

Deconstructing Chronic Low Back Pain in the Older Adult -
Step by Step Evidence and Expert-Based Recommendations
for Evaluation and Treatment.
Part II: Myofascial PainThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: Pain Medicine 2015 (Jul); 16 (7): 1282–1289 ~ FULL TEXT
OPEN ACCESS Anthony J. Lisi, Paula Breuer, Rollin M. Gallagher, Eric Rodriguez,
Michelle I. Rossi, Kenneth Schmader, Joel D. Scholten, Debra K. Weiner
VA Connecticut Healthcare System
2Rehabilitation and Prosthetics Services,
Veterans Health Administration.
3University of Bridgeport College of Chiropractic.OBJECTIVE: To present an algorithm of sequential treatment options for managing myofascial pain (MP) in older adults, along with a representative clinical case.
METHODS: A modified Delphi process was used to synthesize evidence-based recommendations. A multidisciplinary expert panel developed the algorithm, which was subsequently refined through an iterative process of input from a primary care physician panel.
RESULTS: We present an algorithm and supportive materials to help guide the care of older adults with MP, an important contributor to chronic low back pain (CLBP). Addressing any perpetuating factors should be the first step of managing MP. Patients should be educated on self-care approaches, home exercise, and the use of safe analgesics when indicated. Trigger point deactivation can be accomplished by manual therapy, injection therapy, dry needling, and/or acupuncture.
CONCLUSIONS: The algorithm presented gives a structured approach to guide primary care providers in planning treatment for patients with MP as a contributor to CLBP.
KEYWORDS: Chronic Low Back Pain; Chronic Pain; Degenerative Disc Disease; Elderly; Low Back Pain; Lumbar; Myofascial Pain; Older Adults; Pain Disorder; Spinal Stenosis
From the FULL TEXT Article:
Introduction
Myofascial pain (MP) as first described by Travell and Simons, is defined by a localized region of palpable tightness and tenderness within a muscle that is characterized by resistance to passive elongation, and reproduction of a predictable pattern of referred pain on palpation. [1] The pathogenesis of MP is not fully understood, but can be a local muscle response to underlying mechanical factors (postural abnormalities, biomechanical faults, chronic strain), or a response to altered neurotrophic factors secondary to spondylosis. [2–4]
A characteristic feature of MP is the presence of localized palpable tender regions called trigger points (TrP). These have been identified on microscopic evaluation of involved muscles [5] and exhibit a distinct biochemical profile (i.e., inflammatory mediators, neuropeptides, cytokines, and catecholamines) as compared with normal muscles. [6, 7] The elevated tissue tension in TrP was shown to be decreased by the administration of general anesthesia, supporting a spinal segmentally mediated etiology. [8] It has also been proposed that TrP may have a bidirectional relationship with central sensitization, being both a cause as well as an effect. Preliminary evidence suggests the prolonged nociceptive input from TrP can sensitize dorsal horn neurons, whereas the referred pain phenomenon seen in TrP may in fact be the result of central sensitization. [9]
Studies have reported TrP prevalence ranging from 30% to 93%. [10] Using a structured examination, latent or active TrPs were identified in 93% of community-dwelling older adults with chronic low back pain (CLBP) attending a university-based pain management program. [11] Latent TrPs are painful when palpated, but the palpation-induced pain does not reproduce the patient's spontaneously reported pain, as occurs with active TrP. A subsequent study in older veterans with CLBP identified active TrP in approximately half of participants (Weiner, unpublished data), supporting MP as an important treatment target in older adults with CLBP.
Despite the commonplace nature of MP, allopathic medical education does not routinely include instruction in its evaluation and treatment. Thus, primary care physicians are often not confident in their ability to diagnose MP [12] and overlook it as a contributor to CLBP. This can result in misdirected, often suboptimal, unnecessarily invasive, and potentially dangerous treatments being prescribed. [13] This article presents an algorithm for managing MP in older adults, along with an illustrative case description.
Methods
This work was part of a larger project described previously. [14] We used a modified Delphi process to develop an algorithm (Figure 1) and evidence table (Table 1) providing the rationale for the individual algorithm components. The project principle investigator (DW) drafted an evidence-based treatment algorithm and evidence table, which were subsequently refined by an expert panel. The panel used the strongest available published evidence, supplemented by expert opinion and clinical experience as appropriate. The panel comprised five members, selected based on their recognition and expertise in their individual fields, representing geriatric medicine, pain medicine, physical medicine and rehabilitation, physical therapy and chiropractic.
Figure 1.
Algorithm for the evaluation and treatment of myofascial pain in an older adult.
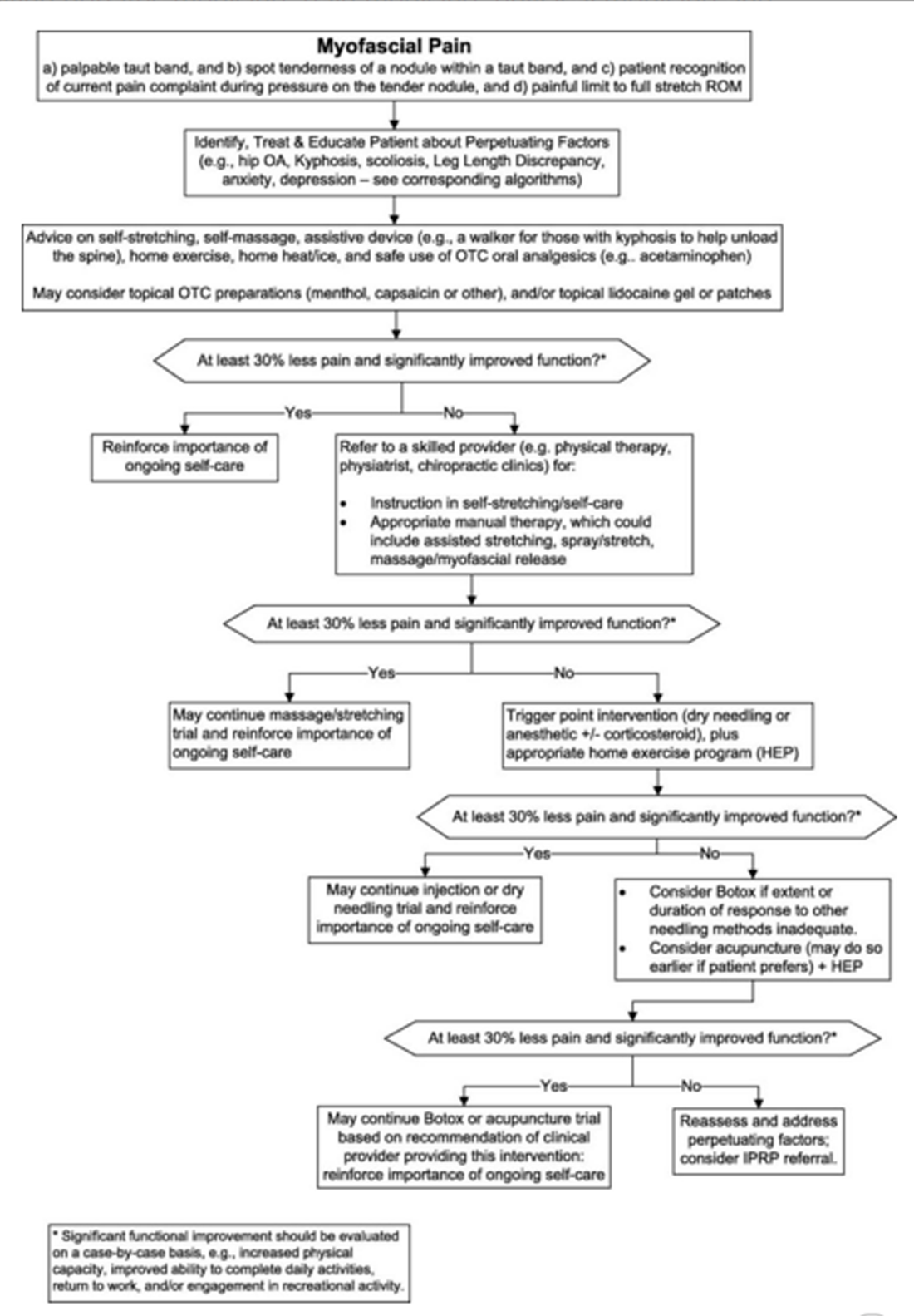
Table 1.
Maladaptive coping: theoretical and pragmatic underpinnings of algorithm recommendations
Algorithm component Comments 30% pain reduction as significant Data on 2724 subjects from 10 placebo controlled trials of pregabalin in diabetic neuropathy, postherpetic neuralgia, CLBP, fibromyalgia, and OA. Myofascial pain was not one of the conditions studied. In older adults with chronic low back pain myofascial pain is most often a pain comorbidity (i.e., accompanies the above conditions) rather than a sole contributor. Importance of identifying and treating perpetuating factors Travell and Simons published the seminal and authoritative textbook on myofascial pain. Within this textbook, numerous treatment techniques are outlined in detail. Key role of manual therapy Trigger point deactivation with injection versus dry needling. Numerous studies have been performed with highly variable quality, supporting trigger point injection, with or without injectate (i.e., dry needling). One study purported that the critical therapeutic element is the local twitch response. Botulinum toxin There is neither strong evidence for nor against the use of botulinum toxin for the treatment of myofascial pain. Based on this, we recommend referral only if other interventions have failed. Oral medications A number of variable quality trials have been performed that suggest a number of oral agents may benefit those with myofascial pain including tizanidine, cyclobenzaprine, clonazepam, alprazolam, diazepam, and amitriptyline. Because of the potential for adverse CNS effects, these medications should be used with extreme caution in older adults. Topical medications As with oral medications, trials of topical medications have been of variable quality and, therefore, there is no strong evidence to recommend their use. Because of their favorable safety profile topical lidocaine, methylsalicylate, menthol, diclofenac, and thiocolchicoside can be tried.
The materials were then distributed to a 9–member primary care provider review panel that provided feedback using a structured questionnaire. The expert panel used this feedback to make additional modifications, and the process was repeated until no further revisions were recommended.
Case Presentation
Relevant History
The patient is a 72–year-old male presenting to his Department of Veterans Affairs (VA) primary care provider (PCP) with a 4–year history of low back pain attributed to a motor vehicle accident. He complains of a burning pain in the right lower lumbar region radiating to the right upper gluteal region with an average intensity of 5/10. The pain is present every day, approximately 50% of waking time each day, typically brought on by prolonged standing or walking, and relieved by sitting or lying down, although prolonged sitting also could cause pain. He denies lower extremity pain, weakness, numbness, tingling, unexplained weight loss, and bladder or bowel problems. He states, at the time of the accident, he was diagnosed with a bulging disc, and was prescribed ibuprofen, physical therapy (ultrasound, bicycle exercise, and core strengthening), chiropractic care (lumbar spinal manipulation and traction) and two rounds of epidural steroid injections. He reports no lasting improvement after any of these interventions. He continued to take over the counter ibuprofen 400 mg, two times per day. Over the past 4 months, his pain has been preventing him from bowling and playing with his grand-daughter, so he increased ibuprofen to 600 mg, two times per day, but he began to experience abdominal pain. He was prescribed oxycodone 5 mg/acetaminophen 325 mg, one pill every 6 hours as needed for pain, however, this caused sedation that interfered with activities of daily living.
Relevant Physical Examination
The patient is a pleasant, alert, and cooperative African American male in no apparent distress. His gait and station are unremarkable. There is increased low back pain at the end range of flexion and when returning to neutral from a flexed position. Straight leg raise, lumbar, hip, and sacroiliac orthopedic testing are painless. [21] Lower extremity motor strength, reflexes, and light touch are within normal limits. The lower lumbar paraspinal muscles are tight but not tender. There is a taut band in the right paraspinal musculature at the T12–L1 level, that when palpated reproduces the patient's spontaneously reported pain in the right lower lumbar and upper gluteal region.
Imaging
Lumbar magnetic resonance imaging (MRI) ordered by a previous provider revealed mild to moderate multilevel degenerative disc disease, and mild to moderate bilateral foraminal stenosis at L4–5 and L5–S1.
Clinical Course
The patient was referred to the VA chiropractic clinic, where he received manual myofascial release [22] of the thoracolumbar paraspinal muscles, and was instructed in self-massage with topical capsaicin cream. He was also taught a few key stretching and postural exercises. After four sessions, the patient reported pain had decreased to 2/10 intensity on average, and frequency had decreased to 3 days per week, with duration decreased to 10% of waking hours on those days. He was able to resume bowling and playing with his grand-daughter. He rated this as a 75% global impression of change. He was treated another two times with no further improvement, thus was offered a consultation for TrP injection. The patient was satisfied with the current outcome and declined any additional follow-up.
Approach to Management (Figure 1)
This patient with CLBP was presumed to have MP as a major contributor to his pain and functional limitations. Even the burning and radiating quality of his pain, which may indicate a neuropathic origin, are consistent with the diagnosis of MP. As is typical of most older adults with CLBP [11], there likely was more than one pain generator in this patient. Nevertheless, the key exam features seen here (muscle tightness [i.e., taut bands] with palpable point tenderness [i.e., TrP] as well as reproduction of distant [referred] pain that reproduces the patient's spontaneously reported pain) give the clinician a plausible mechanical target that can be approached in a systematic fashion. Common locations of TrPs thought to contribute to CLBP are the lower thoracic and lumbar erector spinae, the quadratus lumborum, and the gluteal muscles. [1]
It is worth noting that before presenting to his VA PCP, this patient was initially managed by a private physician group that ordered the MRI and gave the patient the diagnosis of lumbar disc disease. In the majority of low back pain cases, advanced imaging is not indicated [23], and the findings have no bearing on the diagnosis of MP.
As shown in the algorithm (Figure 1), the initial approach to managing MP is to address any potential perpetuating factors such as scoliosis, leg length discrepancy, hip pathology, depression or anxiety, as these all may contribute to chronic muscle tightness. Appropriate treatment of any of these factors improves the likelihood of success of subsequent treatment of the MP. When possible, any factors that acutely precipitated an episode of MP also should be identified, and ameliorated to the greatest extent possible. Medication contributors to muscle pain and/or dysfunction, such as statins, also should be modified if possible. [24] Systemic illnesses such as Parkinson's disease that cause muscle dysfunction and may perpetuate myofascial pain also should be targeted as part of comprehensive treatment. [25]
Providers should counsel patients on the importance of self-care including stretching, superficial heat/ice, self-massage, appropriate topical preparations, and the use of acetaminophen or other safe analgesics when indicated. These relatively simple measures are often beneficial and can empower patients to be active participants in their own health. However when these measures alone are insufficient, the next management step would be TrP deactivation. As the name implies, TrP deactivation aims to neutralize the chronically hyperactive/hypersensitive region of the involved muscle. This can be accomplished by manual therapy, injection therapy, and/or dry needling.
The manual therapy approaches to TrP deactivation essentially fall into two categories: stretching techniques (postisometric relaxation, spray and stretch, etc.) and/or massaging techniques (ischemic compression, myofascial release, etc.). In this case, the manual therapy was provided by chiropractors, yet these treatments also can be provided by medical/osteopathic physicians, physical therapists, and others with appropriate training. The choice of technique and degree of mechanical load applied must be tailored to the individual patient's tolerance. In this case, the patient reported satisfactory improvement after a short course of manual therapy and home instructions. Had there been little or no improvement to this initial intervention, management could have evolved in a stepwise fashion to include wet needling (i.e., injection of an anesthetic +/? corticosteroid) or dry needling, Botox injections or acupuncture. While wet needling is practiced commonly, evidence does not indicate that it is superior to dry needling, the introduction and subsequent manipulation of an acupuncture or hypodermic needle into a TrP, without any injectate. [26] The critical therapeutic element in both approaches is thought to be obtaining a local twitch response. [27] As depicted in Figure ?, all of these interventions should be accompanied by ongoing self-care and reassurance. Patients should be educated that judicious use of passive therapies can be appropriate, but that long-term benefit requires active patient engagement. As many factors may facilitate or impede a patient's engagement in self-care and physical activity [28], appropriate management should include collaborative goal setting. [29]
Another important aspect of treating MP in older adults is building muscular resilience. As noted in the introduction to this CLBP series, a central concept in gerontology and in caring for older adults is homeostenosis, defined as the progressive restriction of an organism's ability to respond to stress as it ages. [30, 31] Sarcopenia, diminished muscle bulk and quality associated with normal aging, is one component of homeostenosis in the muscular system. [32, 33] It is possible that sarcopenia is an important perpetuating factor for MP in older adults, possibly accounting for the high prevalence of MP in these patients. Because of the undeniable presence of sarcopenia in all older patients, resilience building through a long-term home exercise program is essential.
The biopsychosocial model of chronic pain requires clinicians to consider other aspects in addition to the involved musculoskeletal target tissue. This patient exhibited no significant psychosocial contributors to his CLBP and difficulty functioning, For many patients with chronic musculoskeletal pain in general [34–36], and MP in particular [37], depression, anxiety, and other mental health conditions do contribute to pain and disability. Concurrent treatment of depression and pain has been shown to yield more favorable outcomes for both conditions. [38] Depending on the factors that have been identified to perpetuate the patient's MP, optimal management may require a collaborative team approach including psychiatry, health psychology, addiction medicine, social work, and/or other disciplines. [39, 40] A patient-centered approach including shared decision making has been shown to result in improved outcomes. [41]
Resolution of Case
The patient continued to follow with his PCP and reported no significant back pain for the next 12 months. He remained fully functional in his desired activities of daily living. He was somewhat compliant with the active care instructions over this time, but he eventually discontinued his home exercise and self-management program. One year later he returned with increased symptoms and no new precipitating incident or perpetuating factors. His PCP advised the patient to resume home exercises and self-care, and if the pain remains bothersome he will be referred for manual therapy or TrP injection.
It is worth highlighting most nonsteroidal anti-inflammatory drugs (NSAIDs) are included in Beers Criteria for potentially inappropriate medications in older adults. [42] The older patient presented developed adverse effects associated with NSAIDs that could have been avoided had treatment been targeted specifically to his MP.
Summary
Providers should consider the contribution of MP in older adults with CLBP. Current evidence supports a number of interventions for CLBP but none have been shown to be clearly superior. [23, 43] Identification and treatment of MP as part of the CLBP syndrome [14] requires neither imaging nor exposure to procedures with significant risk. Thus, prioritizing its identification and treatment, along with factors that precipitate and/or perpetuate it has the potential to substantially benefit quality of life with minimal associated risk. The MP algorithm presented in this article provides guidance for a stepwise approach that takes into account risk/benefit and patient preference. For optimal treatment planning, goals and expectations must be congruent among patients, primary care providers, and specialists.
Box 1 Myofascial pain diagnostic criteriaHistory: Acute, subacute or chronic regional muscle pain and/or stiffness
Exam findings: Palpable taut band(s) within the muscle
Spot tenderness of a nodule within a taut band, palpation of which produces:
Concordant pain
Predictable pattern of referred pain
Local muscle twitch response
Painful limit to full stretch ROM
Acknowledgments
This material is based on work supported by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development, Rehabilitation Research and Development Service. The contents of this report do not represent the views of the Department of Veterans Affairs or the US government. The authors thank Dave Newman for his thoughtful review of the manuscript.
Funding sources:
Veterans Administration Rehabilitation Research & Development Service.
Conflict of interest:
The authors have indicated that they have no conflicts of interest regarding the content of this article.
References:
Travell JG, Simons DG.
Myofascial pain and dysfunction: The trigger point manual.
Baltimore: Williams and Wilkins; 1983Gunn CC, Milbrandt WE.
Early subtle signs in low back sprain.
Spine 1978;3:267–81Gunn CC, Milbrandt WE.
Tenderness at motor points––a diagnostic and prognostic aid for low back injury.
J Bone Joint Surg 1976;58A:815–25Gunn CC.
Neuropathic myofascial pain syndromes.
In: Loeser J, Butler S, Chapman C, eds. Bonica's Management of Pain,
3rd edition. Philadelphia, PA:
Lippincott Williams & Wilkins; 2001: 522–9Mense S, Simons DG, Hoheisel U, Quenzer B.
Lesions of rat skeletal muscle after local block of acetylcholinesterase and neuromuscular stimulation.
J Appl Physiol 2003;94:2494–501Shah J, Phillips T, Danoff J, Gerber LH.
An in vivo microanalytical technique for measuring the local biochemical milieu of human skeletal muscle.
J Appl Physiol 2005;99:1977–84Shah JP, Danoff JV, Desai MJ, et al.
Biochemicals associated with pain and inflammation are elevated in sites near to and remote from active myofascial trigger points.
Arch Phys Med Rehabil 2008;89:16–23Buchmann J, Neustadt B, Buchmann-Barthel K, et al.
Objective measurement of tissue tension in myofascial trigger point areas before and during the administration of anesthesia with complete blocking of neuromuscular transmission.
Clin J Pain 2014;30(3):191–8Fernández-de-las-Peńas C, Dommerholt J.
Myofascial trigger points: Peripheral or central phenomenon?
Curr Rheumatol Rep 2014;16(1):395Simons D.
Clinical and etiological update of myofascial pain from trigger points.
J Musculoskel Pain 1996;4:93–2122Weiner DK, Sakamoto S, Perera S, Breuer P.
Chronic low back pain in older adults: Prevalence, reliability, and validity of physical examination findings.
J Am Geriatr Soc 2006;54:11–20Cayea D, Perera S, Weiner DK.
Chronic low back pain in older adults: What physicians know, what they think they know, and what they should be taught.
J Am Geriatr Soc 2006;54(11):1772–7Gerwin R.
Diagnosis of myofascial pain syndrome.
Phys Med Rehabil Clin N Am 2014;25(2):341–55Weiner DK.
Deconstructing Chronic Low Back Pain in the Older Adult -
Shifting the Paradigm from the Spine to the Person
Pain Med 2015; 16 (5): 881–885Farrar JT, Young JP, Jr, LaMoreaux L, Werth JL, Poole MR.
Clinical Importance of Changes in Chronic Pain Intensity
Measured on an 11-point Numerical Pain Rating Scale
Pain 2001 (Nov); 94 (2): 149-158Simons D, Travell J, Simons L.
Travell and Simon's Myofascial Pain and Dysfunction: The Trigger Point Manual. 2nd edition.
Baltimore, MD: Williams & Wilkins; 1999Hong CZ.
Lidocaine injection versus dry needling to myofascial trigger point. The importance of the local twitch response.
Am J Phys Med Rehabil 1994;73:256–63Annaswamy TM, et al.
Emerging concepts in the treatment of myofascial pain: A review of medications, modalities, and needle-based interventions.
PM&R 2011;3:940–61Soares A, Andriolo RB, Atallah AN, daSilva EM.
Botulinum toxin for myofascial pain syndromes in adults.
Cochrane Database Syst Rev 2014;25;7: CD007533The American Geriatrics Society 2012 Beers Criteria Update Expert Panel.
American Geriatrics Society updated Beers Criteria for potentially inappropriate medication use in older adults.
J Am Geriatr Soc 2012;60:616–31Hoppenfeld S.
Physical examination of the lumbar spine.
In: Hoppenfeld S, Hutton R, Hugh T, eds. Physical Examination of the Spine and Extremities.
Norwalk, CT: Appleton & Lange; 1976Furlan AD, Brosseau L, Imamura M, Irvin E.
Massage for low-back pain: A systematic review within the framework of the cochrane collaboration back review group.
Spine 2002;27:1896–910Chou R, Qaseem A, Snow V, Casey D, Cross JT Jr., Shekelle P, Owens DK:
Diagnosis and Treatment of Low Back Pain: A Joint Clinical Practice Guideline
from the American College of Physicians and the American Pain Society
Annals of Internal Medicine 2007 (Oct 2); 147 (7): 478–491Valiyil R, Christopher-Stine L.
Drug-related myopathies of which the clinician should be aware.
Curr Rheumatol Rep 2010;12:213–220Rana AQ, Kabir A Jesudasan M, Siddiqui I, Khondker S.
Pain in Parkinson's disease: analysis and literature review.
Clin Neurol Neurosurg 2013;115:2313–7Kalichman L, Vulfsons S.
Dry needling in the management of musculoskeletal pain.
J Am Board Fam Med 2010;23:640–6Vulfsons S, Ratmansky M, Kalichman L.
Trigger point needling: Techniques and outcome.
Curr Pain Headache Rep 2012;16:407–12Newitt R, Barnett F, Crowe M.
Understanding factors that influence participation in physical activity among people with a neuromusculoskeletal condition: A review of qualitative studies.
Disabil Rehabil 2015;14:1–10Tan V, Cheatle MD, Mackin S, Moberg PJ, Esterhai JL Jr..
Goal setting as a predictor of return to work in a population of chronic musculoskeletal pain patients.
Int J Neurosci 1997;92:(3-4):161–70Becker PM, Cohen HJ.
The functional approach to the care of the elderly: A conceptual framework.
J Am Geriatr Soc 1984;32:923Karp JF, Shega JW, Morone NE, Weiner DK.
Advances in understanding the mechanisms and management of persistent pain in older adults.
Br J Anaesth 2008;101:111–20Moore AZ, Caturegli G, Metter EJ, et al.
Difference in muscle quality over the adult life span and biological correlates in the Baltimore longitudinal study of aging.
J Am Geriatr Soc 2014;62:230–6Studenski SA, Peters KW, Alley DE, et al.
The FNIH sarcopenia project: Rationale, study description, conference recommendations, and final estimates.
J Gerontol A Biol Sci Med Sci 2014;69:547–58Morone NE, Belnap BH, He F, et al.
Pain adversely affects outcomes to a collaborative care intervention for anxiety in primary care.
J Gen Intern Med 2012;28:58–66Lew HL, Otis JD, Tun C, et al.
Prevalence of chronic pain, posttraumatic stress disorder, and persistent postconcussive symptoms in OIF/OEF veterans: Polytrauma clinical triad.
J Rehabil Res Dev 2009;46:697–702Stratton KJ, Clark SL, Hawn SE, et al.
Longitudinal interactions of pain and posttraumatic stress disorder symptoms in U.S. Military service members following blast exposure.
J Pain 2014;15(10):1023–32Velly AM, Gornitsky M, Philippe P.
Contributing factors to chronic myofascial pain: A case-control study.
Pain 2003;104:491–99Kroenke K, Bair MJ, Damush TM, et al.
Optimized antidepressant therapy and pain self-management in primary care patients with depression and musculoskeletal pain: A randomized controlled trial.
JAMA 2009;301:2099–110Gatchel RJ, McGeary DD, McGeary CA, Lippe B.
Interdisciplinary chronic pain management: Past, present, and future.
Am Psychol 2014;69:119–30Stanos S.
Focused review of interdisciplinary pain rehabilitation programs for chronic pain management.
Curr Pain Headache Rep 2012;16:147–52Stewart M, Brown JB, Donner A, et al.
The impact of patient-centered care on outcomes.
J Fam Pract 2000;49:796–804American Geriatrics Society Beers Criteria Update Expert Panel.
American Geriatrics Society updated Beers criteria for potentially inappropriate medication use in older adults.
J Am Geriatr Soc 2012;60:616–31Haldeman S, Dagenais S.
A Supermarket Approach to the Evidence-informed Management of Chronic Low Back Pain
Spine Journal 2008 (Jan); 8 (1): 1–7
Return to LOW BACK PAIN
Since 1-22-2016


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |