

Neck-tongue Syndrome This section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: J Manipulative Physiol Ther 2004 (Jun); 27 (5): E 8 ~ FULL TEXT
OPEN ACCESS Cameron Borody, BSc, DC
Assistant Professor,
Clinical Education,
Canadian Memorial Chiropractic College,
1900 Bayview Avenue,
Toronto, Ontario M4G 3E6, Canada.OBJECTIVE: To discuss a case of neck-tongue syndrome (NTS) affecting a dancer/figure skater, review literature summarizing the pathogenesis and treatment, and offer new categorization of neck-tongue syndrome.
CLINICAL FEATURES: A 24ûyear-old female dancer/skater sought treatment for recurrent episodes of right-sided upper neck pain with associated ipsilateral numbness of her tongue following brisk active rotation. Radiographs revealed a narrowing of the left para-odontoid space. Physical examination revealed a mildly painful restriction in rotation at C1û2 with no apparent muscular hypertonicity.
INTERVENTION AND OUTCOME: The patient had sought chiropractic treatment for this condition several times since she was 8 years old. Diversified chiropractic adjustments were applied to restrictions throughout the cervical spine as determined by the clinician. No other interventions were employed. The patient experienced significant improvement in frequency and intensity of the neck and tongue symptoms following spinal manipulative therapy applied to her cervical spine.
CONCLUSION: There are 2 categories of NTS: complicated NTS due to the presence of an underlying disease process (inflammatory or degenerative) and uncomplicated NTS (idiopathic or trauma-related). This case report is of uncomplicated NTS that responded favorably to spinal manipulative therapy directed at the cervical spine. In the absence of upper cervical instability, spinal manipulative therapy appears to be beneficial and should be considered in all cases of uncomplicated NTS.
From the Full-Text Article:
Introduction
Neck-tongue syndrome (NTS) is defined as unilateral upper nuchal and/or occipital pain accompanied by ipsilateral numbness of the tongue. [1] The classic presentation of symptoms is brought on by a brisk, sudden axial rotation of the head. Neck and tongue symptoms will dissipate in approximately 60 seconds. [1] NTS has been reported over a wide age range (6û65 years). There is no clear sex or age predilection, although 10 of 45 cases have reported an onset prior to 20 years of age. [2, 3] Differential diagnoses for paresthesia in and around the mouth include carotid artery pathology and vertebrobasilar insufficiency, which are both contraindications for spinal manipulative therapy (SMT). [1, 4] A review of the relevant anatomy, mechanisms, and reported treatments will facilitate diagnosis and appropriate therapeutic intervention.
Case Report
A 24ûyear-old woman entered the clinic complaining of short episodes of upper neck pain. The recurrent episodes of right-sided neck pain began insidiously when she was 8 years old. She explained that the neck pain was ôsharpö or ôpiercingö and rated the intensity as a 7 on a 10ûpoint scale (10 = most intense pain). She related that the pain occurred when she briskly turned her head to the right. As an avid dancer/figure skater, the pain limited her ability to perform; although she was still physically able to dance, the intensity and unpredictability of the symptoms affected her concentration and expressiveness. The frequency of the complaint varied with her activity level. She had virtually no neck or tongue symptoms until she committed the aggravating head movement. The intensity of the neck and tongue symptoms was consistent from one episode to the next. There was no past medical history of trauma or inflammatory arthritide that could predispose her to upper cervical instability. The patient was a nonsmoker; did not report any headaches or vestibular or visual symptoms; and was otherwise in excellent health.
Her vital signs were normal. Assessment of the cervical ranges of motion (active and passive) did not reproduce the chief complaint. She reported that slow deliberate movement did not aggravate the condition and demonstrated that a quick glance over her right shoulder (ie, shoulder-checking while driving) elicited the neck pain. She said that her neck pain was usually accompanied by a sensation of ô7ûUp bubblingö on the right half of her tongue that lasted roughly 30 seconds despite moving her head back to neutral. A neurological examination was within normal limits. There were no positive orthopedic tests in the cervical spine. Motion and static palpation of the cervical spine and associated soft tissues revealed a mildly painful restriction in left rotation at C1û2 with no apparent muscular hypertonicity.
Radiographs were ordered (Figure 1, A through C) due to the longevity of the complaint and revealed a persistent narrowing of the left para-odontoid space. The radiology report stated that the asymmetry might be due to a right rotational malposition of atlas, possibly a compensatory response to a left rotational fixation of axis.
Figure 1.
A: Neutral and angulated anteroposterior open month
radiographs reveal a narrowed left para-odontoid space.
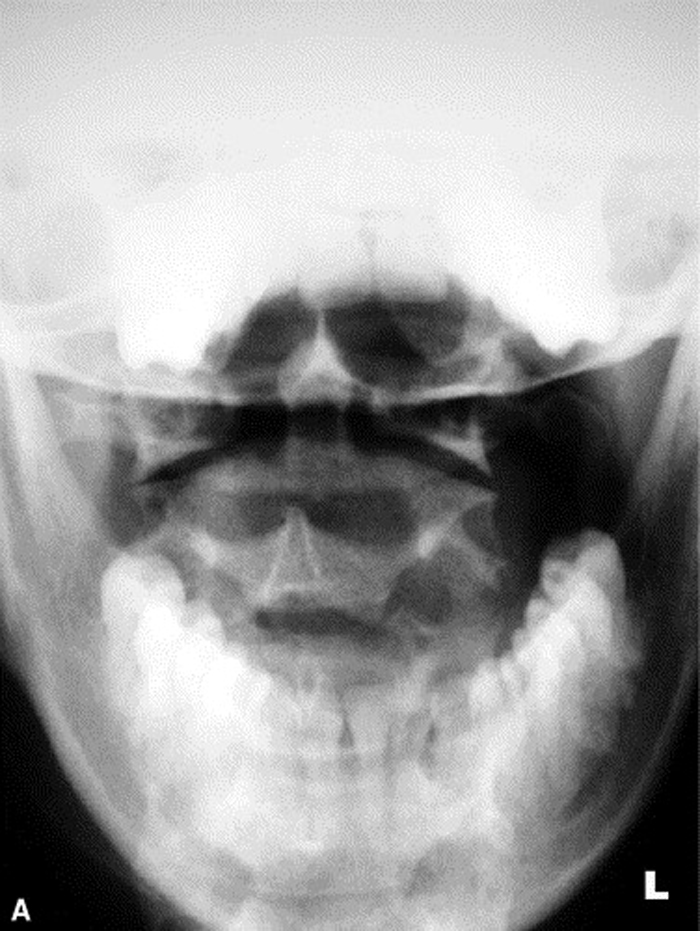
Figure 2.
B: Neutral and angulated anteroposterior open month
radiographs reveal a narrowed left para-odontoid space.
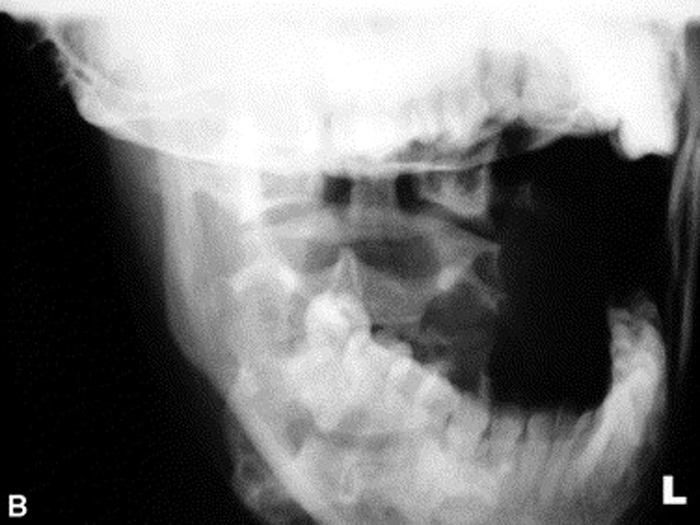
Figure 3.
C: Neutral and angulated anteroposterior open month
radiographs reveal a narrowed left para-odontoid space.

The patient was treated by a chiropractor (not the author) who applied diversified SMT to the restrictions throughout the cervical spine only. Other reported treatment options (cervical collar, exercises, medication) were not employed. The patient has sought treatment at various intervals since the onset of the symptoms and reported a significant reduction in the frequency and intensity of the symptoms, particularly the paresthesia felt in the tongue. Although she still experiences NTS, she describes its occurrence as ôrareö and is able to perform without pain.
Discussion:
The patient was diagnosed with neck-tongue syndrome. This is the third reported case of NTS surfacing during athletic activity. [1, 5]. Lance and Anthony [1] described a 15ûyear-old male whose neck and tongue symptoms followed a quick rotation of the head to the right or left while swimming, bowling, playing softball, or swinging on parallel bars. Webb et a [5] described a case of a 34ûyear-old male competitive swimmer whose neck and tongue symptoms had plagued him consistently throughout his swimming career. Turning his head to either side to breathe while swimming elicited pain and paresthesia severe enough to force him to stop racing. [5] No treatment was described for either case. [1, 5]. Various case reports suggest that symptoms may not be limited to the classic presentation and that patients with neck-tongue syndrome may describe any of the following associated symptoms: jaw pain, anterolateral neck pain, a sensation of choking, ôear pressure,ö radiation along the ipsilateral trapezius muscle to the acromioclavicular joint, paresthesia (ulnar distribution), dysarthria, nasal congestion, sialorrhea (excessive salivation), radiation to the vertex of the skull, and retroauricular numbness. [4û8]
Anatomy
There is no obvious anatomical connection between the upper cervical spine and the tongue. However, a review of the relevant anatomy helps explain how the two may be linked. The C2 nerve roots unite to form a short spinal nerve that splits into ventral and dorsal rami. The rami lie dorsal to the C1û2 facet joint deep to the inferior oblique muscle, intimately associated with both the joint capsule and muscle via strong fascia. The most important detail is that the C2 ventral ramus, which innervates the C1û2 facet joint, arches over the vertebral artery and joins the cervical plexus. [7] Somatic sensory afferents to the anterior two thirds of the tongue originate from the lingual nerve, which arises from the posterior division of the mandibular nerve (third branch of cranial nerve V) [9] (See Figure 2)
Mechanism
Two main theories exist which attempt to explain the pathogenesis of neck-tongue syndrome:
Subluxation/compression
Bogduk [7] suggests that a temporary incongruency of the C1û2 facet joint on sudden axial rotation of the head stretches the joint capsule causing local pain in the neck. Subsequent mechanical irritation of the closely associated C2 ventral ramus results in paresthesia of the ipsilateral half of the tongue. [7] This theory is supported by experimental findings that suggest that the hypoglossal nerve contains more than just motor efferents to the intrinsic muscles of the tongue. Adatia and Gehring [10] discovered afferent discharges in the hypoglossal nerve of cats and monkeys during passive stretching of the tongue. They also found that position sense and control of the tongue were retained despite blocking of the lingual and inferior alveolar nerves (which together carry sensory afferents for the entire tongue). [10] Bowman and Combs [11] found that in rhesus monkeys, hypoglossal afferents entered the central nervous system through the ventral rami of C2 and C3. These findings support the theory that the hypoglossal nerve has a proprioceptive component or, more importantly, that it is not a purely motor nerve.
Bogduk [7] suggests that the pathogenesis of NTS parallels that of Bell's palsy. Patients with Bell's palsy frequently report the sensation of numbness, even though the trigeminal nerve (cranial nerve V) is intact. He suggests that their sensation of numbness is explained by compression of the proprioceptive fibers that run in the facial nerve (affected in Bell's palsy). Thus, in both NTS and Bell's palsy, the numbness does not imply a loss of touch and pain sensation but rather reflects the impaired function of deep proprioceptive afferents. [7] In both conditions, the neuropathology is not as expected according to the symptom presentation, and the well-known Bell's palsy provides indirect support for the proposed neural connections of NTS. Bogduk [7] suggests that the dysfunction of the C1û2 facet joint compresses the ventral ramus of C2, which compromises the proprioceptive afferents entering the central nervous system via the connections between the hypoglossal nerve, the ansa cervicalis, and the cervical plexus.
Muscle spasm
Cassidy and Diakow [8] propose a different mechanism for NTS, which focuses on the anatomical relationships at the C1û2 facet joint. They report 3 cases of NTS that responded positively to a course of spinal manipulative therapy to the C1û2 articulation. [8] The close association of the C1û2 facet joint, the ventral ramus of C2, and the inferior oblique muscle forms the foundation of their theory. Only one fourth of the reported cases of NTS have any evidence of pathology at the occipitoatlantoaxial joints. [6] Because of the lack of evidence that a temporary incongruency or instability is actually occurring at C1û2, they propose that a spasm or hypertonicity of the inferior oblique muscle is responsible for the syndrome. All 3 cases demonstrated significantly reduced range of axial rotation, a movement that receives significant contributions from both the C1û2 articulation and the inferior oblique muscle. All 3 cases had complete resolution of their tongue symptoms and improved range of axial rotation in response to spinal manipulative therapy to the upper cervical spine daily for 2 weeks. They contend that if the C1û2 articulation was compromised by instability secondary to an inflammatory process or trauma then spinal manipulative therapy would be contraindicated because of the potential for further damage to the joint or possibly the spinal cord. [8] An additional 7 cases are described by Terrett, [3] where positive outcomes were achieved in response to SMT to the cervical spine.
Review of Treatment Options
The diagnosis of NTS is based on key information gathered from the history and physical examination, notably reproduction of unilateral tongue symptoms following rapid axial rotation of the cervical spine. Clinically, provocation of signs may be difficult and differential diagnoses including transient ischemic attack (TIA), migraine headaches, vertebrobasilar insufficiency (VBI), and carotid insufficiency may elevate concern. [1, 4]. Paresthesia in and around the mouth is common in VBI and migraine but not in half of the tongue in isolation. Hemianesthesia of the face, commonly associated with carotid insufficiency, may include half of the tongue but would not occur on its own. Lance and Anthony [1] explain that the occurrence of neck pain and tongue numbness is simultaneous; however, it is not possible to invoke vascular occlusion by neck rotation as a mechanism. Therefore, a direct neural connection is indicated. [1] There are no guidelines for the use of diagnostic imaging with regard to NTS, despite the hypothesis that in some cases degeneration of the occipitoatlantoaxial joints play a contributory role. [2û6, 8, 12].
Treatment of NTS has consisted of nerve root resection, atlantoaxial fusion, immobilization with a cervical collar, and SMT. [2, 4û6, 8, 13]. There are no randomized controlled studies comparing the relative success rates of the therapies listed above; therefore, the following review of treatment options will attempt to clarify the issue.
All of the literature available on NTS is case studies, with the exception of the paper by Bogduk [7] proposing the subluxation theory of its pathogenesis. [1û6, 8, 12û15]. In the absence of higher quality evidence, we review all available data concerning treatment options, outcomes, and suggest 2 categories of NTS that may facilitate appropriate clinical decision making.
Since the initial publication by Lance and Anthony [1] in 1980, there have been a total of 44 published cases of NTS. [1û6, 8, 12û15]. Historically, isolated cases of NTS are described as rare; however, Webb [5] suggests that tongue symptoms in association with cervical arthritis are not uncommon and are simply ôsubmerged in the plethora of neck-related symptoms.ö Bogduk [7] has also contradicted the notion that NTS is rare, based on discussions with other neurologists and neurosurgeons.
After reviewing the 2 proposed mechanisms of NTS, it seems logical that either the atlantoaxial subluxation theory by Bogduk [7] or the inferior oblique spasm theory by Cassidy and Diakow [8] may be accurate, depending on the clinical presentation and response to treatment.
In order to clarify this analysis, we can divide all reported cases of NTS into 2 groups based on clinical presentation: uncomplicated NTS and complicated NTS. Uncomplicated NTS (U-NTS) is defined as cases of NTS that appear to be idiopathic or began in association with trauma (ie, motor vehicle accident). Complicated NTS (C-NTS) is defined as NTS in association with an underlying disease process, such as severe degenerative joint disease, inflammatory disease (ie, ankylosing spondylitis), and myelopathic conditions (Table 1)
Of the 44 reported cases of NTS, only 23 discuss the treatment rendered. [1û6, 8, 12û15]. Of those 23 cases, 19 were categorized as U-NTS and 4 were deemed C-NTS. Thirteen were treated with spinal manipulative therapy directed at the cervical spine, while some also received SMT to other areas of the spine. [3, 8]. Four were treated with a cervical collar alone, and 1 wore a cervical collar and performed isometric exercises for the posterior musculature of the neck. [4, 5]. One was treated with stabilizing exercises for the posterior musculature of the neck only. [14] Two were treated with surgical intervention, and 1 was treated with a local steroid injection. Two were prescribed medication.
One case was categorized as C-NTS due to the presence of inflammatory disease and was successfully treated with fusion of the upper 2 cervical vertebrae. [3] The other surgical case was U-NTS, and resection of the C2 ventral ramus was only partially successful. [13] The use of a soft cervical collar appears to be of some benefit, particularly when combined with postural retraining of neck musculature and patient education. [4, 5].
The drug carbamazepine is used to control epileptic seizures and manic-depressive disorder and to relieve severe pain caused by damage to the cranial nerves.16 It was prescribed to a patient whose NTS was complicated by the presence of transverse thoracic spinal cord myelopathy, where it successfully relieved the NTS symptoms. [12] Amitriptyline is a tricyclic antidepressant that is used to help control bed-wetting in children. [16] It was prescribed to a 13ûyear-old male with U-NTS whose symptoms resolved; however, follow-up was nonspecific. [2]
O'Meara and Wise [17] described neck and tongue symptoms in 2 young females, 8 and 12 years of age, respectively. These cases were atypical presentations according to the classic description of NTS, and several signs suggested diagnoses such as lymphoma, juvenile rheumatoid arthritis, and chronic recurrent multifocal osteomyelitis. [17] These 2 cases lacked the characteristic hemiglossal paresthesia following axial rotation of the head and were excluded from this review of treatment options for NTS.
A large majority of the cases of NTS that describe the treatment rendered fit the criteria as uncomplicated (19/23). Of the 19 cases of U-NTS, 13 were successfully treated with SMT, particularly to the cervical spine. All 13 patients reported a significant reduction in symptom intensity and frequency (Table 2)
This review of treatment lends support to both the subluxation theory by Bogduk [7] and the spasm theory by Cassidy and Diakow [8] as the mechanism of NTS. Those patients who were categorized as C-NTS because of the presence of inflammatory disease (a leading contributor to acquired upper cervical instability) were successfully treated by stabilization through surgery, use of a cervical collar, and/or exercise. [3û5, 13, 14]. Those cases of U-NTS of idiopathic or trauma-related origin responded well to SMT. [3, 8].
Conclusion
Based on the information presented in this article, we suggest that the majority of cases of NTS are uncomplicated and the onset is usually idiopathic or secondary to trauma. Although upper cervical instability secondary to inflammatory disease, myelopathy, or severe degenerative joint disease (DJD) is unlikely in athletes, it remains a possibility. Intuitively, NTS in athletes is more likely caused by a spasm of the inferior oblique muscle, and once upper cervical instability is ruled out, it should be categorized as uncomplicated. In conclusion, SMT appears to be beneficial and should be considered in all cases of uncomplicated NTS, once upper cervical instability has been ruled out. Use of a cervical collar and postural training exercises in conjunction with patient education may be beneficial in all cases of NTS.
References
Lance, JW and Anthony, M.
Neck-tongue syndrome on sudden turning of the head.
J Neurol Neurosurg Psychiatry. 1980; 43: 97û101Chedrawi, AK, Fishman, MA, and Miller, G.
Neck-tongue syndrome.
Pediatr Neurol. 2000; 22: 397û399Terrett, AGJ.
Neck-tongue syndrome and spinal manipulative therapy.
In: H Vernon (Ed.) Upper cervical syndrome: chiropractic diagnosis and treatment.
Williams & Wilkins, Baltimore; 1988: 223û229Fortin, CJ and Biller, J.
Neck-tongue syndrome.
Headache. 1985; 25: 255û258Webb, J, March, L, and Tyndall, A.
The neck-tongue syndrome: occurrence with cervical arthritis as well as normals.
J Rheumatol. 1984; 11: 530û533Orrell, R and Garsden, CD.
The neck-tongue syndrome.
J Neurol Neurosurg Psychiatry. 1994; 57: 348û352Bogduk, N.
An anatomical basis for the neck-tongue syndrome.
J Neurol Neurosurg Psychiatry. 1981; 44: 202û208Cassidy, D and Diakow, P.
Treatment of neck-tongue syndrome by spinal manipulation: a report of three cases.
Pain Clinic. 1986; 1: 41û46Moore, K.
In: Clinically oriented anatomy. 3rd ed.
Williams & Wilkins, Baltimore; 1992: 664û875Adatia, AK and Gehring, EN.
Proprioceptive innervation of the tongue.
J Anat. 1971; 110: 215û220Bowman, JP and Combs, CM.
The cerebrocortical projection of hypoglossal afferents.
Exp Neurol. 1969; 23: 291û301Noda, S and Vmezaki, H.
Spinal neck-tongue syndrome.
J Neurol Neurosurg Psychiatry. 1984; 47: 751Elisevich, K, Stratford, J, Bray, G, and Finlayson, G.
Neck-tongue syndrome: operative management.
J Neurol Neurosurg Psychiatry. 1984; 44: 497û499Lois, I.
The cervicolingual syndrome.
Man Med. 1987; 3: 63û66Hankey, GJ.
Neck-tongue syndrome on sudden neck rotation.
Aust N Z J Med. 1988; 18: 181In: MS Berner, GN Rotenberg (Eds.) New guide to prescription and over the counter drugs.
Canadian Medical Association, Ottawa; 1996: 240O'Meara, M and Wise, G.
Painful torticollis with tongue atrophyùa different neck-tongue syndrome.
Neuropediatrics. 1995; 26: 276û280
Return to CASE STUDIES
Since 9-18-2004


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |