine fixation and misalignment as well as the effects of various therapeutic interventions.
References:
Council on Chiropractic Practice.
Vertebral Subluxation in Chiropractic Practice
Council on Chiropractic Practice, Chandler (Ariz); 2003Hendrickson, RM.
Chiropractic Science and Practice in the United States:
A Report to the White House Commission on Complementary and Alternative Medicine Policy
on Behalf of the International Chiropractors Association
[monograph on the Internet].
The International Chiropractors Association, 2001 Feb. 14.
([cited 2006 Aug. 18]. Available from:)
www.chiropractic.org/pdf/commission_report.pdfMcDonald W.P., Durkin K.F., Pfefer M.
How Chiropractors Think and Practice: The Survey of North American Chiropractors
Semin Integr Med 2004; 2: 92–98Bryner, P.
Isn't it time to abandon anachronist terminology?.
J Aust Chiropr Assoc. 1987; 17: 53–58Mootz, RD.
Theoretic models of subluxation.
in: MI Gatterman (Ed.) Foundations of chiropractic: subluxation. 2nd ed.
Elsevier Mosby, St. Louis (Mo); 2005: 227–244J Keating, K Charton, J Grod, S Perle, D Sikorski, J Winterstein
Subluxation: Dogma or Science?
Chiropractic & Osteopathy 2005 (Aug 10); 13: 17Henderson, CNR.
Three neurophysiologic theories on the chiropractic subluxation.
in: MI Gatterman (Ed.) Foundations of chiropractic: subluxation. 2nd ed.
Elsevier Mosby, St. Louis (Mo); 2005: 296–303Rosner, AL.
The Role of Subluxation in Chiropractic
Foundation for Chiropractic Education and Research,
Des Moines, IA; 1997Goldstein, M.
The research status of spinal manipulative therapy: NINCDS monograph no. 15.
U.S. Department of Health, Education, and Welfare, Bethesda; 1975Hawk, C, Meeker, WC, and Hansen, DT.
The national workshop to develop the chiropractic research agenda.
J Manipulative Physiol Ther. 1997; 20: 147–149Brennan, PC, Cramer, GD, Kirstukas, SJ, and Cullum, ME.
Basic science research in chiropractic: the state of the art and recommendations for a research agenda.
J Manipulative Physiol Ther. 1997; 20: 150–168Henderson, CNR.
Animal models in the study of subluxation and manipulation: 1964-2004.
in: MI Gatterman (Ed.) Foundations of chiropractic: subluxation. 2nd ed.
Mosby Yearbook Inc, St. Louis (Mo); 2005: 47–10352nd World Medical Association General Assembly.
World Medical Association Declaration of Helsinki:
Ethical principles for medical research involving human subjects (5th revision)
[monograph on the Internet]. World Medical Association Website; 2004 Sept. 10.
([cited 2006 Jan. 14]. Available from:)
http://www.wma.net/e/policy/b3.htmWhite, A and Panjabi, MM.
Clinical biomechanics of the spine. 2nd ed.
J.B. Lippincott Company, Philadelphia; 1990Vernon, H.
Basic scientific evidence for chiropractic subluxation.
in: MI Gatterman (Ed.) Foundations of chiropractic: subluxation.
Mosby, St. Louis (Mo); 1995: 35–55Cleveland, CS.
Researching the subluxation on the domestic rabbit: a pilot research program conducted at the Cleveland Chiropractic College, Kansas City, Missouri.
Int Rev Chiropr: Science Edition. 1965; 1: 1–23Thomas, JW, Touchman, JW, Blakesley, RW, Bouffard, GG, Beckstrom-Sternberg, SM, Margulies, EH et al.
Comparative analyses of multi-species sequences from targeted genomic regions.
Nature. 2003; 424: 788–793Colpaert, FC, De Witte, P, Maroli, AN, Awouters, F, Niemegeers, CJ, and Janssen, PA.
Self-administration of the analgesic suprofen in arthritic rats: evidence of Mycobacterium butyricum induced arthritis as an experimental model of chronic pain.
Life Sci. 1980; 27: 921–928De Castro, C, De Sutter, P, Gybels, J, and Van Hees, J.
Adjuvant-induced arthritis in rats: a possible animal model of chronic pain.
Pain. 1981; 10: 173–185Levine, JD, Clark, R, Devor, M, Helms, C, Moskowitz, MA, and Basbaum, AI.
Intraneuronal substance P contributes to the severity of experimental arthritis.
Science. 1984; 226: 547–549Budgell, B and Sato, A.
Somatoautonomic reflex regulation of sciatic nerve blood flow.
J Neuromusculoskel Syst. 1994; 2: 170–177Kim, YI, Sik, N, Han, JS, and Hong, SK.
Critical role of the capsaicin-sensitive nerve fibers in the development of the causalgic symptoms produced by transecting some but not all of the nerves innervating the rat tail.
J Neurosci. 1995; 15: 4133–4139Mantyh, PW, DeMaster, E, Malhotra, A, Ghilardi, JR, Rogers, SD, Mantyh, CR et al.
Receptor endocytosis and dendrite reshaping in spinal neurons after somatosensory stimulation.
Science. 1995; 268: 1629–1631Elliott, DM and Sarver, JJ.
Young investigator award winner: validation of the mouse and rat disc as mechanical models of the human lumbar disc.
Spine. 2004; 29: 713–722Smit, TH.
The use of a quadruped as an in vivo model for the study of the spine—biomechanical considerations.
Eur Spine J. 2002; 11: 137–144Kumar, N, Kukreti, S, Ishaque, M, Sengupta, DK, and Mulholland, RC.
Functional anatomy of the deer spine: an appropriate biomechanical model for the human spine.
Anat Rec. 2002; 266: 108–117Wilke, HJ, Kettler, A, and Claes, LE.
Are sheep spines a valid biomechanical model for human spines?.
Spine. 1997; 22: 2365–2374Wilke, HJ, Kettler, A, Wenger, KH, and Claes, LE.
Anatomy of the sheep spine and its comparison to the human spine.
Anat Rec. 1997; 247: 542–555Abbott, A.
Laboratory animals: the Renaissance rat.
Nature. 2004; 428: 464–466Gibbs, RA, Weinstock, GM, Metzker, ML, Muzny, DM, Sodergren, EJ et al.
Genome sequence of the Brown Norway rat yields insights into mammalian evolution.
Nature. 2004; 428: 493–521Machida, M, Murai, I, Miyashita, Y, Dubousset, J, Yamada, T, and Kimura, J.
Pathogenesis of idiopathic scoliosis: experimental study in rats.
Spine. 1999; 24: 1985–1989Tanaka, H, Kimura, Y, and Ujino, Y.
The experimental study of scoliosis in bipedal rat in lathyrism.
Arch Orthop Trauma Surg. 1982; 101: 1–27Gloobe, H and Nathan, H.
Osteophyte formation in experimental bipedal rats.
J Comp Pathol. 1973; 83: 133–141Yamada, K, Sakamoto, K, Ushikubo, S, and Sato, Y.
Study of intervertebral disc herniation in bipedal rats.
Tokushima J Exp Med. 1960; 7: 93–103Henderson, CNR, DeVocht, JW, and Cramer, GD.
Evaluating reversible spinal fixation in biped rats [database on the Internet].
National Institutes of Health Crisp Database;
National Center for Complementary and Alternative Medicine; 2006.
([cited 2006 Aug. 20]. Grant Number: 5R21AT000784-02 Available from:)
http://crisp.cit.nih.gov/Cramer G.D., Fournier J.T., Henderson C.N., Wolcott C.C.
Degenerative Changes Following Spinal Fixation in a Small Animal Model
J Manipulative Physiol Ther 2004 (Mar); 27 (3): 141–154Henderson, CNR, DeVocht, JW, Kirstukas, SJ, and Cramer, GD.
in: In vivo biomechanical assessment of a small animal model of the vertebral subluxation.
Foundation for Chiropractic Education and Research,
Des Moines (Iowa); 2000: 193–195Henderson, C.N., Cramer, G.D., Zhang, Q.,
DeVocht, J.W., and Fournier, J.T.
Introducing the External Link Model for Studying Spine Fixation and Misalignment:
Part 2, Biomechanical Features
J Manipulative Physiol Ther 2007 (May); 30 (4): 279–294Bakkum, BW, Hong, S-P, Cramer, GD, and Henderson, CNR.
Preliminary morphologic evidence that vertebral hypomobility induces synaptic plasticity in the spinal cord [database on the Internet].
Society for Neuroscience; 2005.
([cited 2006 Aug. 20]. Available from:)
http://sfn.scholarone.com/itin2005/Meeker, WC.
Mechanisms and effects of chiropractic manipulation [database on the Internet].
National Institutes of Health Crisp Database;
National Center for Complementary and Alternative Medicine; 2006.
([cited 2006 Aug. 20]. Grant Number: 5U19AT002006-03.
Available from:)
http://crisp.cit.nih.gov/Bradfield, JF, Schachtman, TR, McLaughlin, RM, and Steffen, EK.
Behavioral and physiologic effects of inapparent wound infection in rats.
Lab Anim Sci. 1992; 42: 572–578Genaro, G and Schmidek, WR.
The influence of handling and isolation postweaning on open field, exploratory and maternal behavior of female rats.
Physiol Behav. 2002; 75: 681–688Lastres-Becker, I, de Miguel, RDPL, Makriyannis, A, Di Marzo, V, and Fernandez-Ruiz, J.
Compounds acting at the endocannabinoid and/or endovanilloid systems reduce hyperkinesia in a rat model of Huntington's disease.
J Neurochem. 2003; 84: 1097–1109Kaplan, RM, Ganiats, TM, Sieber, W, and Anderson, JP.
The quality of well-being scale.
Med Outcomes Trust Bull. 1996; 4: 1–4
Return to SUBLUXATION DEGENERATION
Since 5–08–2007


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |