

Magnetic Resonance Spectroscopy Displays the Structural
and Biochemical Effects of Spinal Manipulative Therapy
in Lumbar Discogenic Pain: A Case ReportThis section is compiled by Frank M. Painter, D.C.
Send all comments or additions to:7 Frankp@chiro.org




FROM: Research Square (Apr 4, 2023) ~ FULL TEXT

OPEN ACCESS Jessica F Billham, Erika Evans Roland, Matthew F. Gornet, Kelly Brinkman,
Francine Schranck, Jim Cox, Norman W. Kettner
Logan University
Chesterfield, MO, US
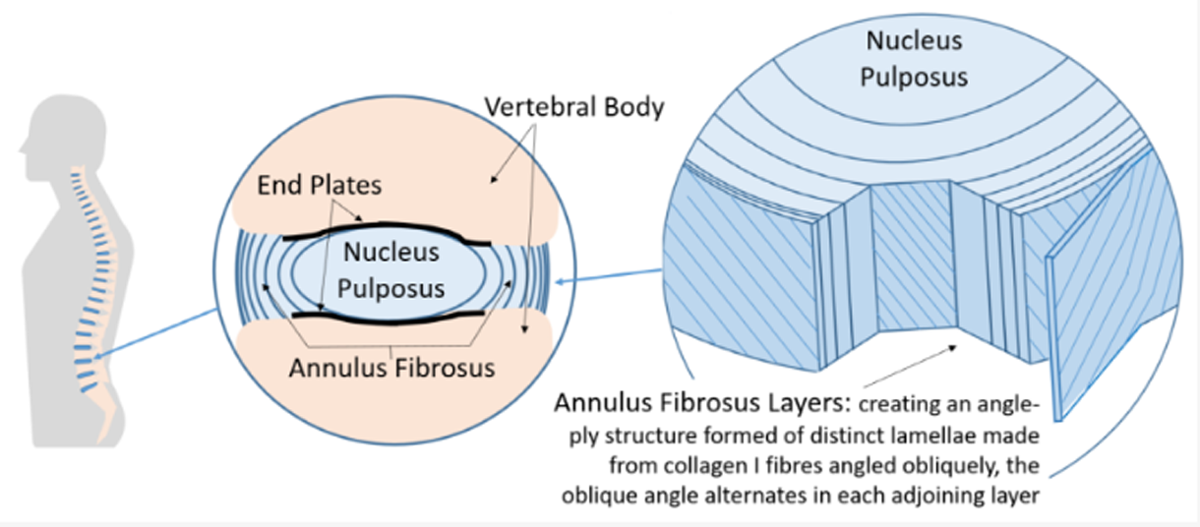
The Vertebral Disc
FROM: Bioengineering 2022INTRODUCTION: We report utility of magnetic resonance spectroscopy (MRS) in identifying the intervertebral disc (IVD) as a pain generator, describe the contemporary pathophysiology underlying biochemical and structural components of discogenic low back pain (DLBP), and exhibit therapeutic responses to spinal manipulation.
CLINICAL FEATURES: A 29-year old man presented with uncomplicated low back pain (LBP). The nonspecific presentation and clinical exam findings were consistent with non-specific LBP with the IVD as likely pain generator.
INTERVENTION AND OUTCOMES: Conventional magnetic resonance imaging showed findings of IVD degeneration including Modic Type 1 changes consistent with a diagnosis of DLBP. MRS was utilized for structural and biochemical analysis of the IVDs. Altered spectral features confirmed a DLBP diagnosis. The patient underwent 12 Cox Flexion Distraction treatments at a chiropractic teaching clinic. Follow-up MRS revealed improved IVD spectral features including decreased biochemical pain markers and increased glycoprotein biosynthesis suggesting improved IVD structural integrity.
CONCLUSION: We report the first utilization of magnetic resonance spectroscopy (MRS) to quantify structural integrity and biochemical pain profile of the IVD in a conservatively managed discogenic low back pain (DLBP) patient. Findings of this case suggest spinal manipulative therapy in DLBP management may improve the structural integrity of IVDs and alter pain biochemistry.
From the Full-Text Article:
Introduction
Low back pain (LBP) is a common health problem and cause of disability affecting upwards of 80% of adults in their lifetime. [1–3] In the USA, LBP healthcare costs have increased substantially in the past decade, making this health problem a burden on both the individual and society. [2,3] Pain is multifaceted including physiologic and psychosocial factors that should be considered in the workup of all LBP cases. [4–6]
Francio et al stated we should “aim to pursue the right treatment, for the right patient, at the right time, and directed to the correct anatomical target”. [7] Currently, 90% of LBP is classified as nonspecific (NSLBP) and lacks a clear pathoanatomical etiology, or anatomical target. Specific LBP, however, has a clear pathoanatomic source, either spinal or non-spinal in origin. [8, 9] Often discogenic low back pain (DLBP) is initially classified as nonspecific Low back pain (NSLBP) due to vague presentations and inconclusive diagnostic test results. [10]
Qualitative imaging predominates attempts to specify a pathoanatomic source in NSLBP, however imaging findings correlate poorly with clinical presentations. [11] Qualitative imaging may show intervertebral disc (IVD) degeneration, such as reduced IVD height and endplate sclerosis on lumbar radiography and reduced IVD fluid signal intensity on conventional magnetic resonance imaging (MRI). [12–14] Pfirrmann grading is a common method to evaluate IVD degeneration on MRI but is insensitive to early biochemical alterations. Instead, it centers on the IVD fluid signal intensity, distinctions between separate IVD structures, and IVD height. It is a 5-class system with 1 representing normal and 5 representing severe degeneration. [15, 16] Provocative discography is another qualitative IVD analysis performed by injecting contrast into the suspected IVD to highlight morphologic changes under fluoroscopy. [4, 13]
The IVD is considered the pathoanatomic source of LBP when there is LBP with conventional MRI evidence of IVD degeneration, also termed “active discopathy”. [17, 18] When the IVD undergoes degeneration, many structural and biochemical changes occur that may trigger pain. Degenerative IVD findings on MRI include Modic Type 1 bone marrow edema, annular fissures and herniation of the nucleus pulposis (NP) due to loss of IVD structural integrity, and high intensity zones. [6, 12, 19–22] However, these findings are also present in asymptomatic patients. [12–14]
To support qualitative MRI findings, quantitative imaging such as magnetic resonance spectroscopy (MRS) in identifying the intervertebral disc (IVD) can quantify the structural and biochemical changes that occur with IVD degeneration. [4, 15, 23, 24] Metabolites like proteoglycans and lactate serve as biomarkers to identify specific IVDs as the pathoanatomic source of pain when quantified using novel NOCISCAN technology. [19, 22]
Patients with DLBP should be carefully paired with appropriate treatment options. This may include surgery, opioid prescription, and/or conservative care. Non-surgical, non-pharmacologic treatments are the commonest interventions and include patient education, spinal manipulative therapy (SMT) and exercise. [25] In fact, for some patients, these conservative options may be as effective as surgery and opioids in reducing pain, but are more cost effective with a lower risk of complication. [26, 27]
We describe a case of NSLBP with qualitative lumbar IVD degeneration suggesting a DLBP diagnosis. Spectroscopic analysis of the lumbar IVDs quantified structural and biochemical changes and confirmed the IVD as the pathoanatomic source of pain. The IVD structural integrity and degenerative pain biochemistry after a course of conservative management were markedly improved. This is the first publication of MRS utilized to quantify IVD structural integrity and biochemical changes pre- and post- SMT in a case of DLBP. The authors aim to show utility of MRS to identify the IVD as a pathoanatomic source of pain in LBP patients, review the current understanding of the structural and biochemical components of DLBP, and demonstrate how conservative management of DLBP may provide benefit.
Case Report
A 29-year-old male office worker and military reservist presented to a chiropractic teaching clinic with an acute flare of LBP of 2 days duration. His goals were to reduce pain, return to weight-lifting and golfing, and improve function for a long-term active lifestyle. Maintaining an adequate level of fitness to perform necessary military duties was important to him.
Prior episodes of similar pain occurred over the past several years that the patient attributed to jumping out of helicopters while in active military duty. The patient sought care at the emergency department for a comparable episode approximately 1 year prior and received an epidural steroid injection and pain medications which provided minimal relief. Subsequent physical therapy provided no perceived benefit. Pain recurred 6 months prior to presentation and was self-managed with rest, ice, and over-the-counter pain-relievers until resolution over the course of 3 months.
The presenting pain was rated a 40 on the Numeric Pain Rating Scale (NPRS) and localized to the lower lumbar region without radiation into the extremities. It was described as a constant deep ache that progressed to a sharp pain with lumbar flexion. Standing and stretching provided some relief. The Oswestry Disability Index (ODI) was a 34%, representing moderate disability. The patient denied experiencing any night pain, night sweats, unexplained weight loss, or uncontrolled bowel/bladder function. Psychosocial contributors included fear-avoidance of tasks related to military physical fitness like deadlifting and running.

Figure 1
page 13

Figure 2
page 14

Figure 3
page 15The patient presented without antalgia, although needed support when transitioning postures for fear of increased pain. Clinical examination of lower extremity dermatomes, myotomes, and deep tendon reflexes revealed normal neurological function. The lumbar paraspinal musculature was taut and tender bilaterally. Active lumbar ranges-of-motion in flexion, extension, and lateral bending were full but reproduced pain. The patients’ pain was exacerbated with Yeoman and Kemp orthopedic tests bilaterally. Milgram and Dejerine Triad were negative.
Based on the non-specific presentation and absence of red flags, the patient was diagnosed with NSLBP with suspicion of discogenic etiology and identified as a good candidate for conservative care. Because fear-avoidance behaviors were present, reassurance and education about the benign and self-resolving nature of LBP were emphasized throughout treatment. After 3 sessions of Cox flexion-distraction SMT over 3 weeks without improvement, lumbar radiography was acquired (Figure 1a). A lumbar MRI was obtained 6 weeks after initiation of care for qualitative evaluation of the IVDs (Fig. 1b,d).
Magnetic resonance spectroscopy was performed with the MRI to analyze the lumbar IVD structural integrity and biochemical composition. Utilizing NOCISCAN post-processing, structural and biochemical markers were charted (Fig. 2a,b and Fig. 3a,b). The results signified the L3/4 and L5/S1 IVDs were the patients’ pain generators with significant loss of structural integrity of the L5/S1 IVD.
These quantitative findings confirmed a DLBP diagnosis. Care continued with emphasis on patient reassurance and 16 treatments of Cox flexion-distraction SMT over 16 weeks. There were subjective improvements in pain and functional levels with full return to weight lifting and golf. However, the patient experienced another similar flare while getting dressed in the 16th week of care. He rated the pain at a 60 on the NPRS and the ODI increased to 46%. Based on patient preference, high-velocity low-amplitude SMT was utilized for 2 treatments over 2 weeks. At this time a repeat MRI was utilized to evaluate any qualitative IVD changes (Fig. 1c,e). A post-treatment quantitative MRS performed with the MRI demonstrated marked improvement in the structural and biochemical findings (Fig. 2c,d and Fig. 3c,d).
Discussion
Qualitative imaging features of IVD degeneration are seen in 40% of people under 30-years-old and in 90% of people over 50-years-old. [15] Many do not experience symptoms which speaks to the fact that IVD degeneration is common age-related change. [7] However, the severity of IVD degeneration predicts first time LBP episodes. [21] The structural and biochemical mechanisms of discogenic low back pain (DLBP) attempt to explain the transformation from asymptomatic IVD degeneration to symptomatic IVD degeneration, or active discopathy. [6, 7, 17, 18]

The Vertebral Disc
FROM: Bioengineering 2022The nucleus pulposis (NP) of a healthy IVD resists compression, a function derived from high water and proteoglycan content paired with an intact annulus fibrosis (AF). [22, 23, 28] An early characteristic of IVD degeneration is proteoglycan breakdown. Proteoglycans notochordal cell derivatives that decline in quantity with increasing age. [29, 30] Because notochordal cells are less abundant, intact proteoglycan content decreases and allows for reduced imbibition of water and nutrients into the NP. The hoop stress forces of the AF are reduced as the NP water content decreases, allowing for structural causes of discogenic low back pain (DLBP) such as annular tears and disc herniations. [21, 32] In disc herniation patients, presentation is often with radicular symptoms and less often with centralized pain. In this case, the patient had a disc protrusion but no radicular symptoms further suggesting the pain source as a degenerative IVD and not nerve root.
Other considerations in DLBP are biochemical changes that occur. The healthy IVD is avascular requiring the use of glycolysis as its energy system which creates lactate as a byproduct. In a healthy IVD with imbibition of water and nutrients in exchange for waste products, lactate levels stay low. Decreased imbibition occurs with reduced intact proteoglycan. As the degenerative process continues, cartilaginous endplates calcify further reducing imbibition. [30, 31] Lactate accumulation results in a reduced intradiscal pH, accelerated degeneration of proteoglycans, hypoxia, and cell death [15, 19, 22]. With increased lactate there is also increased proinflammatory cytokines, more so in IVDs with adjacent Modic Type 1 changes. [6, 15, 20, 30, 32]
Structural changes occur synchronously with biochemical changes. A healthy IVD is only innervated in the outer layers of the AF. When annular fissures occur, inflammatory cells and lactate present in a degenerative IVD can reach the normally innervated outer layers of the AF. These tissues have acid sensing ion channels making them sensitive to pH changes. [31–33] More importantly, biochemical changes and annular fissures create an environment conducive to vascular granulation tissue formation and neoneural growth. New nerves grow into previously aneural areas of the AF as deep as the nucleus pulposis (NP) where intradiscal pH is low and proinflammatory cytokines are high. [15, 21, 31, 32]
While the structural and biochemical causes of DLBP are known, there are no adequate sensitive and specific clinical tests for DLBP. The goal of clinical evaluation of LBP patients should be to identify or rule out serious pathology. Once accomplished, more benign pathoanatomic diagnoses for LBP can be considered. Precise clinical characteristics of DLBP are not well defined and complex pathomechanisms of LBP frequently make diagnosis uncertain. Although centralized LBP is highly sensitive for DLBP, it is not specific. [21] Adding further complexity to LBP patients are contributing psychosocial factors as they impact prognosis and treatment options. These individual contextual factors and lack of specificity in clinical diagnosis lead to significant variability in management of DLBP patients. [4, 11]
A solution to the lack of specificity in the diagnosis of DLBP may be a more specific imaging modality. Magnetic resonance spectroscopy is a non-invasive, objective quantification of chemical differences in tissues used to evaluate specific biomarkers. It takes advantage of the fact that all metabolites interact slightly differently with a magnetic field based on the distribution of electrons in a molecule. Consequently, each metabolite resonates at slightly different magnetic resonance frequencies, or chemical shifts. [4, 34] Specific to DLBP, Keshari et al showed metabolites like proteoglycans and lactate serve as spectroscopically quantifiable biomarkers in ex-vivo IVD specimens. [22]
Recently, Gornet et al utilized MRS to characterize in-vivo metabolic features of painful lumbar IVDs using NOCISCAN technology. [19] NOCISCAN utilizes MRS to assess relative biomarkers for IVD degeneration and pain. Imaging technologists mark a region-of-interest inside the IVD NP on a conventional MRI sequence to represent a voxel of information. NOCISCAN-LS exam proprietary protocol is run and a cloudbased post-processing analysis creates MRS spectra, or graph of relative peaks of resonance plotted right to left on the x-axis. The y-axis describes the degree of chemical shift. These spectral signatures are measured with a diagnostic algorithm to generate a NOCISCORE ratio of proteoglycans to lactate which has been correlated to provocative discography.
Gornet et al showed that the spectral measurements of proteoglycans and lactate that were used to create the NOCISCORE score showed reliable identification of painful IVDs when compared to provocative discography and had a significant favorable impact on surgical outcomes. [19] Because provocative discography is painful, invasive, and leads to accelerated degeneration in the injected segments, there is debate about the overall usefulness of the modality. [4, 13, 14] Gornet et al showed MRS compares to provocative discography without the associated risks. [19]
In our case, the painful IVDs identified were L5/S1 which demonstrated qualitative changes, but also L3/4 which interestingly did not show any qualitative changes. This suggests that MRS and NOCISCAN technology can identify degenerative IVD changes earlier than qualitative imaging modalities. After treatment, both of these levels improved in their structural and biochemical components. Unfortunately, this patient experienced an exacerbation of pain days prior to the second scan. The L5/S1 NOCISCORE was increased in the post-treatment scan, but since NOCISCORE is a ratio of the proteoglycans to lactate, it likely represents the significant increase in proteoglycans associated with the SI-SCORE (Fig. 2a,c and Fig. 3a,c). Alternatively, it may be explained by variability in temporal expression of molecular scale events from those at tissue levels. Additionally, there is a known disconnect between degenerative structure and function that varies in both frequency and duration. More research could assess IVD structural integrity and pain biomarkers to understand when these changes occur and how they evolve during treatment.
Non-surgical treatments are most commonly utilized in DLBP patients. The most non-invasive option is patient education, which should be employed alongside every other treatment used in accordance with the Biopsychosocial patient care model. Patient education includes advice to stay active, assurance of the benignity of DLBP, and its favorable course, which was utilized in our case. Relaxation techniques and Cognitive Behavioral Therapy can modify negative thoughts and rumination about pain/disability and to address any coexisting catastrophization and kinesophobia. [9, 26, 35]
Other non-surgical treatments include opioid analgesic therapy (OAT), exercise, and SMT. Whedon et al compared long-term outcomes for patients who initiated LBP care with SMT to patients who initiated care with OAT. They concluded the SMT group had fewer rates of escalated care thereby reducing overall cost burden. [27] A systematic review and meta-analysis performed by Paige et al showed statistically significant improvement of SMT similar to NSAIDs, but with only transient minor musculoskeletal harms, whereas NSAIDS can be very harmful to certain patient populations. [9, 36]
SMT is used to treat mechanical aspects of pain, specifically in areas of hypomobility. [37] With a decreased water content in degenerative IVDs, it is theorized that motion segments become unstable or hypermobile. However, there is much literature supporting the idea that the IVD may become stiffer due to the changes in AF collagen. [32, 38] This has significant implications for treatment. Improving motion segment mobility may provide better outcomes than surgical fusion in the properly selected patient. In the presented case, Cox flexion-distraction SMT was utilized. Flexion-distraction SMT employs a slow manual traction and mobilization of spinal motion segments. [39] The outcomes of flexion-distraction SMT include increased mobility and a decrease in perceived pain. Choi et al evaluated the biomechanical aspect of flexion-distraction SMT on the IVD and concluded it also results in increased imbibition, movement of metabolites into the IVD, and increased IVD height. [37]
In the appropriate patient, surgical treatment of specific lumbar segments can be beneficial. Gornet et al showed a significant impact on surgical outcomes in DLBP cases when the correct pathoanatomic target was identified. In their study, Gornet et al used MRS and NOCISCAN technology to identify painful IVDs. The outcomes were better in the group who had surgical treatment of only the specific levels identified as painful by NOCISCAN in comparison to the group whose surgical treatment included IVDs NOCISCAN identified as non-painful. [19] The lack of specificity of the correct pathoanatomic target may explain why outcomes of surgical intervention for DLBP have a low success rate.
In this case, post-treatment MRS and NOCISCORE results showed an improvement in structural integrity and biochemical changes. These results suggest that conservative management can improve those factors of IVD degeneration and DLBP. The mechanisms to explain why SMT may improve DLBP are variable. It may be as simple as increasing motion at those segments and improving imbibition to restore IVD homeostasis. Another area of study, mechanobiology, proposes it is mechanotransduction and cellular response to mechanical load.
Mechanotransduction is the process cells use to detect and respond to mechanical signals and may be the mechanism underlying clinical benefits of SMT. [40] There is no evidence to support spontaneous regeneration of the IVD. However, the notochordal cells within the NP from which proteoglycans are derived, have in animal models shown to differentiate into mature NP cells under mechanical stimulation. [29, 32] Ex vivo studies have demonstrated that both NP cells and AF cells have a strong direct response to external mechanical stimuli. The response is dose dependent with low magnitude and moderate frequency tensile load promoting proteoglycan production. In contrast to therapeutic effects, elevated magnitude and frequency tensile or compressive loads are catabolic, decreasing proteoglycan production and activating metalloproteinases. [29, 38, 40] More research in this field mayhelp develop appropriate load dose in conservative management of DLBP.
Case report results cannot be generalized to a larger population. However, this case report was heavily influenced by innovative work done by Gornet et al who showed MRS and NOCISCAN technology can identify painful IVDs across a larger sample of patients with DLBP. As this is the first case where posttreatment MRS was performed in a conservatively treated patient, a larger study is needed to validate the results.
Conclusion
We present the first utilization of magnetic resonance spectroscopy (MRS) in identifying the intervertebral disc (IVD) in a conservatively managed patient to confirm a pathoanatomic pain source as the IVD. Pre- and post-treatment IVD structural integrity and biochemical pain profile was quantified. The findings of this case suggest Cox flexion-distraction SMT in discogenic low back pain (DLBP) management may improve the IVD structural integrity and diminish pain biochemistry. The structural and biochemical contributors to DLBP are complex and more research may provide more understanding.
Authors Contributions:
All listed authors provided substantial contribution to the conception and/or design of the manuscript. JFB drafted the work with critical appraisal and revisions provided by the remaining authors. EER managed all patient interactions and correspondence as well as significantly contributed to the case report section of the manuscript. MFG, FK, and NK provided significant contribution to the conception and design of this work. KB and JC provided substantial revisions to the work. All authors have approved the final version of the manuscript and agree to be personally accountable to their contributions. Each author agrees that questions related to the accuracy or integrity of this work will be appropriately investigated, resolved, and resolution documented in the literature.
Funding:
No funding was received for this manuscript.
Conflicts of interest/Competing interests:
Erika Evans Roland, Matthew F Gornet, and Francine Schranck disclose ownership of stock in Aclarion.
Ethics:
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, and with the 1964 Helsinki declaration, and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
References:
Balagué F, Mannion AF, Pellisé F, Cedraschi C.
Non-specific low back pain.
Lancet. 2012 Feb 4;379(9814):482-91.
doi: 10.1016/S0140-6736(11)60610-7Fatoye F, Gebrye T, Odeyemi I.
Real-world incidence and prevalence of low back pain
using routinely collected data.
Rheumatol Int. 2019 Apr;39(4):619-626.
doi: 10.1007/s00296-019-04273-0Geurts JW, Willems PC, Kallewaard JW, van Kleef M, Dirksen C.
The Impact of Chronic Discogenic Low Back Pain: Costs and Patients' Burden.
Pain Res Manag. 2018 Oct 1;2018:4696180.
doi:10.1155/2018/4696180Brayda-Bruno M, Tibiletti M, Ito K, Fairbank J, Galbusera F, Zerbi A, Roberts S.
Advances in the diagnosis of degenerated lumbar discs
and their possible clinical application.
Eur Spine J. 2014 Jun;23 Suppl 3:S315-23.
doi: 10.1007/s00586-013-2960-9Chou R, Baisden J, Carragee EJ, Resnick DK, Shaffer WO, Loeser JD (2009)
The Global Spine Care Initiative: A Narrative Review of Psychological
and Social Issues in Back Pain in Low- and Middle-income Communities
European Spine Journal 2018 (Sep); 27 (Suppl 6): 823–837Fujii K, Yamazaki M, Kang JD, Risbud MV, Cho SK, Qureshi SA, Hecht AC, Iatridis JC.
Discogenic Back Pain: Literature Review of Definition, Diagnosis, and Treatment.
JBMR Plus. 2019 Mar 4;3(5):e10180.
doi: 10.1002/jbm4.10180Tieppo Francio V, Sherwood D, Twohey E, Barndt B, Pagan-Rosado R, Eubanks J, Sayed D.
Developments in Minimally Invasive Surgical Options for Vertebral Pain:
Basivertebral Nerve Ablation - A Narrative Review.
J Pain Res. 2021 Jun 23;14:1887-1907.
doi: 10.2147/JPR.S287275Ohtori S, Inoue G, Miyagi M, Takahashi K.
Pathomechanisms of discogenic low back pain in humans and animal models.
Spine J. 2015 Jun 1;15(6):1347-55.
doi: 10.1016/j.spinee.2013.07.490Chiarotto A, Koes BW.
Nonspecific Low Back Pain.
N Engl J Med. 2022 May 5;386(18):1732-1740.
doi: 10.1056/NEJMcp2032396Hancock MJ, Maher CG, Latimer J, Spindler MF, McAuley JH, Laslett M, Bogduk N.
Systematic review of tests to identify the disc, SIJ or facet joint as the source of low back pain.
Eur Spine J. 2007 Oct;16(10):1539-50.
doi: 10.1007/s00586-007-0391-1Han CS, Hancock PM, Maher PC.
Reconsidering non-specific low back pain:
Where to from here?
Spine J. 2022 Aug 6:S1529-9430(22)00840-3.
doi: 10.1016/j.spinee.2022.08.001Koes BW, van Tulder MW, Thomas S.
Diagnosis and Treatment of Low Back Pain
British Medical Journal 2006 (Jun 17); 332 (7555): 1430–1434Manchikanti L, Glaser SE, Wolfer L, Derby R, Cohen SP.
Systematic review of lumbar discography as a diagnostic test
for chronic low back pain.
Pain Physician. 2009 May-Jun;12(3):541-59Cuellar JM, Stauff MP, Herzog RJ, Carrino JA, Baker GA, Carragee EJ.
Does provocative discography cause clinically important injury to
the lumbar intervertebral disc? A 10-year matched cohort study.
Spine J. 2016 Mar;16(3):273-80.
doi: 10.1016/j.spinee.2015.06.051Tamagawa S, Sakai D, Nojiri H, Sato M, Ishijima M, Watanabe M.
Imaging Evaluation of Intervertebral Disc Degeneration and
Painful Discs-Advances and Challenges in Quantitative MRI.
Diagnostics (Basel). 2022 Mar 14;12(3):707.
doi: 10.3390/diagnostics12030707Zuo J, Joseph GB, Li X, Link TM, Hu SS, Berven SH, Kurhanewitz J, Majumdar S.
In vivo intervertebral disc characterization using magnetic resonance
spectroscopy and T1? imaging: association with discography and
Oswestry Disability Index and Short Form-36 Health Survey.
Spine (Phila Pa 1976). 2012 Feb 1;37(3):214-21.
doi: 10.1097/BRS.0b013e3182294a63Boisson M, Lefčvre-Colau MM, Rannou F, Nguyen C.
Active discopathy: a clinical reality.
RMD Open. 2018 Apr 17;4(1):e000660.
doi: 10.1136/rmdopen-2018-000660Splendiani A, Bruno F, Marsecano C, Arrigoni F, Di Cesare E, Barile A, Masciocchi C.
Modic I changes size increase from supine to standing MRI correlates
with increase in pain intensity in standing position: uncovering
the "biomechanical stress" and "active discopathy" theories
in low back pain.
Eur Spine J. 2019 May;28(5):983-992.
doi: 10.1007/s00586-019-05974-7Gornet MG, Peacock J, Claude J, Schranck FW, Copay AG, Eastlack RK, Benz R, Olshen A, Lotz JC.
Magnetic resonance spectroscopy (MRS) can identify painful lumbar
discs and may facilitate improved clinical outcomes of
lumbar surgeries for discogenic pain.
Eur Spine J. 2019 Apr;28(4):674-687.
doi: 10.1007/s00586-018-05873-3Dudli S, Fields AJ, Samartzis D, Karppinen J, Lotz JC.
Pathobiology of Modic changes.
Eur Spine J. 2016 Nov;25(11):3723-3734.
doi: 10.1007/s00586-016-4459-7Fields AJ, Ballatori A, Liebenberg EC, Lotz JC.
Contribution of the endplates to disc degeneration.
Curr Mol Biol Rep. 2018 Dec;4(4):151-160.
doi: 10.1007/s40610-018-0105-yKeshari KR, Lotz JC, Link TM, Hu S, Majumdar S, Kurhanewicz J.
Lactic acid and proteoglycans as metabolic markers
for discogenic back pain.
Spine (Phila Pa 1976). 2008 Feb 1;33(3):312-7.
doi:10.1097/BRS.0b013e31816201c3Zuo J, Saadat E, Romero A, Loo K, Li X, Link TM, Kurhanewicz J, Majumdar S.
Assessment of intervertebral disc degeneration with
magnetic resonance single-voxel spectroscopy.
Magn Reson Med. 2009 Nov;62(5):1140-6.
doi: 10.1002/mrm.22093Wang AM, Cao P, Yee A, Chan D, Wu EX.
Detection of extracellular matrix degradation in intervertebral
disc degeneration by diffusion magnetic resonance spectroscopy.
Magn Reson Med. 2015 May;73(5):1703-12.
doi: 10.1002/mrm.25289Mu X, Peng W, Ou Y, Li P, Li Z, Wei J.
Non-surgical therapy for the treatment of chronic low back pain in
patients with Modic changes: A systematic review of the literature.
Heliyon. 2022 Jun 30;8(7):e09658.
doi: 10.1016/j.heliyon.2022.e09658Tagliaferri SD, Miller CT, Ford JJ, Hahne AJ, Main LC, Rantalainen T, Connell DA.
Randomized Trial of General Strength and Conditioning Versus Motor
Control and Manual Therapy for Chronic Low Back Pain
on Physical and Self-Report Outcomes.
J Clin Med. 2020 Jun 3;9(6):1726.
doi: 10.3390/jcm9061726Whedon JM, Kizhakkeveettil A, Toler AW, Bezdjian S, Rossi D, Uptmor S,
MacKenzie TA, Lurie JD, Hurwitz EL, Coulter I, Haldeman S.
Initial Choice of Spinal Manipulation Reduces Escalation of Care
for Chronic Low Back Pain Among Older Medicare Beneficiaries
Spine (Phila Pa 1976) 2022 (Feb 15); 47 (4): E142–E148Hwang D, Kim S, Abeydeera NA, Statum S, Masuda K, Chung CB, Siriwanarangsun P, Bae WC.
Quantitative magnetic resonance imaging of the lumbar intervertebral discs.
Quant Imaging Med Surg. 2016 Dec;6(6):744-755.
doi: 10.21037/qims.2016.12.09Fearing BV, Hernandez PA, Setton LA, Chahine NO.
Mechanotransduction and cell biomechanics of the intervertebral disc.
JOR Spine. 2018 Sep;1(3):e1026.
doi: 10.1002/jsp2.1026Theodore N.
Degenerative Cervical Spondylosis.
N Engl J Med. 2020 Jul 9;383(2):159-168.
doi:10.1056/NEJMra2003558Lyu FJ, Cui H, Pan H, Mc Cheung K, Cao X, Iatridis JC, Zheng Z.
Painful intervertebral disc degeneration and inflammation:
from laboratory evidence to clinical interventions.
Bone Res. 2021 Jan 29;9(1):7.
doi: 10.1038/s41413-020-00125-xIto K, Creemers L.
Mechanisms of intervertebral disk degeneration/injury and pain: a review.
Global Spine J. 2013 Jun;3(3):145-52.
doi: 10.1055/s-0033-1347300Immke DC, McCleskey EW.
Lactate enhances the acid-sensing Na+ channel on ischemia-sensing neurons.
Nat Neurosci. 2001 Sep;4(9):869-70.
doi: 10.1038/nn0901-869Gaillard F, Yap J, Saber M, et al.
MR spectroscopy.
Reference article, Radiopaedia.org (Accessed on 15 Feb 2023)
https://doi.org/10.53347/rID-1630Zgierska AE, Burzinski CA, Garland EL, Lennon RP, Jamison R, Nakamura Y, Barrett B.
Mindfulness-based therapy compared to cognitive behavioral therapy
for opioid-treated chronic low back pain: Protocol for
a pragmatic randomized controlled trial.
Contemp Clin Trials. 2021 Nov;110:106548.
doi:10.1016/j.cct.2021.106548Paige NM, Miake-Lye IM, Booth MS, Beroes JM, Mardian AS, Dougherty P.
Association of Spinal Manipulative Therapy With Clinical Benefit and
Harm for Acute Low Back Pain: Systematic Review and Meta-analysis
JAMA. 2017 (Apr 11); 317 (14): 1451–1460Choi J, Hwangbo G, Park J, Lee S.
The Effects of Manual Therapy Using Joint Mobilization and
Flexion-distraction Techniques on Chronic
Low Back Pain and Disc Heights.
J Phys Ther Sci. 2014 Aug;26(8):1259-62.
doi: 10.1589/jpts.26.1259Lotz JC, Hsieh AH, Walsh AL, Palmer EI, Chin JR.
Mechanobiology of the intervertebral disc.
Biochem Soc Trans. 2002 Nov;30(Pt 6):853-8.
doi: 10.1042/bst0300853Gudavalli MR, Cambron JA, McGregor M, Jedlicka J, Keenum M.
A randomized clinical trial and subgroup analysis to compare
flexion-distraction with active exercise for chronic low back pain.
Eur Spine J. 2006 Jul;15(7):1070-82.
doi: 10.1007/s00586-005-0021-8Molladavoodi S, McMorran J, Gregory D.
Mechanobiology of annulus fibrosus and nucleus pulposus cells in intervertebral discs.
Cell Tissue Res. 2020 Mar;379(3):429-444.
doi: 10.1007/s00441-019-03136-1
Return to LOW BACK PAIN
Return to DISC HERNIATION & CHIROPRACTIC
Since 4-10-2023


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |